Chapter 2: Matter, Energy & Life
2.1 The Building Blocks of Life
The Building Blocks of Life
Matter, Elements, Atoms and Molecules

At its most fundamental level, life is made of matter. Matter is something that occupies space and has mass. All matter is composed of elements, substances that cannot be broken down or transformed chemically into other substances. Each element is made of atoms, each with a constant number of protons and unique properties. Each element is designated by its chemical symbol (such as H, N, O, C, and Na), and possesses unique properties. These unique properties allow elements to combine and to bond with each other in specific ways.
An atom is the smallest component of an element that retains all of the chemical properties of that element. For example, one hydrogen atom has all of the properties of the element hydrogen, such as it exists as a gas at room temperature and it bonds with oxygen to create a water molecule (H20). Hydrogen atoms cannot be broken down into anything smaller while still retaining the properties of hydrogen. At the most basic level, all organisms are made of a combination of elements. They contain atoms that combine together to form molecules. Molecules are chemicals made from two or more atoms bonded together. Some molecules are very simple, like oxygen (O2), which is comprised of just two oxygen atoms. Some molecules used by organisms, such as DNA, are made of many millions of atoms. In multicellular organisms, such as animals, molecules can interact to form cells that combine to form tissues, which make up organs. These combinations continue until entire multicellular organisms are formed.
Biological Molecules
Besides water, the molecules necessary for life are organic. Organic molecules are those that contain carbon covalently bonded to hydrogen. In addition, they may contain oxygen, nitrogen, phosphorus, sulfur, and additional elements. There are four major classes of organic molecules: carbohydrates, lipids, proteins, and nucleic acids. Each is an important component of the cell and performs a wide array of functions.
Carbon
It is often said that life is “carbon-based.” This means that carbon atoms, bonded to other carbon atoms or other elements, form the fundamental components of many of the molecules found uniquely in living things. Other elements play important roles in biological molecules, but carbon certainly qualifies as the “foundation” element for molecules in living things. It is the bonding properties of carbon atoms that are responsible for its important role.
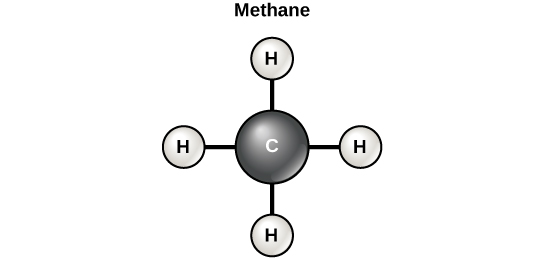
Carbon can form four covalent bonds with other atoms or molecules. The simplest organic carbon molecule is methane (CH4), in which four hydrogen atoms bind to a carbon atom (Figure 1. below).
Lipids include a diverse group of compounds that are united by a common feature. Lipids are hydrophobic (“water-fearing”), or insoluble in water, because they are non-polar molecules(molecules that contain non-polar covalent bonds). Lipids perform many different functions in a cell. Cells store energy for long-term use in the form of lipids called fats. Lipids also provide insulation from the environment for plants and animals. For example, they help keep aquatic birds and mammals dry because of their water-repelling nature. Lipids are also the building blocks of many hormones and are an important constituent of cellular membranes. Lipids include fats, oils, waxes, phospholipids, and steroids.
Proteins are one of the most abundant organic molecules in living systems and have the most diverse range of functions of all macromolecules. They are all polymers of amino acids. The functions of proteins are very diverse because there are 20 different chemically distinct amino acids that form long chains, and the amino acids can be in any order. Proteins can function as enzymes, hormones, contractile fibers, cytoskeleton rods, and much more.
Enzymes are vital to life because they act as catalyst in biochemical reactions (like digestion). Each enzyme is specific for the substrate (a reactant that binds to an enzyme) upon which it acts. Enzymes can function to break molecular bonds, to rearrange bonds, or to form new bonds.
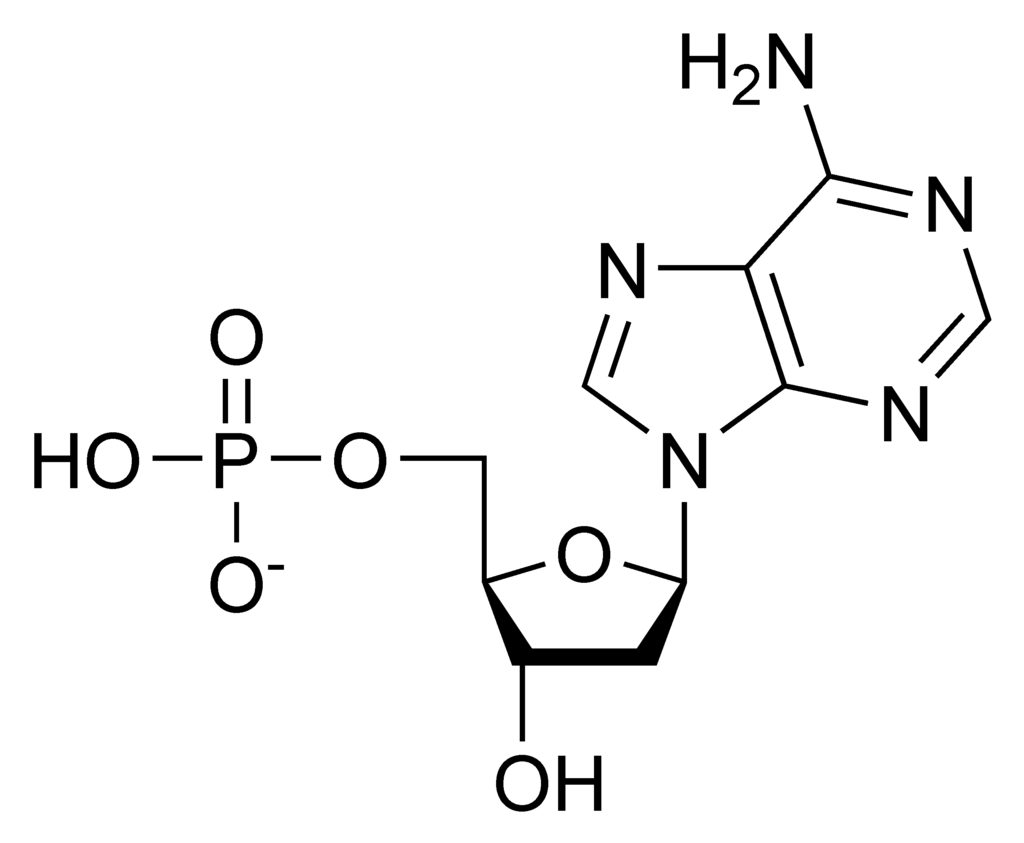
Nucleic acids are very large molecules that are important to the continuity of life. They carry the genetic blueprint of a cell and thus the instructions for its functionality. The two main types of nucleic acids are deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). DNA is the genetic material found in all organisms, ranging from single-celled bacteria to multicellular mammals like humans. The other type of nucleic acid, RNA, is mostly involved in protein synthesis. The DNA molecules never leave the nucleus, but instead use an RNA intermediary to communicate with the rest of the cell. Other types of RNA are also involved in protein synthesis and its regulation. DNA and RNA are made up of small building blocks known as nucleotides. The nucleotides combine with each other to form a polynucleotide: DNA or RNA. Each nucleotide is made up of three components: a nitrogenous base, a pentose (five-carbon) sugar, and a phosphate. DNA has a beautiful double-helical structure (Figure 2 below).

Cells and Cell Theory
Close your eyes and picture a brick wall. What is the basic building block of that wall? It is a single brick, of course. Like a brick wall, your body is composed of basic building blocks and the building blocks of your body are cells. Your body has many kinds of cells, each specialized for a specific purpose. Just as a home is made from a variety of building materials, the human body is constructed from many cell types. For example, bone cells help to support and protect the body. Cells of the immune system fight invading bacteria, and red blood cells carry oxygen throughout the body. Each of these cell types plays a vital role during the growth, development, and day-to-day maintenance of the body. In spite of their enormous variety, however, all cells share certain fundamental characteristics.
The microscopes we use today are far more complex than those used in the 1600s by Antony van Leeuwenhoek, a Dutch shopkeeper who had great skill in crafting lenses. Despite the limitations of his now-ancient lenses, van Leeuwenhoek observed the movements of single-celled organism and sperm, which he collectively termed “animalcules.” In a 1665 publication called Micrographia, experimental scientist Robert Hooke coined the term “cell” (from the Latin cella, meaning “small room”) for the box-like structures he observed when viewing cork tissue through a lens. In the 1670s, van Leeuwenhoek discovered bacteria and protozoa. Later advances in lenses and microscope construction enabled other scientists to see different components inside cells.
By the late 1830s, botanist Matthias Schleiden and zoologist Theodor Schwann were studying tissues and proposed the unified cell theory, which states that all living things are composed of one or more cells, that the cell is the basic unit of life, and that all new cells arise from existing cells. These principles still stand today. There are many types of cells, and all are grouped into one of two broad categories: prokaryotic and eukaryotic. Animal, plant, fungal, and protist cells are classified as eukaryotic, whereas bacteria and archaea cells are classified as prokaryotic.
All cells share four common components: 1) a plasma membrane, an outer covering that separates the cell’s interior from its surrounding environment; 2) cytoplasm, consisting of a jelly-like region within the cell in which other cellular components are found; 3) DNA, the genetic material of the cell; and 4) ribosomes, particles that synthesize proteins. However, prokaryotes differ from eukaryotic cells in several ways.
Components of Prokaryotic Cells
A prokaryotic cell is a simple, single-celled (unicellular) organism that lacks a nucleus, or any other membrane-bound organelle. We will shortly come to see that this is significantly different in eukaryotes. Prokaryotic DNA is found in the central part of the cell: a darkened region called the nucleoid (Figure 5 below).

Unlike Archaea and eukaryotes, bacteria have a cell wall made of peptidoglycan (molecules comprised of sugars and amino acids) and many have a polysaccharide capsule. The cell wall acts as an extra layer of protection, helps the cell maintain its shape, and prevents dehydration. The capsule enables the cell to attach to surfaces in its environment. Some prokaryotes have flagella, pili, or fimbriae. Flagella are used for locomotion. Pili are used to exchange genetic material during a type of reproduction called conjugation. Fimbriae are protein appendages used by bacteria to attach to other cells.
Eukaryotic Cells
A eukaryotic cell is a cell that has a membrane-bound nucleus and other membrane-bound compartments or sacs, called organelles, which have specialized functions (see Figure 6). The word eukaryotic means “true kernel” or “true nucleus,” alluding to the presence of the membrane-bound nucleus in these cells. The word “organelle” means “little organ,” and, as already mentioned, organelles have specialized cellular functions, just as the organs of your body have specialized functions.

Cell Size
At 0.1–5.0 µm (micrometre) in diameter, most prokaryotic cells are significantly smaller than eukaryotic cells, which have diameters ranging from 10–100 µm (Figure 7 below). The small size of prokaryotes allows ions and organic molecules that enter them to quickly spread to other parts of the cell. Similarly, any wastes produced within a prokaryotic cell can quickly move out. However, larger eukaryotic cells have evolved different structural adaptations to enhance cellular transport. Indeed, the large size of these cells would not be possible without these adaptations. In general, cell size is limited because volume increases much more quickly than does cell surface area. As a cell becomes larger, it becomes more and more difficult for the cell to acquire sufficient materials to support the processes inside the cell, because the relative size of the surface area through which materials must be transported declines.

Animal Cells versus Plant Cells
Despite their fundamental similarities, there are some striking differences between animal and plant cells. Animal cells have centrioles, centrosomes, and lysosomes, whereas plant cells do not. Plant cells have a cell wall, chloroplasts, plasmodesmata, and plastids used for storage, and a large central vacuole, whereas animal cells do not.
The Cell Wall
In Figure 8 below, the diagram of a plant cell shows a structure external to the plasma membrane called the cell wall. The cell wall is a rigid covering that protects the cell, provides structural support, and gives shape to the cell. Fungal and protist cells also have cell walls. While the chief component of prokaryotic cell walls is peptidoglycan, the major organic molecule in the plant cell wall is cellulose, a polysaccharide made up of long, straight chains of glucose units. When nutritional information refers to dietary fibre, it is referring to the cellulose content of food.

Chloroplasts
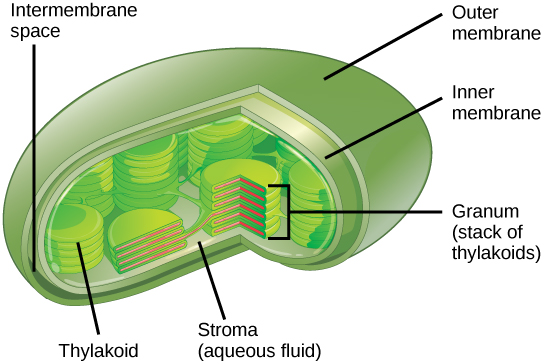
Chloroplasts function in photosynthesis and can be found in eukaryotic cells such as plants and algae. In photosynthesis, carbon dioxide, water, and light energy are used to make glucose and oxygen. This is the major difference between plants and animals: plants are able to make their own food, like glucose, whereas animals must rely on other organisms for their organic compounds or food source. Chloroplasts have outer and inner membranes, but within the space enclosed by a chloroplast’s inner membrane is a set of interconnected and stacked, fluid-filled membrane sacs called thylakoids (Figure 9 below). Each stack of thylakoids is called a granum (plural = grana). The fluid enclosed by the inner membrane and surrounding the grana is called the stroma.
Embedded in the thylakoid membrane are molecules of chlorophyll, a pigment (a molecule that absorbs light) through which the entire process of photosynthesis begins.
Attribution
Essentials of Environmental Science by Kamala Doršner is licensed under CC BY 4.0. Modified from the original.
Attribution
Concepts of Biology by OpenStax is licensed under CC BY 4.0. Modified from original.

