Chapter IV: Introduction to Leaching
4. Overview of Types of Leaching Methods
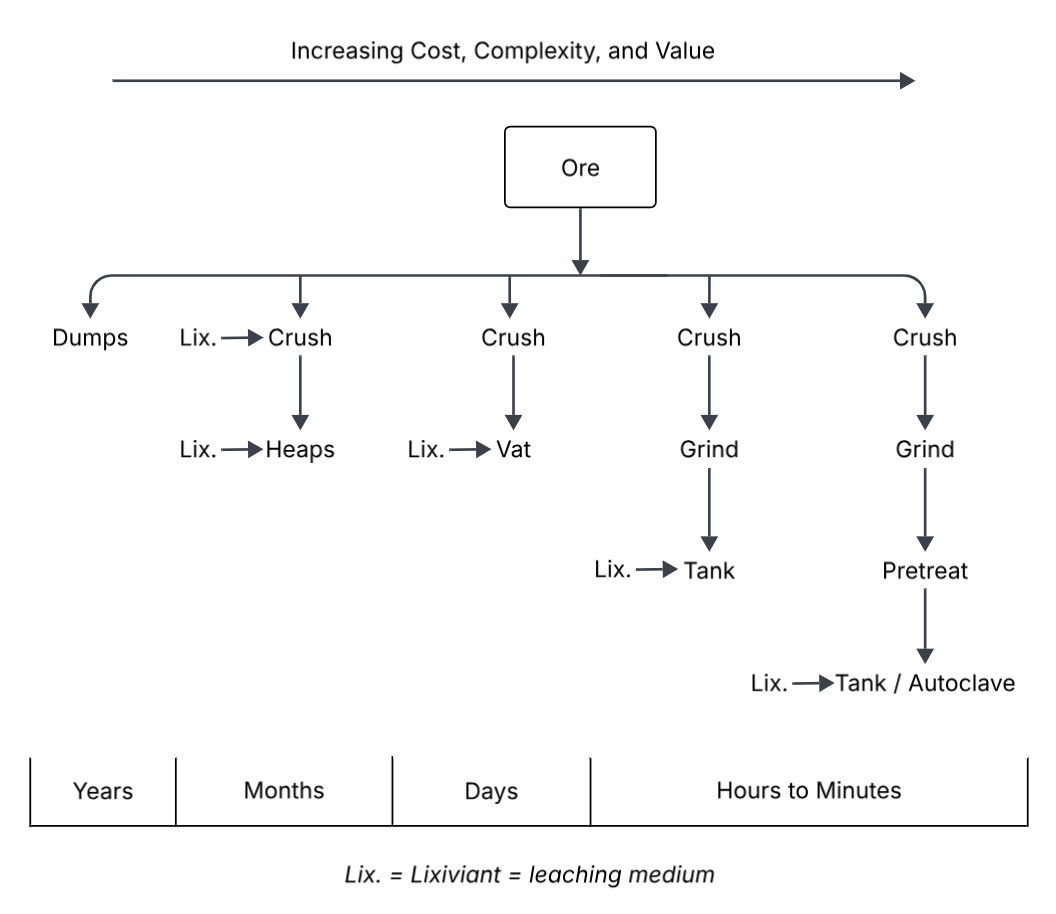
There are many different types or categories of leaching. Figure 4.1 below provides a comparison of some of the common types. The boxes in the figure indicate specific types of steps involved in the process. Obviously complexity and costs increase with the number of discrete steps involved. The value contained in the ore based on the minerals present (actually, the fraction thereof that can be extracted!) must be high enough to justify the effort and cost to be expended. From an economic point of view, it is the $/tonne value of the ore that must be considered. A gold ore containing only 10 g Au/tonne of ore (10 ppm) might be worth running the most involved kind of process. A copper ore containing 0.5% Cu (5000 ppm) may warrant no more cost and effort than a heap leach.
| Table 4.1 - Summary of common leaching processes. | |||||
|---|---|---|---|---|---|
| Feed | Solution | Oxidant | Temp. | Pressure | Equipment/Method |
| Oxidized Cu ore (oxides etc.) | Dil. H2SO4 | None | Ambient | Atmospheric | Heaps |
| Sulfide Cu ore | Dil. H2SO4 | Ferric, bacteria | Ambient | Atmospheric | Heaps |
| Cu,Ni,Co,Zn concentrates/mattes | Dil. H2SO4 | Ferric, oxygen | ~100 or >150°C | 1-10 bar | Tanks, autoclaves |
| Zinc calcine (ZnO) | Dil. H2SO4 | None | 40-90°C | Atmospheric | Tanks |
| Ni matte | Dil. HCl | Ferric, cupric | <100°C | Atmospheric | Tanks |
| Ni laterite ore | Dil. H2SO4 | None | ~250°C | ~50 bar | Autoclaves |
| Reduced laterite ore | NH3/CO2 | Air, Cupric | 40-80°C | Atmospheric | Tanks |
| Uranium ore | Dil. H2SO4 | Ferric + MnO2 | 40-50°C | Atmospheric | Tanks, in-situ |
| Oxidized gold ore | NaCN | Air | Ambient | Atmospheric | Heaps, tanks |
| Refractory gold ore, concentrates | 1 H2SO4, then: | 1 Ferric, then: | 1 150-200°C, then: | 1 30-40 bar, then | 1 Autoclaves, then |
| 2 NaCN | 2 air | 2 ambient | 2 atmospheric | 2 tanks | |
| Bauxite i.e. Al(O)OH | NaOH | None | 150-200°C | 20-40 bar | Autoclaves |
| Roasted vanadium ore | Water | None | 50-90°C | Atmospheric | Tanks, columns |
| Scheelite, i.e. CaWO4 | Soda ash (Na2CO3) | None | 200°C | 40 bar | Autoclaves |
Note that when we specify an ore grade (lie 0.5% Cu), we mean 0.5% by weight, that is 5000 g Cu/tonne of ore or 5 kg/tonne. What this does not specify is the mineral form in which the Cu resides. This causes no end of confusion. To clarify:
\[5000\;\text{g Cu/t ore} \times \frac{1\;\text{mol Cu}}{63.546\;\text{g Cu}} \times \frac{1\;\text{mol CuFeS}_2}{1\;\text{mol Cu}} \times \frac{185.513\;\text{g CuFeS}_2}{\text{mol CuFeS}_2} = 14{,}439\;\text{g CuFeS}_2/\text{t ore}\]
\[= 1.44\%\;\text{CuFeS}_2 \tag{21}\]
Recall that crushing and grinding are stages of size reduction for an ore; first it is crushed, and then ground where applicable. Lixiviant is an archaic term for leaching solution, and it is still commonly in use.
Media Attributions
- Ch4_F7_Leaching_Classes © Bé Wassink and Amir M. Dehkoda adapted by Jeno Hwang is licensed under a CC BY-NC (Attribution NonCommercial) license

