Main Body
Chapter 7
Topic 7 – Humidity
Water in its gaseous state, commonly called water vapour, is present in variable amounts from place to place in the atmosphere. The amount of water vapour in an air mass is called its ‘humidity’. Some air masses hold significant amounts of water vapour, whereas others are very dry. The amount of water vapour an air mass holds has significant implications for water delivery to Earth’s continents, without which the ecological systems that exist on the continents would fail. Here is a water vapour interactive exercise and further explanation: https://earthobservatory.nasa.gov/global-maps/MYDAL2_M_SKY_WV
There are three basic ways in which we define humidity:
- Absolute humidity – mass of water per volume of air (usually in g/m3)
- Specific humidity – mass of water per mass of air (usually in g/kg)
- Relative humidity – ratio of water vapour compared to potential capacity of air mass to hold water vapour (usually expressed as %)
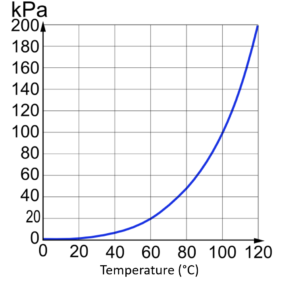
Each method has its own uses, but we will focus on relative humidity as it is most useful in considering weather patterns. In order to fully understand humidity, there is another concept useful to consider: vapour pressure. Earlier in these readings we discussed air pressure. Air pressure is the result of air molecules pushing outwards on a surface as they move in a system. Similarly, vapour pressure is the result of water vapour molecules pushing outwards on a surface. At sea level, the pressure an air mass exerts is a combination of the air molecules and water vapour molecules pushing outwards in the system. If you imagine a cubic metre of air, the more water vapour molecules you squeeze into that cubic metre, the higher the vapour pressure. In the critical zone, water is squeezed into air masses through the process of evaporation. An increase in vapour pressure corresponds to an increase in humidity. But air masses have a limit on the amount of vapour pressure they can sustain before water vapour is forced back out into liquid form through condensation. This limit is called the saturation vapour pressure . When an air mass reaches saturation vapour pressure, no more water vapour can be forced into it through evaporation without a corresponding amount being released through condensation. Relative humidity measures how close an air mass is to saturation vapour pressure. Further reading on saturation pressure here: https://www.chemguide.co.uk/physical/phaseeqia/vapourpress.html
![]()
Critical to understanding saturation vapour pressure is that it is dependent on temperature. The higher the temperature, the higher the saturation vapour pressure. In other words, the warmer the air mass, the more water molecules it can hold.
Imagine a cubic metre of air that is at 100% relative humidity (that is, it is at its saturation vapour pressure). According to the above statement on the relationship of temperature and saturation vapour pressure, you could create condensation from that air mass in two ways:
- Evaporate more water into the air mass, causing some to be forced out as condensation because it is already at capacity.
- Without adding more water vapour to the system, simply reduce the temperature of the air mass, decreasing the saturation vapour pressure, and forcing some of the water vapour to condense.
You can see from the above example, that relative humidity and temperature have an inverse relationship . Without the addition or subtraction of water vapour to a system, if you increase the temperature, the capacity to hold moisture increases, and the relative humidity goes down. If you decrease the temperature, the capacity to hold moisture decreases and the relative humidity goes up.
When you apply this principle to the atmosphere you find that a change in temperature is enough to force condensation to occur, resulting in precipitation. The temperature at which an air mass will reach saturation (or 100% relative humidity) is called the dew point temperature.
You can probably think of several ways in which an air mass could be forced to reach its dew point temperature. Over the course of a 24 hour day, an air mass will go through big temperature swings as it is warmed by the sun, and cools at night. These temperature differences may be enough for it to reach its dew point. Alternatively air masses may be forced to rise through the atmosphere as they encounter low pressure, or are forced above topographic obstacles, like mountain ranges. In the troposphere, this change in elevation usually comes with a decrease in temperature, pushing air masses towards their dew point. The elevation at which the dew point is reached is referred to as the lifting condensation level. The rate at which temperature change occurs in the atmosphere with elevation change is called the lapse rate. There are three significant atmospheric lapse rates to be aware of:
- Environmental lapse rate (ELR): the actual, measured change in temperature as one moves upward through the atmosphere at specific location. Usually about 6.5°C/1000 m, but variable.
- Dry adiabatic lapse rate (DAR): the average rate at which an unsaturated (<100% humidity) air mass will cool as it rises through the troposphere. 10°C/1000 m
- Moist adiabatic lapse rate (MAR): the average rate at which a saturated (100% humidity) air mass will cool as it rises through the troposphere. 6°C/1000 m
If an air mass encounters environmental conditions that are cooler than it is, it will rise because it is less dense. If the air mass is unsaturated, it will rise at the dry adiabatic lapse rate, until it reaches the lifting condensation level, at which point it will rise at the moist adiabatic lapse rate.
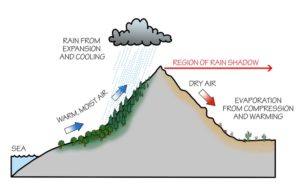
This concept leads into a significant geographic pattern in mountainous environments. Where air masses are forced to rise up and over mountain ranges, they often reach their lifting condensation level, and water vapour is forced out of the air mass. As they reach and pass over the mountain range, they descend the other side, but because they have condensed much of their water vapour, they are no longer saturated and descend at the dry adiabatic lapse rate, creating the opportunity for little precipitation on the far side of the mountain range. This process is called the rain-shadow effect and can be used to explain drier climates located behind topographic barriers like mountain ranges.
the gaseous phase of water (Wiki)
water in its gaseous state-instead of liquid or solid (ice) (weatherstreet)
Generally, some measure of the water vapor content of air (AMS)
A general term used to describe the amount of water vapor found in the atmosphere (PhysGeo.net)
The ratio of the vapor pressure to the saturation vapor pressure with respect to water (AMS)
Pressure exerted by water vapor molecules in a given quantity of atmosphere (PhysGeo.net)
The pressure exerted by the molecules of a given vapour (AMS)
The vapor pressure of a system, at a given temperature, for which the vapor of a substance is in equilibrium with a plane surface of that substance's pure liquid or solid phase (AMS)
The vapor pressure at which the gaseous phase of a substance can coexist with the liquid or solid phase in equilibrium at a given temperature (Wiki)
Is any aqueous deposit, in liquid or solid form, that develops in a saturated atmosphere (relative humidity equals 100%) and falls to the ground generally from clouds (PhysGeo)
- movement of water from the atmosphere to the earth’s surface (ItsEdu)
All liquid or solid phase aqueous particles that originate in the atmosphere and fall to the earth's surface (AMS)
the temperature at which water vapor saturates from an air mass into liquid or solid usually forming rain, snow, frost or dew (PhysGeo)
temperature at which air becomes saturated i.e. relative humidity is 100% (ItsEdu)
The level at which a parcel of moist air lifted dry-adiabatically would become saturated (AMS)
The rate of decrease of temperature with elevation, -∂T/∂z, or occasionally ∂T/∂p, where p is pressure (AMS)
change in temperature with altitude above a particular point at a particular time. Average is 6.5 degrees C per 1000m. (ItsEdu)
The rate of air temperature increase or decrease with altitude. The average ELR in the troposphere is an air temperature decrease of 6.5° Celsius per 1000 meters rise in elevation (PhysGeo)
the rate of fall in air temperature by adiabatic change as unsaturated air gains altitude. Approximately 1 degree C per 100m. (ItsEdu)
process lapse rate of temperature, the rate of decrease of temperature with height of a parcel of dry air lifted by a reversible adiabatic process through an atmosphere in hydrostatic equilibrium (AMS)
The rate of decline in the temperature of a rising parcel of air after it has reached saturation. This rate is less than the dry adiabatic lapse rate (9.8° Celsius per 1000 meters) because of the heat energy added to the ascending air parcel from condensation and deposition processes (PhysGeo)
The rate of decrease of temperature with height along a moist adiabat (AMS)
Reduction of precipitation commonly found on the leeward side of a mountain (PhysGeo)
the area on the leeward side of a mountain range which experiences drier conditions than the windward side. This is due to the air being drier as it has experienced condensation and precipitation on the windward side, plus the fact that the air will be sinking and therefore warming meaning relative humidity is falling and condensation cannot occur (ItsEdu)
A region of sharply reduced precipitation on the lee side of an orographic barrier, as compared with regions upwind of the barrier (AMS)
