Chapter 3 Induced Innate Immune Responses
Learning Objectives
By the end of this chapter you will be able to:
- Describe how innate immune cells identify pathogens and engage in eliminating pathogens by phagocytosis.
- Define cytokine signalling and identify relevant cytokines involved in immune responses.
- Describe the physiological events and immune cell recruitment associated with localized inflammation.
- Explain the potential outcomes of systemic inflammation and fever.
- Describe the actions of plasma proteins such as complement.
Case Study
Emily is 48 years old and identifies as a female. Her medical history is unknown and she does not appear to have underlying conditions. Emily presented with signs of COVID-19 and tested positive for the virus that causes this infectious disease (SARS-CoV-2). The viral infection centered within the respiratory tract. Emily was admitted to the hospital on day 10 of infection after experiencing worsening localized respiratory inflammation, including dry cough for 10 days and shortness of breath for 5 days. However, Emily also experienced a systemic response involving a fever of 38.7°C and general malaise. As the infection progressed, Emily’s signs and symptoms became more severe. By day 13, anti-pyretic medication was used to manage the persistent fever and on day 18 a blood test revealed elevated cytokines IL-6, IL-8, and TNF-α. Emily’s conditions began to worsen rapidly and further blood tests revealed excessive accumulation of these same cytokines. Aggressive therapy was used to support breathing and tissue oxygenation and immunomodulatory drugs were administered to suppress the apparent cytokine storm. Following treatment, Emily progressively overcame the infection and recovered.
- Reflect on what happens to tissue when it is injured and inflamed. Why do you think that inflammation of the respiratory tract contributes to difficulty in breathing?
- Cytokines are chemicals used by immune cells to communicate and trigger a specific immune response. What do you think is the relevance of the cytokines identified in Emily’s blood test?
- Why was it important to use immunomodulatory drugs to suppress the immune response in this case of COVID-19?
Answers to these questions are at the end of the chapter.
3.1 Phagocytosis
Many human cells, including epithelial cells lining the body surface and endothelial cells around the blood vessels, are capable of consuming dead human cells and foreign microbes through a process called phagocytosis. However, these cells would be regarded as non-professional phagocyte cells. Immune cells, including neutrophils, macrophages and dendritic cells, are professional phagocytes, also simply referred to as phagocytes. These cells differ from non-professional phagocytes because they have specific cell receptors that allow them to efficient detect and eliminate pathogens.
Phagocytes detect the target cell using pattern recognition receptors (PRRs), which include toll-like receptors (TLRs). These receptors do not identify specific pathogens but rather detect molecular patterns that are commonly found on the surface of pathogens. These pathogen-associated molecular patterns are abbreviated as PAMPs. Examples of PAMPs include components of the bacterial cell wall, such as peptidoglycan and lipopolysaccharide (LPS).
Upon binding a microbial cell, the phagocyte cell surface undergoes structural changes that wrap the phagocyte cell membrane around to the microbe. When fully encapsulated, the phagocyte surface fuses to fully contain the microbe inside a bubble within the cell, called a phagosome. Specialized organelles, called lysosomes, contain chemicals and enzymes that are capable of killing and digesting the microbial cell. These lysosomes fuse with the phagosome to produce a structure called the phagolysosome.
Figure 3-1 Phagocytosis. Phagocytes have specialized pattern recognition receptors (PRR) to bind distinguishing patterns (PAMPs) on the surface of microbial cells. The membrane surface of the cell folds and encapsulates the microbe into a vacuole called a phagosome. Lysosomes concentrate within them digestive chemicals and enzymes, and these lysosomes fuse with the phagosome to produce a phagolysosome. The microbial cell is killed within this vacuole and the digested material is expelled from the cell. [Link to Image Description]
Image Source: Modified image from OpenStax CC BY 4.0, via Wikimedia Commons
Two methods are used to kill the microbial cell within the phagolysosome:
- Oxygen-dependent mechanisms can be used to destroy entrapped cells. This mechanism is also known as a respiratory burst or oxidative burst. Metabolism of oxygen produces molecular byproducts that are toxic, called reactive oxygen species (ROS). Normally reactive oxygen species are detoxified within the cell, however, they can also be repurposed to digest captured microbes. Relevant ROS include hydrogen peroxide (H2O2), hydroxyl free radicals (HO–), superoxide anion (O2–), singlet oxygen (O–) or hypochlorite (OCl–).
- Oxygen independent mechanisms include other chemicals and proteins (e.g. enzymes) stored within the lysosome. Lactic acid and nitric oxide (NO) are harmful chemicals. Cationic proteins can damage the bacterial cell membrane. Enzymes include proteases, phospholipases, nucleases and lysozymes.
3.2 Cytokine Signaling
Complex cellular responses, such as inflammation and fever, are coordinated by an equally complex system of chemical communication. This communication is accomplished by secreting small proteins called cytokines. Cytokines are secreted by immune cells as well as other cells that are stressed or damaged. Cytokines bind receptor proteins on the surface of other immune cells to induce a specific response in that target cell. Cytokines are involved in a range of immunological responses, including (but not limited to) an inflammatory response to infection, an anti-inflammatory response to return to normal after infection, as well as induction of fever.
Cytokines may be classified into various groups of signalling molecules:
- Tumor Necrosis Factors (TNF) are multifunctional cytokines that regulate fever and inflammation, and inhibit cancer cell growth and viral replication.
- Interleukins (IL) were originally isolated from leukocytes but are now known to be secreted by other cells as well.
Many cytokines are produced in response to injury and infection but two signalling functions are particularly important:
- Chemotactic cytokines – These chemicals are released by both immune cells and other cells in the tissue to direct immune cells to the site of infection. Immune cells follow the chemotactic gradient from regions of low cytokine concentration to regions of higher concentration. The immune cells will ultimately swarm the tissue region where these chemotactic agents were produced and secreted.
- Examples of chemoattractants and chemotactic cytokines include:
- Fibrin and Platelet activating factors (PAF) – proteins involved in blood clotting attract leukocytes to the site of injury.
- Collagen – tissue repair protein attracts leukocytes.
- Mast cell chemotactic factors – specific proteins secreted by mast cells to recruit leukocytes when they encounter pathogens.
- Examples of chemoattractants and chemotactic cytokines include:
- Vasoactive cytokines – These chemicals alter the structure of the blood vessels, or vasculature. For example, vasoactive cytokines are responsible for vasodilation that enables white blood cells to extravasate from the bloodstream into tissues.
- Examples of vasoactive cytokines include:
- Histamine – stored in granules within mast cells to rapidly induce vasodilation at the early stages of inflammation.
- Bradykinin – produced found in tissues and blood, this protein acts on endothelium to produce vasodilation.
- Serotonin – often known for its function as a neurotransmitter in the nervous system, serotonin is also released by mast cells and platelets, causing both vasodilation and vasocontriction
- Prostaglandins – complex chemical mediators that can induce vasoconstriction or vasodilation, depending on the biological context.
- Examples of vasoactive cytokines include:
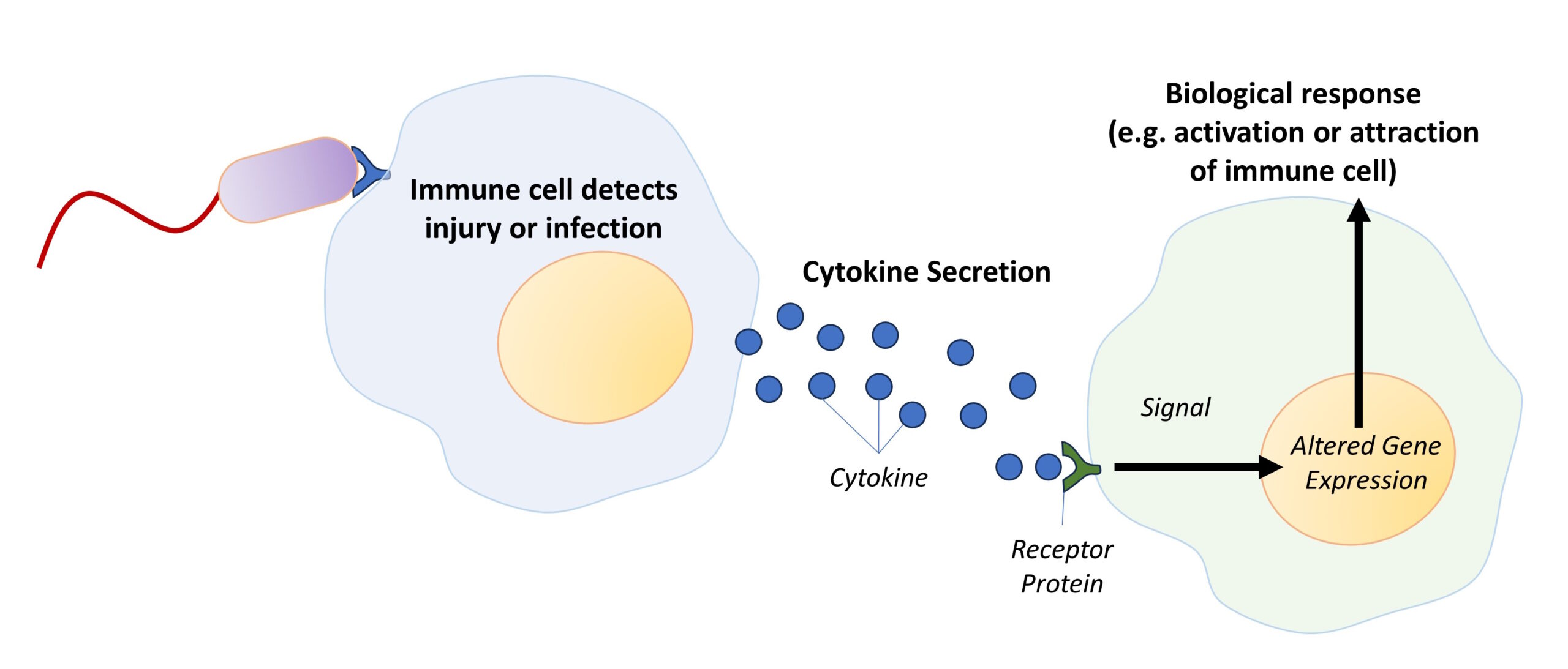
Figure 3-2 Cytokine Secretion. Immune cells communicate by secretion of cytokine proteins, where the secretion of these cytokines by one cell induces a biological response in another cell. The specific biological response depends on both the type and intensity of secreted cytokine. [Link to Image Description]
3.3 Localized Inflammation in Response to Infection
Inflammation is a physiological response induced by cytokine signalling. These inflammatory cytokines may be produced and released by damaged, infected or stressed cells or these cytokines may be secreted by leukocyte immune cells upon detection of infection or injury. The immediate consequence of inflammation is to alter blood vessels in the local tissues so that leukocytes can be recruited to the site for infection control, removal of dead cells and tissue repair. However, excessive or prolonged inflammation can result in tissue damage, dysfunction or even death.
Acute Inflammation
Immediately following an injury, blood vessels briefly constrict (vasoconstriction) to stop the flow of blood and to minimize blood loss. However, as platelets help form a blood clot, the constriction of blood vessels is no longer required. Once the clot forms, inflammation drives a series of events that lead to (1) expansion or vasodilation of blood vessels to cause more blood cells to collect around the site of injury/infection and (2) increased blood vessel (vascular) permeability to allow blood plasma fluid and leukocytes to enter into the tissues.
Vasodilation increases blood flow into vasculature around the site of infection. In this context, blood is actually flowing slowly through the blood vessels and the increase in blood flow means that blood is collecting in these tissues. The increased volume of fluid in blood vessels and tissues dilutes potential toxins produced by the pathogen. Also, leukocytes in the blood will flow closer to the walls of the blood vessels in a process called margination. Once at the site of injury, these immune cells may undergo diapedesis to extravasate from the bloodstream into the inflamed tissues.
Edema occurs as the blood vessels become more permeable and blood plasma fluid leaks into the tissue. This fluid may include extravasated leukocytes, clotting factors, defensive chemicals such as complement proteins and many other factors to support an efficient immune defense. When the fluid is rich in leukocytes, it may appear white and is commonly called pus.
Manifestation of localized inflammation stems from both these processes of vasodilation and edema. Vasodilation increases the flow of blood to the site of injury or infection. The tissue becomes red (erythema) and warm, owing to these characteristics of blood. Edema causes fluid to enter the tissue and cause swelling and altered function. An additional outcome of inflammation may be pain as swollen tissues impact on sensitive nerve endings.
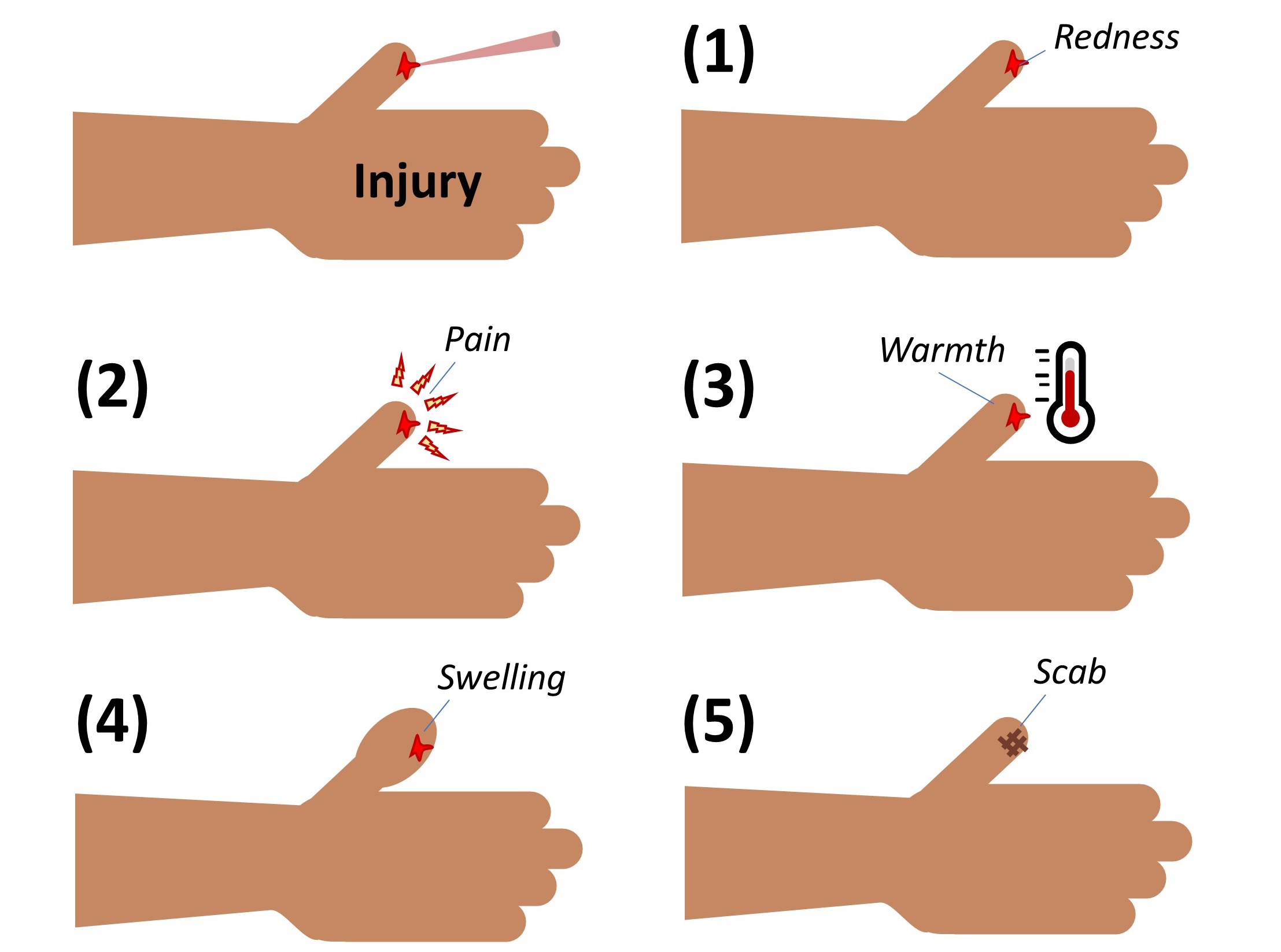
Five cardinal signs of inflammation have been described, including rubor (redness), dolor (pain), calor (warmth at the site), tumor (swelling) and loss of function. These signs are directly related to the effects of vasodilation and edema, as illustrated in Figure 3-3.
Figure 3-3 Cardinal Signs of Inflammation. In response to infection, injury or damage, inflammation may produce (1) Rubor – Redness as a consequence of increased blood flow to the site; (2) Dolor – Pain due to activation of nerve endings; (3) Calor – Heat due to accumulation of blood; (4) Tumor – where tissue swells as a consequence of edema; (5) Loss of function – where pain, swelling and/or rigid scab formation reduces the mobility of the tissue in order to prevent further injury. [Link to Image Description]
Acute Inflammation in Recruitment of Immune Cells
A primary purpose of the inflammatory process is to respond to infection and injury by recruiting immune cells and components to the inflamed tissue. Cells are attracted to the inflamed site by specific proteins that are expressed on the surface of endothelial cells in the blood vessels around the affected site. Leukocytes are then able to deform and migrate into the local tissue, through the pores formed during vasodilation of the blood vessel. Once inside the tissue, leukocytes follow chemotactic cytokine gradients to the origin of the inflammation signals.
The process by which leukocytes migrate out from the blood vessels into the tissues involves a series of steps:
- Margination: The leukocytes form temporary adhesions to proteins on the blood vessel endothelial wall. These proteins are called selectins. The endothelial cells near the site of inflammation express additional proteins called intercellular adhesion molecules (ICAMs). The leukocytes have integrin proteins on their surface that specifically bind ICAMs and form tight adhesion in the vicinity of the inflammatory site.
- Extravasation: The leukocytes flatten and pass through gaps between endothelial cells. This process is sometimes also called diapedesis.
- Chemotaxis: Once the leukocytes cross from the blood vessel into the tissue, the cell will follow a cytokine concentration from the distant region of low concentration to the site of inflammation, where the cytokine concentration is much higher.
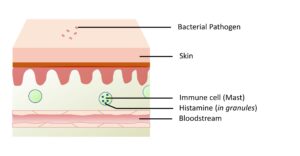 Figure 3-3A. Ubiquitous State. Even prior to infection or injury, mast cells can be found deposited throughout the body. These mast cells have granules with pre-formed histamine that is capable of rapidly inducing an inflammatory response.
Figure 3-3A. Ubiquitous State. Even prior to infection or injury, mast cells can be found deposited throughout the body. These mast cells have granules with pre-formed histamine that is capable of rapidly inducing an inflammatory response.
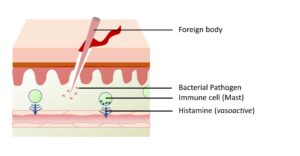
Figure 3-3B. Mast cell degranulation. On detecting a pathogen, mast cells degranulate to secrete the histamine from internal granules into the surrounding tissue.

Figure 3-3C. Cytokine Signalling. Mast cells secrete “mast cell chemotactic factors” that attract local leukocytes. The secreted histamine induces vasodilation which results in accumulation of blood and blood cells in the region of inflammation.
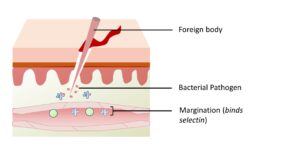
Figure 3-3D. Margination. Leukocytes in the blood stick to the endothelial wall because of temporary binding to selectin proteins.
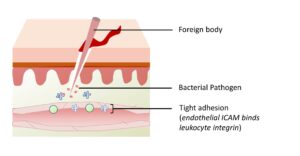
Figure 3-3E. Tight Adhesion. Endothelial cells around the site of inflammation express ICAM proteins. Integrin proteins on the surface of leukocytes bind tightly to ICAM, causing the leukocyte to bind tightly to the blood vessel wall.

Figure 3-3F. Extravasation. Leukocytes flatten and migrate through pores that form between the endothelial cells. Within the tissues, these cells follow a chemotactic gradient back to the site of infection. These newly-arrived leukocytes also secrete cytokines that make the inflammatory response even stronger.
 Figure 3-3A. Ubiquitous State. Even prior to infection or injury, mast cells can be found deposited throughout the body. These mast cells have granules with pre-formed histamine that is capable of rapidly inducing an inflammatory response.
Figure 3-3A. Ubiquitous State. Even prior to infection or injury, mast cells can be found deposited throughout the body. These mast cells have granules with pre-formed histamine that is capable of rapidly inducing an inflammatory response.

Figure 3-3B. Mast cell degranulation. On detecting a pathogen, mast cells degranulate to secrete the histamine from internal granules into the surrounding tissue.

Figure 3-3C. Cytokine Signalling. Mast cells secrete “mast cell chemotactic factors” that attract local leukocytes. The secreted histamine induces vasodilation which results in accumulation of blood and blood cells in the region of inflammation.

Figure 3-3D. Margination. Leukocytes in the blood stick to the endothelial wall because of temporary binding to selectin proteins.

Figure 3-3E. Tight Adhesion. Endothelial cells around the site of inflammation express ICAM proteins. Integrin proteins on the surface of leukocytes bind tightly to ICAM, causing the leukocyte to bind tightly to the blood vessel wall.

Figure 3-3F. Extravasation. Leukocytes flatten and migrate through pores that form between the endothelial cells. Within the tissues, these cells follow a chemotactic gradient back to the site of infection. These newly-arrived leukocytes also secrete cytokines that make the inflammatory response even stronger.
Chronic Inflammation
Chronic inflammation involves the same biological mechanisms as acute inflammation but the tissue is affected for a longer period of time that persists for weeks, months and even years. Chronic inflammation may occur if the body is continuously exposed to irritants or if a person is affected by autoimmune disease. In chronic inflammation, immune cells like lymphocytes and macrophages continuously release inflammatory cytokines, leading to tissue damage and fibrosis. Unlike acute inflammation, chronic inflammation is often characterized by tissue destruction and repair attempts occurring simultaneously, which can eventually lead to organ dysfunction and long-term health complications. Managing chronic inflammation is essential to prevent further tissue damage and maintain overall health.
3.4 Systemic Inflammation
In contrast to acute and chronic localized inflammation, where a specific tissue becomes red and swollen, systemic events affect the entire body. This may occur if cytokines or activated immune cells migrate from the inflamed tissue into the bloodstream. Systemic responses are particularly likely in the context of a septicemia, which describes a circumstance where a pathogen gains entry and spreads within the bloodstream. Once in the bloodstream, immune cells and cytokines are dispersed throughout the body. Systemic cytokines may stimulate immune cells throughout the body to release inflammatory cytokines of their own This produces a feed-forward loop called hypercytokinemia where excessive amounts of cytokines accumulate in the bloodstream. The dysregulated release of cytokines may be considered a cytokine storm and may lead to sepsis, which is an overwhelming and potentially life-threatening response to infection.
Systemic inflammation may pose an immediate health threat because vasodilation and edema occur throughout the body, resulting in a sudden and dramatic reduction of the fluid volume in the bloodstream. Without sufficient fluid, the blood pressure becomes too low to transport oxygen- and nutrient-rich blood to tissues and organs that need it, which may result in multi-organ failure and possibly even death.
3.5 Fever
Fever is a physiological response triggered by the immune system in response to infection or inflammation. Specifically, fever is triggered by molecules shed from pathogens or by cytokines, which are collectively called pyrogens. Pyrogens act on the hypothalamus, raising the body’s temperature set-point, which leads to an elevation in body temperature.
A higher body temperature may be beneficial as it makes it more difficult for certain pathogens to replicate and survive. Additionally, fever stimulates the production of interferons, which are proteins that help combat viral infections. Elevated body temperature also increases the efficiency of certain immune cells, such as T cells and macrophages, enhancing their ability to target and destroy pathogens.
However, fever can also cause discomfort and fatigue. In more severe cases, very high fevers can lead to dehydration and electrolyte (salt) imbalances, especially in young children or individuals with medical conditions that make them sensitive to electrolyte imbalance. Therefore, while fever is a crucial defense mechanism, managing it with appropriate fever-reducing medications (antipyretic medications) and ensuring adequate fluid and electrolyte intake is important to strike a balance between its beneficial effects and potential risks.
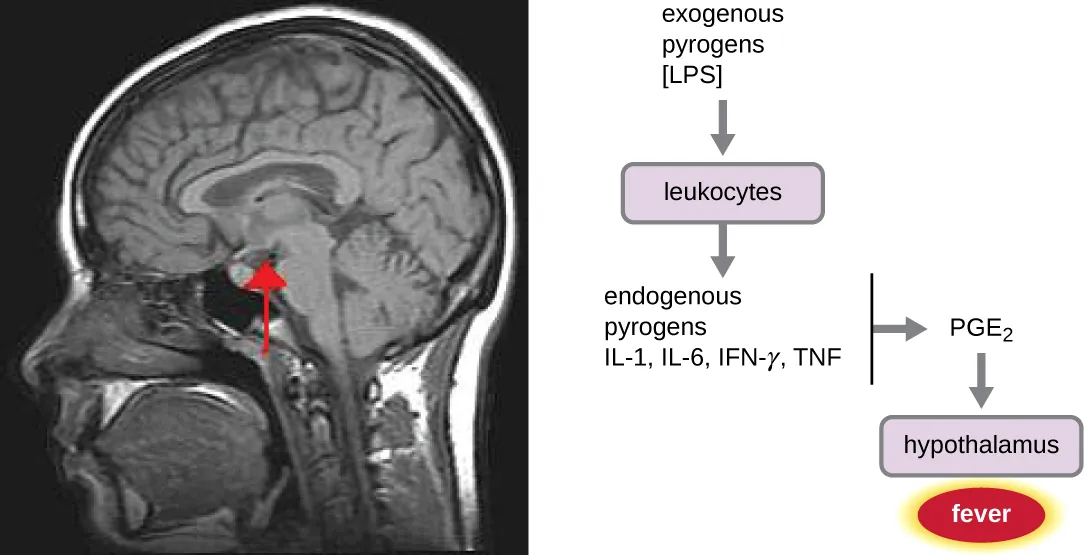
Figure 3-4 Mechanism of Fever. Leukocytes detect exogenous (non-human source) pyrogens, such as bacterial lipopolysaccharide (LPS). These leukocytes then secrete cytokines that serves as endogenous pyrogens in order to signal the hypothalamus of the brain to increase the body temperature set point. [Link to Image Description]
Image Source: By OpenStax CC BY 3.0, via OpenStax Microbiology
3.6 Plasma Protein Inflammatory Mediators
Whole blood is composed of cells and formed elements, as well as a fluid called plasma. Within the blood plasma are electrolytes, nutrients and proteins involved in blood clotting and immune response.
Some plasma proteins, called acute phase proteins (APPs), are produced in the liver and become concentrated in the bloodstream during the acute inflammation stage of the immune response. APPs can be analyzed using blood tests for detecting inflammation.
Examples include:
- C-reactive protein – binds the surface of bacteria and promotes their phagocytosis.
- Ferratin and Transferrin – sequester free iron so that it cannot be absorbed and utilized by microbial pathogens.
- Fibrinogen – a protein that, when activated, forms blood clots.
- Mannose-binding lectin – binds sugars on the surface of pathogen cells and induces a complement cascade.
- Complement proteins – Potent immune mediators that both damage target cells as well as induce potent inflammation.
3.7 Complement Proteins
- Classical complement pathway – The complement protein C1 binds to antibodies, initiating a series of events involving activation of C2 and C4, followed by C3.
- Lectin complement pathway – Mannose-binding lectin accumulates on the surface of microbes, where it activates C2 and C4, and subsequently C3.
- Alternative pathway – spontaneously activates C3.
As all three pathways converge on C3 activation, a selection of other complement proteins are engaged to enact the functions of complement.
- Cell lysis – C6, C7, C8 and C9 cooperate to form a pore through the cell membrane. This pore is called the membrane attack complex (MAC). If enough pores form, the target cell lyses, or “bursts”.
- Opsonization – C3b and C4b remain attached to the cell surface, where they enhance the likelihood the cell will be phagocytized.
- Induce inflammation – C3a and C5a are potent inducers of inflammation.
- Chemotaxis – C5a is a potent attractant for neutrophils, causing them to move toward the source of these chemicals through the process of chemotaxis.
Figure 3-5 Complement Activation. Simplified diagram showing the convergence of the three complement pathways. Note that the classical pathway is unique in its requirement for C1q. Both The classical and lectin pathways use C2 and C4. The alternative pathway involves autocatalytic production of C3, with help from Factor B. [Link to Image Description]
Summary
- The immune system responds to infection by:
- Phagocytosis: Specific immune cells may use pattern recognition receptors (PRRs, e.g. TLR) to detect patterns (PAMPs) on a pathogen surface. The pathogen is internalized within the phagocyte, where it is degraded by oxygen-dependent and oxygen-independent mechanisms.
- Cytokine Signalling: Chemical agents are secreted by immune cells to communicate and coordinate the immune response.
- Chemotactic cytokines attract more immune cells to the site of infection.
- Vasoactive cytokines: act on the local blood vessels to support uptake of immune cells from the bloodstream into the infected tissue.
- Acute Localized Inflammation: Blood vessels dilate and blood plasma (fluid) leaks into infected tissues, causing warmth, swelling and redness of the tissues. Pain and scar formation may also be associated with localized inflammation.
- Chronic inflammation: Inflammation that persists over a longer period of time may damage the tissue.
- Systemic Inflammation: Inflammation may occur systemically throughout the body. The most severe systemic inflammation may produce sudden loss of blood pressure, which can result in death if vital organs do not receive sufficient oxygenated blood.
- Complement proteins contribute to each of these immune responses by causing direct cell lysis, but also by promoting phagocytosis, chemotaxis and vasodilation.
Chapter Review
Case Study Review
Pathogen associated molecular patterns (PAMPs) are the general features that our innate immune system uses to identify foreign agents, like bacteria and viruses. PAMPs are different from "antigens", which are molecules that are specifically identified by cells and antibodies of the adaptive immune system.