Unit 9.4: Diseases of the Gastrointestinal System
Outline
Part 1: Anatomy and Normal Microbiota of the Digestive System
Part 2: Bacterial Infections of the Digestive System
- Staphylococcal Food Poisoning
- Shigellosis (Bacillary Dysentery)
- Salmonellosis
- E. coli Infections
- Cholera
- Peptic Ulcers
- Clostridioides difficile
Learning Objectives
After reading the following, you should be able to:
- Describe the basic structure of the gastrointestinal system, including protective features and defenses.
- Describe any features that promote microbial growth and give one example of a bacterial species that is part of the normal flora.
- Describe any general features of infections of the gastrointestinal system.
- Describe staphylococcal food poisoning, including its causative organism and possible complications.
- Describe shigellosis, including its causative organism and possible complications.
- Describe salmonellosis, including its causative organism and possible complications.
- Describe E. coli infections and possible complications.
- Differentiate between EPEC and EHEC
- Describe cholera, including its causative organism and possible complications.
- Describe peptic ulcers, including their causative organism and possible complications.
- Describe C. difficile infections, including any possible complications.
- Describe hepatitis, including its causative organism and possible complications.
- Differentiate between HAV, HBV and HCV
- Describe giardiasis, including its causative organism and possible complications.
- For all the diseases in this section, describe mode of transmission, portal of entry, modes of detection (where discussed) and treatments (in general, e.g. antiviral medication, vaccine, etc)
Gastrointestinal (GI) diseases are so common that, unfortunately, most people have had first-hand experience with the unpleasant symptoms, such as diarrhea, vomiting, and abdominal discomfort. The causes of gastrointestinal illness can vary widely, but such diseases can be grouped into two categories: those caused by infection (the growth of a pathogen in the GI tract) or intoxication (the presence of a microbial toxin in the GI tract).
Foodborne pathogens like Escherichia coli O157:H7 are among the most common sources of gastrointestinal disease. Contaminated food and water have always posed a health risk for humans, but in today’s global economy, outbreaks can occur on a much larger scale. E. coli O157:H7 is a potentially deadly strain of E. coli with a history of contaminating meat and produce that are not properly processed. The source of an E. coli O157:H7 outbreak can be difficult to trace, especially if the contaminated food is processed in a foreign country. Once the source is identified, authorities may issue recalls of the contaminated food products, but by then there are typically numerous cases of food poisoning, some of them fatal.
Part 1: Anatomy and Normal Microbiota of the Digestive System
The human digestive system, or the gastrointestinal (GI) tract, begins with the mouth and ends with the anus. The parts of the mouth include the teeth, the gums, the tongue, the oral vestibule (the space between the gums, lips, and teeth), and the oral cavity proper (the space behind the teeth and gums). Other parts of the GI tract are the pharynx, esophagus, stomach, small intestine, large intestine, rectum, and anus (Figure 9.33). Accessory digestive organs include the salivary glands, liver, gallbladder, spleen, and pancreas.
The digestive system contains normal microbiota, including archaea, bacteria, fungi, protists, and even viruses. Because this microbiota is important for normal functioning of the digestive system, alterations to the microbiota by antibiotics or diet can be harmful. Additionally, the introduction of pathogens to the GI tract can cause infections and diseases. In this section, we will review the microbiota found in a healthy digestive tract and the general signs and symptoms associated with oral and GI infections.
Anatomy and Normal Microbiota of the GI Tract: As food leaves the oral cavity, it travels through the pharynx, or the back of the throat, and moves into the esophagus, which carries the food from the pharynx to the stomach without adding any additional digestive enzymes. The stomach produces mucus to protect its lining, as well as digestive enzymes and acid to break down food. Partially digested food then leaves the stomach through the pyloric sphincter, reaching the first part of the small intestine called the duodenum. Pancreatic juice, which includes enzymes and bicarbonate ions, is released into the small intestine to neutralize the acidic material from the stomach and to assist in digestion. Bile, produced by the liver but stored in the gallbladder, is also released into the small intestine to emulsify fats so that they can travel in the watery environment of the small intestine. Digestion continues in the small intestine, where the majority of nutrients contained in the food are absorbed. Simple columnar epithelial cells called enterocytes line the lumen surface of the small intestinal folds called villi. Each enterocyte has smaller microvilli (cytoplasmic membrane extensions) on the cellular apical surface that increase the surface area to allow more absorption of nutrients to occur.
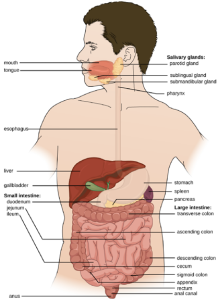
Digested food leaves the small intestine and moves into the large intestine, or colon, where there is a more diverse microbiota. Near this junction, there is a small pouch in the large intestine called the cecum, which attaches to the appendix. Further digestion occurs throughout the colon and water is reabsorbed, then waste is excreted through the rectum, the last section of the colon, and out of the body through the anus.
The environment of most of the GI tract is harsh, which serves two purposes: digestion and immunity. The stomach is an extremely acidic environment (pH 1.5–3.5) due to the gastric juices that break down food and kill many ingested microbes; this helps prevent infection from pathogens. The environment in the small intestine is less harsh and is able to support microbial communities. Microorganisms present in the small intestine can include lactobacilli, diptherioids and the fungus Candida. On the other hand, the large intestine (colon) contains a diverse and abundant microbiota that is important for normal function. These microbes include Bacteriodetes (especially the genera Bacteroides and Prevotella) and Firmicutes (especially members of the genus Clostridium). Methanogenic archaea and some fungi are also present, among many other species of bacteria. These microbes all aid in digestion and contribute to the production of feces, the waste excreted from the digestive tract, and flatus, the gas produced from microbial fermentation of undigested food. They can also produce valuable nutrients. For example, lactic acid bacteria such as bifidobacteria can synthesize vitamins, such as vitamin B12, folate, and riboflavin, that humans cannot synthesize themselves. E. coli found in the intestine can also break down food and help the body produce vitamin K, which is important for blood coagulation.
The GI tract has several other methods of reducing the risk of infection by pathogens. Small aggregates of underlying lymphoid tissue in the ileum, called Peyer’s patches, detect pathogens in the intestines via microfold (M) cells, which transfer antigens from the lumen of the intestine to the lymphocytes on Peyer’s patches to induce an immune response. The Peyer’s patches then secrete IgA and other pathogen-specific antibodies into the intestinal lumen to help keep intestinal microbes at safe levels. Goblet cells, which are modified simple columnar epithelial cells, also line the GI tract. Goblet cells secrete a gel-forming mucin, which is the major component of mucus. The production of a protective layer of mucus helps reduce the risk of pathogens reaching deeper tissues.
The constant movement of materials through the gastrointestinal tract also helps to move transient pathogens out of the body. In fact, feces are composed of approximately 25% microbes, 25% sloughed epithelial cells, 25% mucus, and 25% digested or undigested food. Finally, the normal microbiota provides an additional barrier to infection via a variety of mechanisms. For example, these organisms outcompete potential pathogens for space and nutrients within the intestine. This is known as competitive exclusion. Members of the microbiota may also secrete protein toxins known as bacteriocins that are able to bind to specific receptors on the surface of susceptible bacteria.
General Signs and Symptoms of GI Disease: Infections and intoxications of the lower GI tract often produce symptoms such as nausea, vomiting, diarrhea, aches, and fever. In some cases, vomiting and diarrhea may cause severe dehydration and other complications that can become serious or fatal. Various clinical terms are used to describe gastrointestinal symptoms. For example, gastritis is an inflammation of the stomach lining that results in swelling and enteritis refers to inflammation of the intestinal mucosa. When the inflammation involves both the stomach lining and the intestinal lining, the condition is called gastroenteritis. Inflammation of the liver is called hepatitis. Inflammation of the colon, called colitis, commonly occurs in cases of food intoxication. Because an inflamed colon does not reabsorb water as effectively as it normally does, stools become watery, causing diarrhea. Damage to the epithelial cells of the colon can also cause bleeding and excess mucus to appear in watery stools, a condition called dysentery.
Part 2: Bacterial Infections of the Digestive System
A wide range of gastrointestinal diseases are caused by bacterial contamination of food. Recall that foodborne disease can arise from either infection or intoxication. In both cases, bacterial toxins are typically responsible for producing disease signs and symptoms. The distinction lies in where the toxins are produced. In an infection, the microbial agent is ingested, colonizes the gut, and then produces toxins that damage host cells. In an intoxication, bacteria produce toxins in the food before it is ingested. In either case, the toxins cause damage to the cells lining the gastrointestinal tract, typically the colon. This leads to the common signs and symptoms of diarrhea or watery stool and abdominal cramps, or the more severe dysentery. Symptoms of foodborne diseases also often include nausea and vomiting, which are mechanisms the body uses to expel the toxic materials.
Most bacterial gastrointestinal illness is short-lived and self-limiting; however, loss of fluids due to severe diarrheal illness can lead to dehydration that can, in some cases, be fatal without proper treatment. Oral rehydration therapy with electrolyte solutions is an essential aspect of treatment for most patients with GI disease, especially in children and infants.
Staphylococcal Food Poisoning: Staphylococcal food poisoning is one form of food intoxication. When Staphylococcus aureus grows in food, it may produce enterotoxins that, when ingested, can cause symptoms such as nausea, diarrhea, cramping, and vomiting within one to six hours. In some severe cases, it may cause headache, dehydration, and changes in blood pressure and heart rate. Signs and symptoms resolve within 24 to 48 hours. S. aureus is often associated with a variety of raw or undercooked and cooked foods including meat (e.g., canned meat, ham, and sausages) and dairy products (e.g., cheeses, milk, and butter). It is also commonly found on hands and can be transmitted to prepared foods through poor hygiene, including poor handwashing and the use of contaminated food preparation surfaces, such as cutting boards.
There are at least 21 Staphylococcal enterotoxins and Staphylococcal enterotoxin-like toxins that can cause food intoxication. The enterotoxins are proteins that are resistant to low pH, allowing them to pass through the stomach. They are heat stable and are not destroyed by boiling at 100 °C. Even though the bacterium itself may be killed, the enterotoxins alone can cause vomiting and diarrhea, although the mechanisms are not fully understood. At least some of the symptoms may be caused by the enterotoxin functioning as a superantigen and provoking a strong immune response by activating T cell proliferation.
The rapid onset of signs and symptoms helps to diagnose this foodborne illness. Because the bacterium does not need to be present for the toxin to cause symptoms, diagnosis is confirmed by identifying the toxin in a food sample or in biological specimens (feces or vomitus) from the patient. The condition generally resolves relatively quickly, within 24 hours, without treatment. In some cases, supportive treatment in a hospital may be needed.
Shigellosis (Bacillary Dysentery): When gastrointestinal illness is associated with the rod-shaped, gram-negative bacterium Shigella, it is called bacillary dysentery, or shigellosis. Infections can be caused by S. dysenteriae, S. flexneri, S. boydii, and/or S. sonnei that colonize the GI tract. Shigellosis can be spread from hand to mouth or through contaminated food and water. Most commonly, it is transmitted through the fecal-oral route.
Shigella bacteria invade intestinal epithelial cells. When taken into a phagosome, they can escape and then live within the cytoplasm of the cell or move to adjacent cells. As the organisms multiply, the epithelium and structures with M cells of the Peyer’s patches in the intestine may become ulcerated and cause loss of fluid. Stomach cramps, fever, and watery diarrhea that may also contain pus, mucus, and/or blood often develop. More severe cases may result in ulceration of the mucosa, dehydration, and rectal bleeding. Additionally, patients may later develop hemolytic uremic syndrome (HUS), a serious condition in which damaged blood cells build up in the kidneys and may cause kidney failure, or reactive arthritis, a condition in which arthritis develops in multiple joints following infection. Patients may also develop chronic post-infection irritable bowel syndrome (IBS).
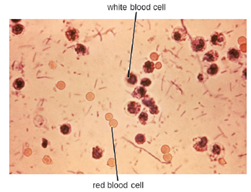
S. dysenteriae type 1 is able to produce Shiga toxin, which targets the endothelial cells of small blood vessels in the small and large intestine by binding to a glycosphingolipid. Once inside the endothelial cells, the toxin targets the large ribosomal subunit, thus affecting protein synthesis of these cells. Hemorrhaging and lesions in the colon can result. The toxin can target the kidney’s glomerulus, the blood vessels where filtration of blood in the kidney begins, thus resulting in HUS. The presence of white blood cells and blood in fecal samples occurs in about 70% of patients (Figure 9.34). Severe cases may require antibiotics such as ciprofloxacin and azithromycin, but these must be carefully prescribed because resistance is increasingly common.
Salmonellosis: Salmonella gastroenteritis, also called salmonellosis, is caused by the rod-shaped, gram-negative bacterium Salmonella. Two species, S. enterica and S. bongori, cause disease in humans, but S. enterica is the most common.
Salmonella is a part of the normal intestinal microbiota of many individuals. However, salmonellosis is caused by exogenous agents, and infection can occur depending on the serotype, size of the inoculum, and overall health of the host. Infection is caused by ingestion of contaminated food, handling of eggshells, or exposure to certain animals. Salmonella is part of poultry’s microbiota, so exposure to raw eggs and raw poultry can increase the risk of infection. Handwashing and cooking foods thoroughly greatly reduce the risk of transmission. Salmonella bacteria can survive freezing for extended periods but cannot survive high temperatures.
Once the bacteria are ingested, they multiply within the intestines and penetrate the epithelial mucosal cells via M cells where they continue to grow (Figure 9.35). They trigger inflammatory processes and the hyper-secretion of fluids. Once inside the body, they can persist inside the phagosomes of macrophages. Salmonella can cross the epithelial cell membrane and enter the bloodstream and lymphatic system. Some strains of Salmonella also produce an enterotoxin that can cause an intoxication.
Infected individuals develop fever, nausea, abdominal cramps, vomiting, headache, and diarrhea. These signs and symptoms generally last a few days to a week. According to the Centers for Disease Control and Prevention (CDC), there are 1,000,000 cases annually, with 380 deaths each year. However, because the disease is usually self-limiting, many cases are not reported to doctors and the overall incidence may be underreported. Diagnosis involves culture followed by serotyping and DNA fingerprinting if needed. Serotyping is important for determining treatment. Oral rehydration therapy is commonly used. Antibiotics are only recommended for serious cases.
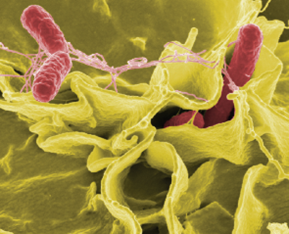
E. coli Infections: The gram-negative rod Escherichia coli is a common member of the normal microbiota of the colon. Although the vast majority of E. coli strains are helpful commensal bacteria, some can be pathogenic and may cause dangerous diarrheal disease. The pathogenic strains have additional virulence factors such as type 1 fimbriae that promote colonization of the colon or may produce toxins.
These virulence factors are acquired through horizontal gene transfer.
Extraintestinal disease can result if the bacteria spread from the gastrointestinal tract. Although these bacteria can be spread from person to person, they are often acquired through contaminated food or water. There are six recognized pathogenic groups of E. coli, but we will focus here on two that are commonly transmitted through food and water.
Enteropathogenic E. coli (EPEC) can cause a potentially fatal diarrhea, especially in infants and those in less developed countries. Fever, vomiting, and diarrhea can lead to severe dehydration. These E. coli inject a protein (Tir) that attaches to the surface of the intestinal epithelial cells and triggers rearrangement of host cell actin from microvilli to pedestals. Tir also happens to be the receptor for Intimin, a surface protein produced by EPEC, thereby allowing E. coli to “sit” on the pedestal. The genes necessary for this pedestal formation are encoded on the locus for enterocyte effacement (LEE) pathogenicity island. The disease is usually relatively mild and self-limiting. If needed, antibiotic treatment with fluoroquinolones, doxycycline, rifaximin, and trimethoprim-sulfamethoxazole
(TMP/SMZ) may shorten infection duration. However, antibiotic resistance is a problem.
The most dangerous strains are enterohemorrhagic E. coli (EHEC), which are the strains capable of causing epidemics. In particular, the strain O157:H7 has been responsible for several recent outbreaks. The O and H refer to surface antigens that contribute to pathogenicity and trigger a host immune response (“O” refers to the O-side chain of the lipopolysaccharide and the “H” refers to the flagella). Similar to EPEC, EHEC also forms pedestals. EHEC also produces a Shiga-like toxin. Because the genome of this bacterium has been sequenced, it is known that the Shiga toxin genes were most likely acquired through transduction (horizontal gene transfer). The Shiga toxin genes originated from Shigella dysenteriae. Prophage from a bacteriophage that previously infected Shigella integrated into the chromosome of E. coli.
EHEC can cause disease ranging from relatively mild to life-threatening. Symptoms include bloody diarrhea with severe cramping, but no fever. Although it is often self-limiting, it can lead to hemorrhagic colitis and profuse bleeding. One possible complication is HUS. Antibiotic therapy is not recommended and may worsen HUS because of the toxins released when the bacteria are killed, so supportive therapies must be used.
Cholera: The gastrointestinal disease cholera is a serious infection often associated with poor sanitation, especially following natural disasters, because it is spread through contaminated water and food that has not been heated to temperatures high enough to kill the bacteria. It is caused by Vibrio cholerae serotype O1, a gram-negative, flagellated bacterium in the shape of a curved rod (vibrio). According to the CDC, cholera causes an estimated 3 to 5 million cases and 100,000 deaths each year.
Because V. cholerae is killed by stomach acid, relatively large doses are needed for a few microbial cells to survive to reach the intestines and cause infection. The motile cells travel through the mucous layer of the intestines, where they attach to epithelial cells and release cholera enterotoxin. The toxin is an A-B toxin with activity through adenylate cyclase. Within the intestinal cell, cyclic AMP (cAMP) levels increase, which activates a chloride channel and results in the release of ions into the intestinal lumen.
This increase in osmotic pressure in the lumen leads to water also entering the lumen. As the water and electrolytes leave the body, it causes rapid dehydration and electrolyte imbalance. Diarrhea is so profuse that it is often called “rice water stool,” and patients are placed on cots with a hole in them to monitor the fluid loss (Figure 9.36).
Cholera is diagnosed by taking a stool sample and culturing for Vibrio. Cholera may be self-limiting and treatment involves rehydration and electrolyte replenishment. Although antibiotics are not typically needed, they can be used for severe or disseminated disease. Tetracyclines are recommended, but doxycycline, erythromycin, orfloxacin, ciprofloxacin, and TMP/SMZ may be used. Recent evidence suggests that azithromycin is also a good first-line antibiotic. Good sanitation—including appropriate sewage treatment, clean supplies for cooking, and purified drinking water—is important to prevent infection (Figure 9.36)

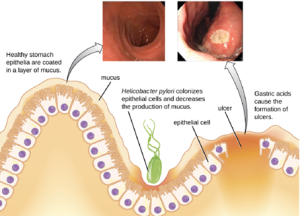
Peptic Ulcers: The gram-negative bacterium Helicobacter pylori is able to tolerate the acidic environment of the human stomach and has been shown to be a major cause of peptic ulcers, which are ulcers of the stomach or duodenum. The bacterium is also associated with increased risk of stomach cancer (Figure 9.37). According to the CDC, approximately two-thirds of the population is infected with H. pylori, but less than 20% have a risk of developing ulcers or stomach cancer. H. pylori is found in approximately 80% of stomach ulcers and in over 90% of duodenal ulcers.
H. pylori colonizes epithelial cells in the stomach using pili for adhesion. These bacteria produce urease, which stimulates an immune response and creates ammonia that neutralizes stomach acids to provide a more hospitable microenvironment. The infection damages the cells of the stomach lining, including those that normally produce the protective mucus that serves as a barrier between the tissue and stomach acid. As a result, inflammation (gastritis) occurs and ulcers may slowly develop.
Signs and symptoms include nausea, lack of appetite, bloating, burping, and weight loss. Bleeding ulcers may produce dark stools. If no treatment is provided, the ulcers can become deeper, more tissues can be involved, and stomach perforation can occur. Because perforation allows digestive enzymes and acid to leak into the body, it is a very serious condition.
To diagnose H. pylori infection, multiple methods are available. In a breath test, the patient swallows radiolabeled urea. If H. pylori is present, the bacteria will produce urease to break down the urea. This reaction produces radiolabeled carbon dioxide that can be detected in the patient’s breath. Blood testing can also be used to detect antibodies to H. pylori. The bacteria themselves can be detected using either a stool test or a stomach wall biopsy. Antibiotics can be used to treat the infection. Although treatment is often valuable, there are also risks to H. pylori eradication. Infection with H. pylori may actually protect against some cancers, such as esophageal adenocarcinoma and gastroesophageal reflux disease.
Clostridioides difficile: Clostridioides difficile is a gram-positive rod that can be a commensal bacterium as part of the normal microbiota of healthy individuals. When the normal microbiota is disrupted by long-term antibiotic use, it can allow the overgrowth of this bacterium, resulting in antibiotic-associated diarrhea caused by C. difficile. Antibiotic-associated diarrhea can also be considered a nosocomial disease. Patients at the greatest risk of C. difficile infection are those who are immunocompromised, have been in health-care settings for extended periods, are older, have recently taken antibiotics, have had gastrointestinal procedures done, or use proton pump inhibitors, which reduce stomach acidity and allow proliferation of C. difficile. Because this species can form endospores, it can survive for extended periods of time in the environment under harsh conditions and is a considerable concern in health-care settings.
This bacterium produces two toxins, Clostridioides difficile toxin A (TcdA) and Clostridioides difficile toxin B (TcdB). These toxins inactivate small GTP-binding proteins, resulting in actin condensation and cell rounding, followed by cell death. Infections begin with focal necrosis, then ulceration with exudate, and can progress to pseudomembranous colitis, which involves inflammation of the colon and the development of a pseudomembrane of fibrin containing dead epithelial cells and leukocytes (Figure 9.38). Watery diarrhea, dehydration, fever, loss of appetite, and abdominal pain can result. Perforation of the colon can occur, leading to septicemia, shock, and death. C. difficile is also associated with necrotizing enterocolitis in premature babies and neutropenic enterocolitis associated with cancer therapies.
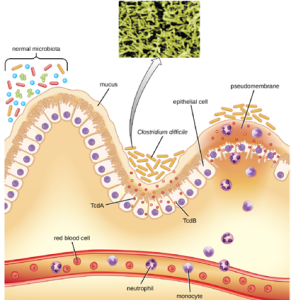
The first step of conventional treatment is to stop antibiotic use, and then to provide supportive therapy with electrolyte replacement and fluids. Metronidazole is the preferred treatment if the C. difficile diagnosis has been confirmed. Vancomycin can also be used, but it should be reserved for patients for whom metronidazole was ineffective or who meet other criteria (e.g., under 10 years of age, pregnant, or allergic to metronidazole). A newer approach to treatment, known as a fecal transplant, focuses on restoring the microbiota of the gut in order to combat the infection. In this procedure, a healthy individual donates a stool sample, which is mixed with saline and transplanted to the recipient via colonoscopy, endoscopy, sigmoidoscopy, or enema. It has been reported that this procedure has greater than 90% success in resolving C. difficile infections.
Part 3: Viral Infections of the Digestive System
Hepatitis: Hepatitis is a general term meaning inflammation of the liver, which can have a variety of causes. In some cases, the cause is viral infection. There are five main hepatitis viruses that are clinically significant: hepatitisviruses A (HAV), B (HBV), C (HCV), D, (HDV) and E (HEV), though only HAV, HAB and HAC will be discussed here). (Figure 9.39).

Although the five hepatitis viruses differ, they can cause some similar signs and symptoms because they all have an affinity for hepatocytes (liver cells). HAV and HEV can be contracted through ingestion while HBV, HCV, and HDV are transmitted by parenteral contact. It is possible for individuals to become long term or chronic carriers of hepatitis viruses.
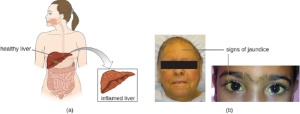
The virus enters the blood (viremia), spreading to the spleen, the kidneys, and the liver. During viral replication, the virus infects hepatocytes. The inflammation is caused by the hepatocytes replicating and releasing more hepatitis virus. Signs and symptoms include malaise, anorexia, loss of appetite, dark urine, pain in the upper right quadrant of the abdomen, vomiting, nausea, diarrhea, joint pain, and gray stool. Additionally, when the liver is diseased or injured, it is unable to break down hemoglobin effectively, and bilirubin can build up in the body, giving the skin and mucous membranes a yellowish color, a condition called jaundice (Figure 9.40). In severe cases, death from liver necrosis may occur.
Despite having many similarities, each of the hepatitis viruses has its own unique characteristics. HAV is generally transmitted through the fecal-oral route, close personal contact, or exposure to contaminated water or food. Hepatitis A can develop after an incubation period of 15 to 50 days (the mean is 30). It is normally mild or even asymptomatic and is usually self-limiting within weeks to months. A more severe form, fulminant hepatitis, rarely occurs but has a high fatality rate of 70–80%. Vaccination is available and is recommended especially for children (between ages one and two), those traveling to countries with higher risk, those with liver disease and certain other conditions, and drug users.
Although HBV is associated with similar signs and symptoms, transmission and outcomes differ. This virus has a mean incubation period of 120 days and is generally associated with exposure to infectious blood or body fluids such as semen or saliva. Exposure can occur through skin puncture, across the placenta, or through mucosal contact, but it is not spread through casual contact such as hugging, hand holding, sneezing, or coughing, or even through breastfeeding or kissing. Risk of infection is greatest for those who use intravenous drugs or who have sexual contact with an infected individual. Health-care workers are also at risk from needle sticks and other injuries when treating infected patients. The infection can become chronic and may progress to cirrhosis or liver failure. It is also associated with liver cancer. Chronic infections are associated with the highest mortality rates and are more common in infants. Approximately 90% of infected infants become chronic carriers, compared with only 6–10% of infected adults. Vaccination is available and is recommended for children as part of the standard vaccination schedule (one dose at birth and the second by 18 months of age) and for adults at greater risk (e.g., those with certain diseases, intravenous drug users, and those who have sex with multiple partners). Health-care agencies are required to offer the HBV vaccine to all workers who have occupational exposure to blood and/or other infectious materials.
HCV is often undiagnosed and therefore may be more widespread than is documented. It has a mean incubation period of 45 days and is transmitted through contact with infected blood. Although some cases are asymptomatic and/or resolve spontaneously, 75%–85% of infected individuals become chronic carriers. Nearly all cases result from parenteral transmission often associated with IV drug use or transfusions. The risk is greatest for individuals with past or current history of intravenous drug use or who have had sexual contact with infected individuals. It has also been spread through contaminated blood products and can even be transmitted through contaminated personal products such as toothbrushes and razors. New medications have recently been developed that show great effectiveness in treating HCV and that are tailored to the specific genotype causing the infection.
Part 4: Protozoan Infections of the Digestive System
Giardiasis: Also called backpacker’s diarrhea or beaver fever, giardiasis is a common disease in the United States caused by the flagellated protist Giardia lamblia (Figure 9.41). To establish infection, G. lamblia uses a large adhesive disk to attach to the intestinal mucosa. The disk is comprised of microtubules. During adhesion, the flagella of G. lamblia move in a manner that draws fluid out from under the disk, resulting in an area of lower pressure that promotes its adhesion to the intestinal epithelial cells. Due to its attachment, Giardia also blocks absorption of nutrients, including fats.
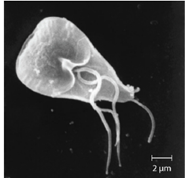
Transmission occurs through contaminated food or water or directly from person to person. Children in day-care centers are at risk due to their tendency to put items into their mouths that may be contaminated. Large outbreaks may occur if a public water supply becomes contaminated. Giardia have a resistant cyst stage in their life cycle that is able to survive cold temperatures and the chlorination treatment typically used for drinking water in municipal reservoirs. As a result, municipal water must be filtered to trap and remove these cysts. Once consumed by the host, Giardia develops into the active tropozoite.
Infected individuals may be asymptomatic or have gastrointestinal signs and symptoms, sometimes accompanied by weight loss. Common symptoms, which appear one to three weeks after exposure, include diarrhea, nausea, stomach cramps, gas, greasy stool (because fat absorption is being blocked), and possible dehydration. The parasite remains in the colon and does not cause systemic infection. Signs and symptoms generally clear within two to six weeks. Chronic infections may develop and are often resistant to treatment. These are associated with weight loss, episodic diarrhea, and malabsorption syndrome due to the blocked nutrient absorption.
Diagnosis may be made using observation under the microscope. A stool ova and parasite (O&P) exam involves direct examination of a stool sample for the presence of cysts and trophozoites; it can be used to distinguish common parasitic intestinal infections. The most common treatments use metronidazole as the first-line choice, followed by tinidazole. If the infection becomes chronic, the parasites may become resistant to medications.

