Chapter I: Introduction to Extractive Metallurgy
3. Natural Resources
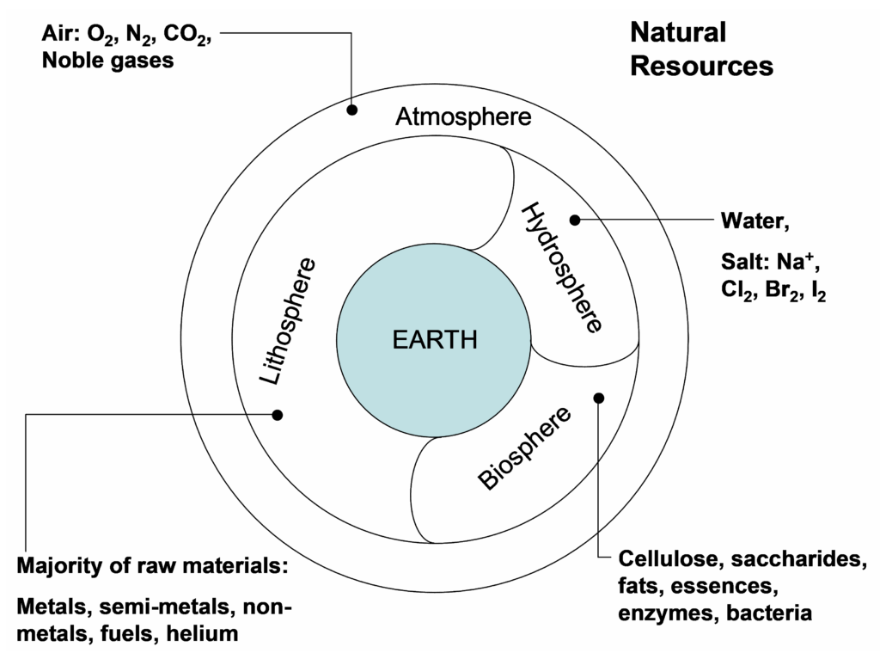
There are four main regions or "spheres" from which raw materials are taken (Figure 3.1). These are the lithosphere (crust), the biosphere (plants, animals, bacteria), the hydrosphere (oceans and freshwater) and the atmosphere. Metals extraction relies almost entirely on the lithosphere for metal bearing raw materials. However, the industry also relies heavily upon each of the other spheres: the hydrosphere for water; the atmosphere for air, or oxygen (concentrated from the air), and; the biosphere, e.g. animal proteins and carbohydrates as additives in metal processing and bacteria used in biological leaching of sulfide ores. It is intriguing to ponder the astounding differentiation between metal compounds in the earth's crust and the oceans. Only three metal ions are found in the oceans at any appreciable concentration: Na+, 0.47%; Mg+2 0.053%, and; Ca+2, 0.010%. Some very valuable metals occur at very low concentrations in the oceans, e.g. gold at ~1 μg/L (~1 part per billion), but as yet there are no economic methods of extracting these. Note too that the lithosphere includes that roughly ¾ of the Earth’s surface which is covered by the oceans. This is largely unexplored. There is interest in this, but land-based mineral resources are still plentiful enough and economically much more viable. Oil and gas are notable exceptions.
How minerals are distributed in the earth's crust is crucially important in the business of metal extraction. There are three aspects to this. One is the natural abundance of the element in the Earth’s crust. This is the average concentration in the lithosphere. A second is the occurrence of mineral deposits of the elements. Often these contain the pertinent minerals at considerably higher concentration than the average in the crust overall. It is fortunate that the elements are often not uniformly distributed. (Some elements are more evenly dispersed on average.) The third factor is that the mineral deposits must be able to be economically produced. The metals need to be present at suitable concentrations and in mineral forms that can be processed. Numerous factors go into the economic viability of a mineral deposit. To illustrate, copper has a natural abundance of 0.0068% by weight (68 g Cu per tonne of rock). But an economically producible copper mineral deposit would have contained around 1.8% Cu 100 years ago, whereas today a deposit with as little as 0.5% Cu could be economic to produce.
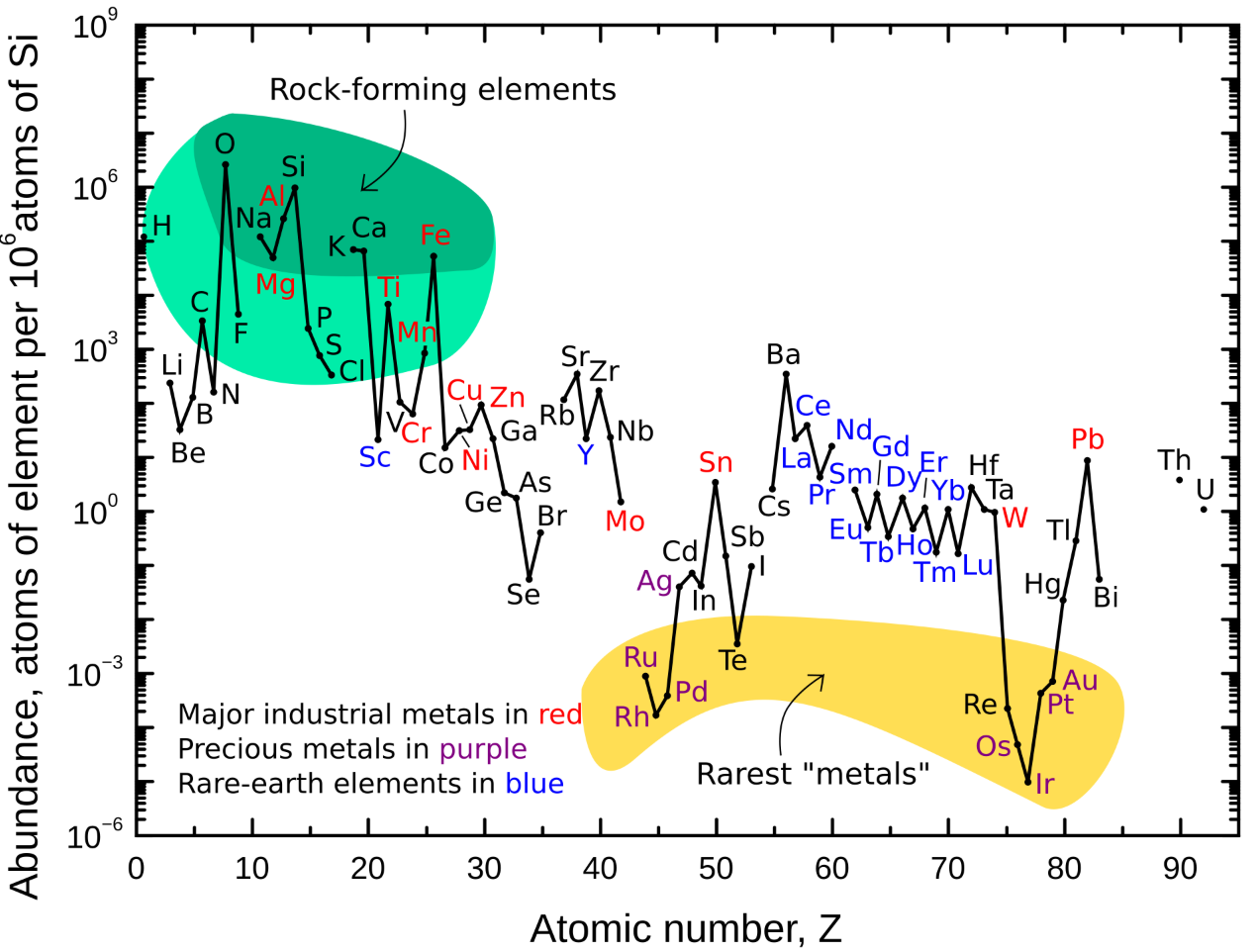
Table 3.1 shows the natural abundances of many commercially important metals and elements in the lithosphere. Most of the Earth’s crust is composed of oxygen (45.5%), silicon (27.2%), aluminum (8.3%), iron (6.2%), calcium, magnesium, sodium and potassium; these 8 elements comprise about 99% of the earth's crust! Most of the transition metals and the other heavier metals are either rare or very rare, with a few exceptions (iron, titanium and manganese - among the earlier of the transition metals). Natural abundances are also illustrated in the form of a periodic table in Figure 3.2. Note that often the lighter elements are the more common, while the heaviest transition metals are relatively rare. Indeed the platinum group metals, gold and silver are among the very rarest elements. There are exceptions to the general rule. The lanthanides (also called the rare earths) are not actually all that rare, compared to some of the less abundant transition metals.
As mentioned previously, many elements are concentrated in mineral deposits. The major types of minerals are listed in Table 3.2. Where a mineral is a compound, it is commonly thought of as formally a combination of cations and anions. However, the nature of the bonds between the atoms is often a lot more complex than this. Components of ionic, covalent and metallic bonds, as well as weaker bonding interactions, like hydrogen bonding, may be involved. Hence one usually cannot regard minerals as actually being salts. PbS, for example, is formally comprised of Pb+2 and S2-, but in reality it is not composed of discrete cations and anions.
| Table 3.1 - Natural Abundances in Percent by Weight of Many Commercially Important Metals and Some Non-metals in the Earth’s Crust | ||||
|---|---|---|---|---|
| Li | Alkali Metals | Ni | 0.099 | Transition Metals |
| Na | Pd | 1.5x10-6 | ||
| K | Pt | 1x10-6 | ||
| Be | Alkaline Earths | Cu | 0.0068 | |
| Mg | Ag | 8x10-6 | ||
| Ca | Au | 4x10-7 | ||
| Sc | Transition Metals | Zn | 0.0076 | |
| Y | Cd | 1.6x10-5 | ||
| La | Hg | 8x10-6 | ||
| Ti | Al | 8.3 | Groups III-VI | |
| Zr | Ga | 0.0019 | ||
| Hf | In | 2.4x10-5 | ||
| V | Tl | 7x10-5 | ||
| Nb | Sn | 0.00021 | ||
| Ta | Pb | 0.0013 | ||
| Cr | Bi | 8x10-7 | ||
| Mo | Lanthanides | 8-660x10-5 | ||
| W | Th | 0.00081 | Actinides | |
| Mn | U | 0.00023 | ||
| Re | Si | 27.2 | Semiconductors | |
| Fe | Ge | 0.00015 | ||
| Ru | As | 0.00018 | ||
| Os | Sb | 2x10-5 | ||
| Co | Te | 1x10-7 | ||
| Rh | Se | 5x10-6 | ||
| Ir | S | 0.034 | Non-metals | |
| Table 3.2 - Classification of Minerals According to their Formally Anionic components. | |
|---|---|
| Anion | |
| Sulfides | S2-, S22- (i.e. a polysulfide) |
| Selenides, tellurides | Se2-, Te2- (rarer than sulfides) |
| Oxides/hydroxides | O2-/OH-(common) |
| Halides | Cl-, F-; rarely Br-, I- |
| Carbonates | CO32- |
| Sulfates | SO42- |
| Nitrates | NO3- |
| Phosphates | PO43- |
| Arsenates | AsO43- |
| Silicates | SiO44-, Si2O76-, many others (common) |
| Aluminosilicates | [Alw(OH)xSiyOz]n- (numerous) |
| Chromates | CrO42- |
| Molybdates | MoO42- |
| Tungstates | WO42- |
| Vanadates | VO43- |
| Borates | B(OH)4-, B4O5(OH)42-, etc. )others |
The formalism is useful for classification purposes and for understanding the chemistry. Minerals are usually classified by the formally anionic components since there are many metals that form precipitates with the same anion, for instance there are many metal sulfides, such as Cu2S, CuFeS2, PbS, ZnS, etc. In many cases the metal values reside in the formally cationic parts of the minerals, e.g. CuS (Cu+2/S2-), AlO(OH) (Al+3/O2-,OH-), and PbCO3 (Pb+2/CO32-). But in some cases the metal of interest resides in the formally anionic part as complex ions, e.g. CaWO4 (Ca+2/WO42-). Oxides/hydroxides, silicates/aluminosilicates and sulfides are the most commonly mined minerals. (There are also other classes of minerals which, in the context of metal production, are not commonly produced.)
A sample list of some of the more important minerals is shown below in Table 3.3. Some rare and valuable metals may be disseminated in a variety of mineral matrices, for example gold may be hosted in quartz or pyrite or arsenopyrite etc. In some instances there are now few available ores of some metals (e.g. silver). In such cases the main means of production is as a by-product of another metal’s production. A partial list of applicable leaching systems for treating the mineral, or conditions under which it may at least partially be dissolved are also indicated in the table. Note the preponderance of acid, most often sulfuric acid (the cheapest of the “mineral acids”).
There are vastly more minerals than listed here, but this gives an indication of some of the more important ones. New minerals are still being discovered. Natural deposits may contain a diverse mixture of minerals, some at very low concentrations, which may have an impact on the extraction process. Thorough study of the mineralogy of an ore is crucial to a successful process. Many a plant has been doomed due to inattention to mineralogy.
In addition to minerals that contain metals of interest, ores inevitably contain gangue minerals. These have no commercial value in the context of the metal extraction process of interest. Very often the gangue constitutes by far the major fraction of an ore. This does not mean that some of the associated metals are not valuable. But, they cannot be economically processed from the given ore. For example, pyrite contains iron, the basis for steel, but there is not economically viable means to make iron from pyrite processed in the context of hydrometallurgy. Common gangue minerals and their reactivity are listed in Table 3.4.
Gangue minerals require careful attention. They may cause excessive consumption of costly reagents. They may host valuable minerals (e.g. fine crystals within gangue minerals, or chemical substituted within the gangue crystals, e.g. Ni in nickeliferrous limonite, Table 3.3). Gangue minerals may cause solid-liquid separation problems, e.g. clay minerals.
| Table 3.3. - Common and important minerals of various metals. | |||
|---|---|---|---|
| Metal | Mineral | Formula | Principal leaching chemistry |
| Al | Gibbsite | Al(OH)3 | Caustic (NaOH) |
| Boehmite | AlO(OH) | NaOH; less reactive than diaspore | |
| Diaspore | AlO(OH) | NaOH; less reactive than gibbsite | |
| Bauxite | Mixtures of above 3 | ||
| Corundum | Al2O3 | NaOH; less reactive than boehmite | |
| Cu | Chalcopyrite | CuFeS2 Main Cu mineral | HCl/oxidant, H2SO4/oxidant. Mainly treated by pyrometallurgy. |
| Cu2S | H2SO4/oxidant | ||
| Chalcocite | CuS | H2SO4/oxidant | |
| Covellite | Cu5FeS4 | H2SO4/oxidant | |
| Bornite | Cu2O | H2SO4/oxidant | |
| Cuprite | Cu metal (rare) | H2SO4/oxidant; NH3/O2 | |
| Native Cu | CuCO3·Cu(OH)2 | Dilute H2SO4 | |
| Malachite | 2CuCO3·Cu(OH)2 | Dilute H2SO4 | |
| Azurite | CuO | Dilute H2SO4 | |
| Tenorite | CuSiO3·2H2O | Dilute H2SO4 | |
| Chrysocolla | 3CuO·CuCl2·3H2O | Dilute H2SO4 | |
| Atacamite | Cu3AsS4 | Difficult to leach or smelt | |
| Enargite | Cu3SbS3 | Difficult to leach or smelt | |
| Tetrahedrite | |||
| Zn | Sphalerite | ZnS Main | H2SO4/oxidant |
| Marmatite | (Zn,Fe)S minerals | H2SO4/oxidant | |
| Zincite | ZnO (uncommon mineral; roasting ZnSZnO; common) | Dilute H2SO4 | |
| Fe | Magnetite | Fe3O4 | Dilute acid |
| Hematite | Fe2O3 | Unreactive | |
| Limonite | FeO(OH)·nH2O | Acid (limonite may bear Ni) | |
| Goethite | FeO(OH) | Acid (goethite may bear Ni) | |
| Siderite | FeCO3 | Dilute acid | |
| Pyrite | FeS2 (may bear Au) | H2SO4/oxidant | |
| Marcasite | FeS2 | H2SO4/oxidant | |
| Pyrrhotite | Fe1-xS, 1-x ~ 1 | Acid or acid/oxidant | |
| Arsenopyrite | FeAsS (may bear Au) | ||
| Pb | Galena | PbS Main mineral; usually smelted | HCl/aq. Cl- salt/oxidant |
| PbO | |||
| Litharge | PbSO4 | HCl/aq. Cl- salt; HNO3 | |
| Anglesite | PbCO3 | HCl/aq. Cl- salt | |
| Cerussite | HCl/aq. Cl- salt | ||
| Ni | Pentlandite | (Ni,Fe)9S8 | H2SO4 or HCl/oxidant |
| Garnierite | (Ni,Mg)6Si4O10(OH)8 | Strong H2SO4 | |
| Millerite | NiS | H2SO4 or HCl/oxidant; NH3/O2 | |
| Laterites, with | Complex oxides/silicates, including (Fe,Ni)O(OH)·nH2O | H2SO4; reductive roasting then leach with NH3/(NH4)2CO3 | |
| nickeliferrous | |||
| limonite | |||
| Co | Smaltite | CoAs2 | |
| Cobaltite | CoAsS | ||
| Linnaeite | Co3S4 | H2SO4 or HCl/oxidant | |
| Laterites | As per Ni | ||
| CoS | "Concentrate" formed from laterite leach; leach with NH3/O2 | ||
| Sn | Cassiterite | SnO2 | Mainly by pyrometallurgy |
| Ti | Ilmenite | FeTiO3 | Very strong acids |
| Rutile | TiO2 | Very strong acids | |
| Mo | Molybdenite | MoS2 | Mainly by pyrometallurgy |
| Molybdite | MoO3 | ||
| Wulfenite | PbMoO4 | ||
| Powellite | Ca(Mo,W)O4 | ||
| W | Scheelite | CaWO4 | HCl, H2SO4 |
| Wolframite | (Fe,Mn)WO4 | NaOH | |
| Sb | Stibnite | Sb2S3 | Generally undesirable impurities. May leach in acid/oxidant |
| Jamesonite | FePb4Sb6S14 | ||
| As | Realgar | As4S4 | Generally undesirable impurities. May leach in H2SO4/oxidant, cyanide/O2 systems |
| Orpiment | As2S3 | ||
| Bi | Bismite | Bi2O3 | Acid/chloride |
| Bismuthinite | Bi2S3 | Acid/oxidant | |
| Native Bi | Elemental Bi | Acid/oxidant | |
| Lanthanides (“Ln”) | Monazite | (La,Th,Ln)PO4 | Hot strong acid; NaOH |
| U | Pitchblende | UO2.x; | H2SO4/oxidant; Na2CO3/oxidant |
| x < 0.67 | |||
| Ag | Argentite | Ag2S | CN-/oxidant; S2O32-/oxidant |
| Cerargyrite | AgCl | NH3; CN- | |
| Native Ag | Ag metal | CN-/oxidant | |
| Au | Native Au | Au metal | CN-/oxidant; S2O32-/oxidant |
| Calaverite | AuTe2 | Hard to leach | |
| Sylvanite | (Au,Ag)Te2 | Hard to leach | |
| Hg | Cinnabar | HgS | Usually by pyrometallurgy |
| PGM’s | Various and complex | ||
| Table 3.4 - Common Gangue Minerals. Superscripted Roman numerals, e.g. MII, Refer to a Formally +2 Metal Cation. | ||
|---|---|---|
| Mineral | Composition | Reactivity (Partial or Total Dissolution) |
| Quartz | SiO2 (may host gold) | Fluorides or strong NaOH |
| Feldspars | M1-M2-Al-silicates M1, M2 = Na, K, Ca |
Relatively unreactive |
| Mica, Chlorite | K2Al4(Si6Al2)O20(OH)4 Mg10Al2(Si6Al2)O20(OH)16 |
Dilute acids |
| Amphiboles | MIIy-(Si4O11)(OH)x: many forms; MII = e.g. Ca, Mg, or both; includes asbestos |
Strong acids |
| Clays | Layered aluminosilicates | Acids |
| Pyroxene,
Olivine |
MIISiO3, (M1II,M2II)SiO3 MII, M1II, M2II = e.g. Ca, Mg (Mg,Fe)2SiO4 |
Acids |
| Garnets,
Staurolite, |
M3IIM2III(SiO4)3 MII = e.g. Ca, Mg, Fe; MIII = e.g. Al, Cr, Fe (FeII,Mg,Zn)2Al9(Si,Al)4O22(OH)2 Al2SiO5 |
Relatively unreactive |
| Andalusite, Sillimanite |
Al2SiO5 Al2SiO5 |
Acids |
| Calcite | CaCO3 | Readily with dilute acids |
| Magnesite, Dolomite |
MgCO3 CaMg(CO3)2 |
Acids |
| Pyrrhotite | Fe1-xS, 1-x ~ 1 | Dilute acids; CN-/O2 |
| Marcasite, Pyrite |
FeS2 FeS2 |
Acids/oxidants; CN-/O2 |
| Magnetite Limonite Goethite Hematite |
Fe3O4 FeO(OH)·nH2O FeO(OH) Fe2O3 |
Acids (rates depend Acids on particle Acids size and crystallinity.) Practically unreactive |
Media Attributions
- Ch1_F6_Terrestrial_sources_raw_materials © Bé Wassink and Amir M. Dehkoda is licensed under a CC BY-NC (Attribution NonCommercial) license
- Ch1_F7_Natural_abundance_elements © Bé Wassink and Amir M. Dehkoda is licensed under a CC BY-NC (Attribution NonCommercial) license
The earth's crustal rocks. The oceanic and non-oceanic lithospheres differ. In practice extractive metallurgy has used the land-based lithosphere. Oil and gas industries have made extensive use of the oceanic lithosphere.
An ore is a mixture of minerals, one or more of which can be economically exploited as a source for one or more materials. These materials might ultimately be produced as metals, alloys, compounds or concentrates. Note that the key is economically viable production. This requires a certain grade of the mineral(s) to be present. Not all mineral deposits are ore bodies, but all ore bodies are mineral deposits. With advances in technology, or increases in price what was only a mineral deposit at one time, might become an ore later.
A concentration unit, usually mg (micrograms) of species of interest per kg of total mass of the sample. Since the density of water is nearly 1000 g/L (998 g/L at 21°C), and the density of a dilute solution is close to that of water, ppb is also often used synonymously with mg/L, though this is not strictly correct.
Strictly speaking, a mineral is a naturally occurring, homogeneous inorganic substance of definite composition and crystal structure. Native elements (e.g. sulfur, gold) and compounds are included. A great many solid materials in nature fit this description. However, some solid materials have variable composition, e.g. pyrrhotite, FeS1−x, and pentlandite, (Fe,Ni)9S8, where, respectively, varying fractions of iron and sulfur and nickel and iron occur. These are still considered minerals. Finally, coal, which is not exactly an inorganic substance nor of definite composition, may also be considered a mineral, broadly speaking.
The average concentration (in % by weight) of an element on the earth. (Strictly speaking it refers to the average concentration of any specified isotope. In hydrometallurgy the usual concern is with the average concentration including all naturally occurring, stable isotopes of an element.)
A mineral deposit is a body of natural material that contains specific minerals.
These include: Ru, Os, Rh, Ir, Pd, Pt. They are generally rare, noble metals and precious. PGM's and some of their compounds are noted for their catalytic properties.
The unwanted minerals that constitute the unwanted minerals of an ore. These are usually substantial fractions of the total mass.

