Chapter V: Solid Liquid Separation
Quiz Solutions (Chapters IV & V)
1. A Fe-S-H2O Eh-pH diagram is provided below. The iron-sulfur species is pyrite (FeS2). Pyrite itself is not valuable in hydrometallurgy, but it may contain finely disseminated gold. Then pyrite can be leached to free the gold, which can then be leached with cyanide. Pyrite leaching can be accomplished using near-ambient conditions (bioleaching using sulfur-oxidizing bacteria) through to autoclave leaching at >200°C.
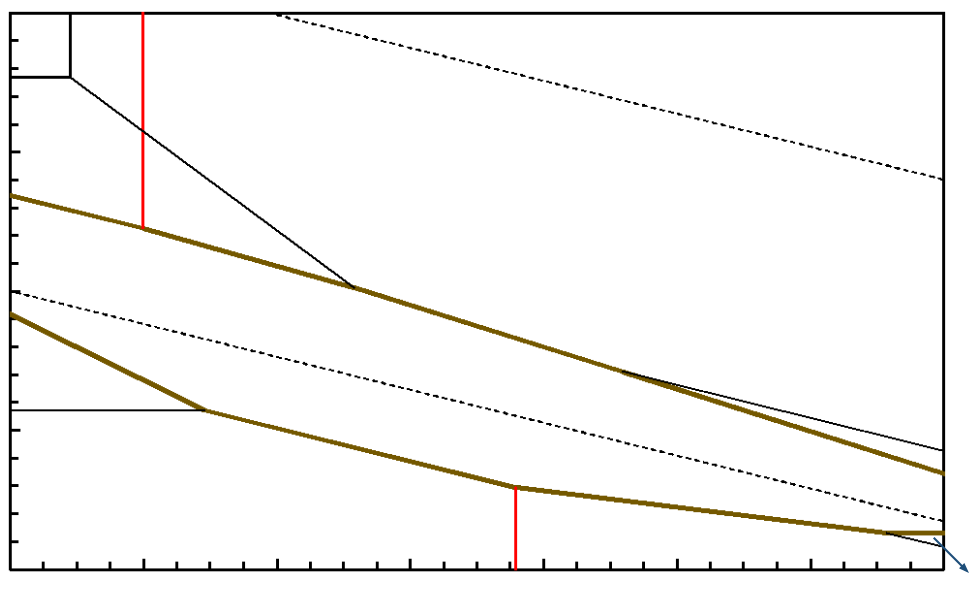
(a) Indicate which of these (if any) involve pyrite oxidation, and which involve pyrite reduction.
Fe+2, H2S g (or H2S aq) Reduction: FeS2 + 4H+ + 2e- = Fe+2 + 2H2S
Fe+2, HSO4- Oxidation: FeS2 + 8H2O = Fe+2 + 2HSO4- + 14H+ + 14e-
Fe+2, SO42- Oxidation: FeS2 + 8H2O = Fe+2 + 2SO42- + 16H+ + 14e-
Fe+3, HSO4- Oxidation: FeS2 + 8H2O = Fe+3 + 2HSO4- + 14H+ + 15e-
(Reduction or oxidation can be determined by writing half reactions for the processes.)
(b) The ferric-ferrous couple in sulfate medium has an E° of 0.68 V; use this value. (In the diagram EhFe+3/Fe+2 is closer to 0.77 V, which would pertain in the absence of sulfate.) If the ratio of Fe+3-to-Fe+2 concentrations is 2, determine the Eh of the ferric-ferrous couple, Eh for pyrite leaching to form ferrous and the appropriate sulfur species at pH 1 and ΔE for the leaching reaction. From the table below select the appropriate half reaction involving pyrite. Write your answers to 3 significant digits.
| Eh = Slope x pH + C | Slope | C = Constant |
| Fe+2/Fe | 0 | -0.4285 |
| Fe+3/Fe+2 | 0 | 0.68 |
| Fe+2, S/FeS2 | 0 | 0.4317 |
| Fe+2, HSO4-/FeS2 | -0.05916 | 0.3444 |
| Fe+2, SO42-/FeS2 | -0.06761 | 0.3613 |
| Fe(OH)2, SO42-/FeS2 | -0.07606 | 0.4094 |
| FeS2/Fe+2, H2S | -0.1183 | -0.08390 |
\[
\ce{Fe^{3+} + e^- -> Fe^{2+}}
\]
\[
E_{h,\ce{Fe^{3+}/Fe^{2+}}} = E^\circ - \frac{2.303RT}{nF} \log\!\left(\frac{[\ce{Fe^{2+}}]}{[\ce{Fe^{3+}}]}\right)
\]
\[
E_h = 0.68 - \frac{2.303 \times 8.314 \times 298.15}{1 \times 96485} \log\!\left(\frac{1}{2}\right)
\]
\[
E_{h,\ce{Fe^{3+}/Fe^{2+}}} = \textcolor{blue}{0.698}~\text{V}
\]
The other half reaction that applies is for leaching of FeS2 to form Fe+2 and HSO4- at pH 1: Fe+2, HSO4-/FeS2
\[ E_h = -0.05916\,\text{pH} + 0.3444 = \textcolor{blue}{0.285}~\text{V} \]
\[ \Delta E = 0.698 - 0.285 = \textcolor{blue}{0.413}~\text{V} \]
The reaction is favorable since ΔE > 0.
(ii) If the ratio of Fe+3-to-Fe+2 concentrations is 10-6 is the reaction still thermodynamically favorable? Why would leaching under these conditions be unsuitable in practice?
\[ E_{h,\ce{Fe^{3+}/Fe^{2+}}} = 0.68 - \frac{2.303 \times 8.314 \times 298.15}{1 \times 96485} \log\!\left(\frac{1}{10^{-6}}\right) = 0.325~\text{V} \]
\[ \Delta E = 0.325 - 0.285 = 0.040~\text{V} > 0 \quad \text{(favorable)} \]
But, the reaction is only slightly favorable, so there is not a strong driving force for leaching to occur, so this will make the reaction slow. Second, the concentration of ferric is so low that together with the low ΔE, the reaction would be extremely slow; too slow to be practical.
(c) According to the diagram what is the maximum pH at which it is reasonable to expect to be able to use a ferric salt to leach pyrite?
The pH beyond which ferric is no longer dominant is the maximum pH under these conditions. This corresponds to pH = 0.90. At pH > 0.90 Fe(OH)3 s is dominant.
2. There are many variations on the leaching step in the Roast-Leach-Electrowin process for zinc. Many of the differences centre on how iron is rejected from the leach solution. The concentrate is roasted to obtain zinc calcine. The concentrate contains ZnS, FeS2 and perhaps other iron minerals, zinc silicates/carbonates, and low levels of lead and copper sulfides: up to 55% Zn (by weight), <10% Fe, <2% Cu, <3% Pb, as well as silicates. Traces of precious metals may also be present. The calcine is introduced in the neutral leach stage, so called because the pH is only weakly acidic (typically ~4.5). This readily dissolves most of the ZnO, but not the ZnFe2O4 that forms as a result of reaction between FeS2 and ZnS in the roaster. The necessary acid is mostly generated in electrowinning by oxidation of water at the anodes. Following the neutral leach iron is precipitated as a jarosite compound. These have the general formula, M(I)Fe3(SO4)(OH)6, where M(I) is a singly charged cation (e.g. Na+, K+, NH4+, H3O+). The K+ form is the least soluble, but the ammonium form costs less to produce. (Note the exception to the rule about alkali metal and ammonium salts solubilities.) Jarosites are a type of basic ferric sulfate salt. Conditions suitable for jarosite precipitation are ~95°C and pH 1.5; this is not sufficient to leach zinc ferrite. Zinc ferrite is leached in the hot acid stage (and possibly also in a subsequent, even more forcing stage - not included here) which uses temperatures of ~90°C and strong acid (pH ~1). The ammonium jarosite precipitate is formed after the hot acid leach stage in the flowsheet below.
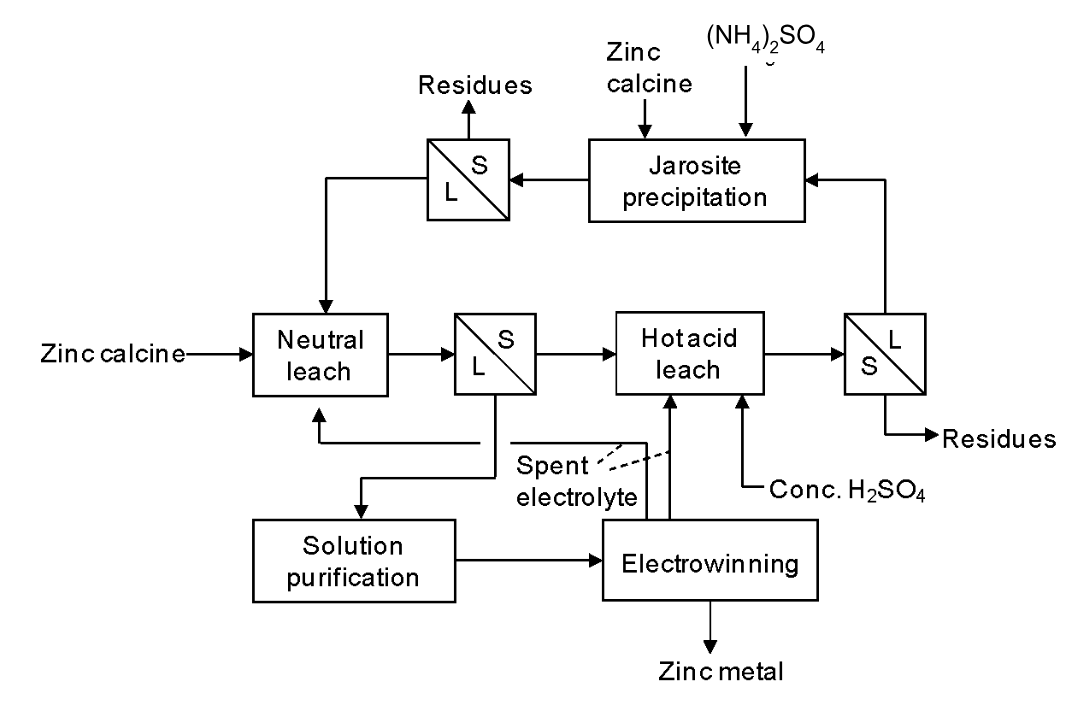
(a) Is this a co-current or counter-current leaching process? Explain your answer.
The process is counter-current. (i) The solids flow in opposite direction to the solutions; (ii) There is a solid-liquid separation stage after each leach stage.
(b) Why is zinc calcine added to the jarosite precipitation stage?
Zinc calcine is added here as a base to neutralize excess acid.
(c) What will happen to the zinc ferrite that is added with the calcine in the jarosite precipitation stage? What is the downside of this?
The pH in the jarosite precipitation step is too high to dissolve zinc ferrite. It passes through to the residues after solid-liquid separation and is lost. This lowers recovery a little. (The amount of calcine used is small.)
3. (i) Would you expect to be able to leach PbS in dilute chloride medium? What about in a very concentrated chloride solution? Briefly explain your answers.
In dilute chloride solution, NO; PbCl2 is insoluble (sparingy soluble) in aqueous solution; Ksp << 1. In a concentrated chloride solution, YES; stable [PbCln]2-n complexes can form and keep Pb(II) in solution.
(ii) Would you expect to be able to leach PbS in a sulfate medium? Briefly explain your answer.
NO; PbSO4 is insoluble (sparingly soluble) in aqueous solution.
4. Nickel and cobalt may be produced by leaching of laterite ores. These are oxidized, near-surface deposits containing oxides, hydroxides and aluminosilicates. Nickel may be present in a (mainly) goethite matrix, Fe(O)OH, at 1-2% grade. Cobalt is usually present at roughly 1/10 this amount. Ores are leached in autoclaves at high pressures and temperatures (≥250°C) using strongly acidic H2SO4 solution. Goethite is converted into hematite, Fe2O3, and in the process nickel and cobalt are leached into solution. After leaching and cooling the slurry is typically subjected to solid-liquid separation via counter-current decantation. This is successfully practiced at Moa Bay, Cuba. (In this case the clarified solution is then treated with hydrogen sulfide to precipitate a Ni-Co-Cu bulk "concentrate," which is shipped to Fort Saskatchewan for conversion into pure nickel and cobalt.) Other nickel laterite processes purify the clarified solution and then recover nickel and cobalt by electrowinning.
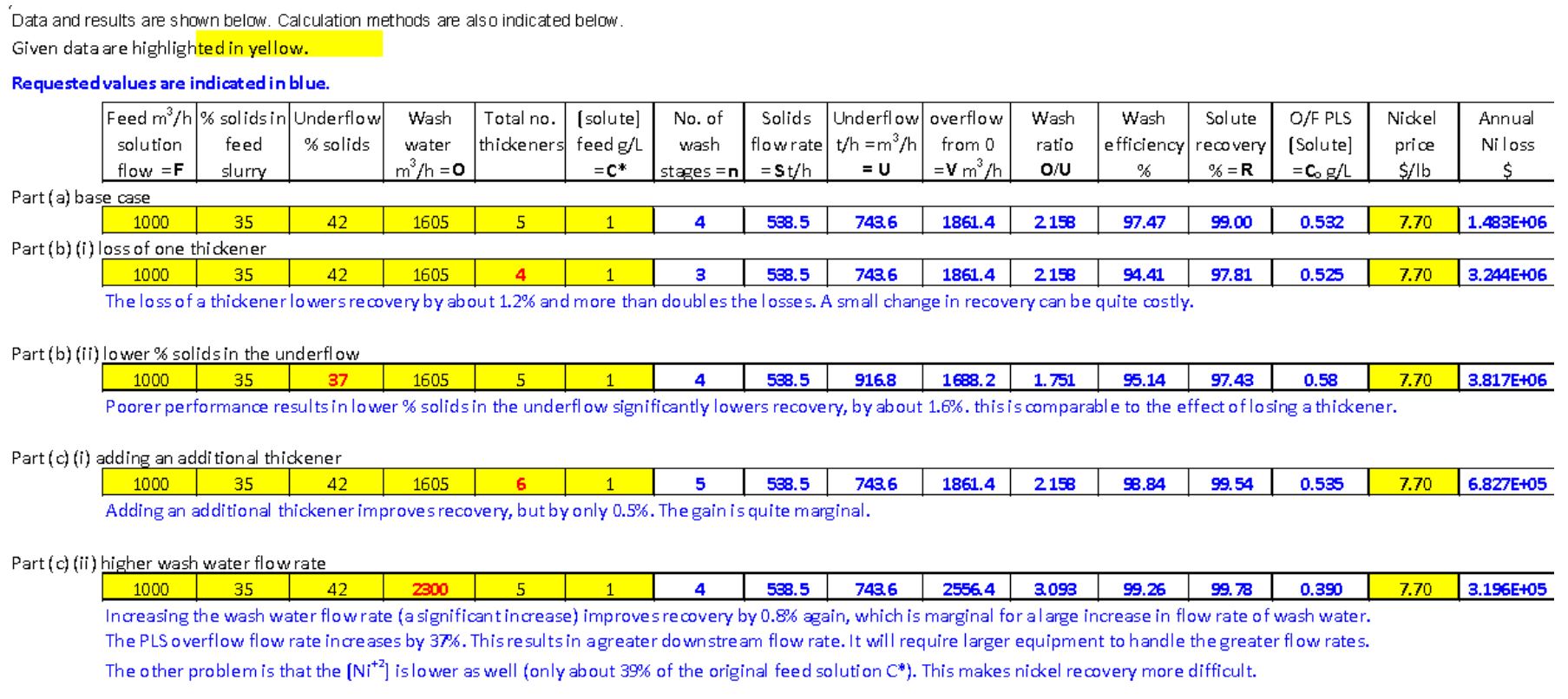
Do all questions using Excel. Handwritten answers will not be accepted, except where otherwise indicated. Show example calculations for your work. (No marks without illustrative calculations.) For parts (b) and (c), calculate the answers using Excel and provide quantitative answers in comparison with part (a).
Use the following symbols and terms as required:
S = solids mass flow rate
O = wash water flow rate
n = number of wash stages
C* = solute concentration in feed solution
F = feed solution flow rate
U = underflow solution flow rate
O/U = wash ratio
V = overflow solution (final PLS) flow rate
Wash efficiency (as per course notes)
R = recovery of solute in %
Co = Solute concentration in lead thickener solution
Dilution of feed = ratio of solute concentration in lead thickener overflow / solute concentration in feed solution
Cn = solute concentration in final underflow solution
Assume all solutions have a density of 1 kg/L (this is an oversimplification for concentrated solutions).
(a) There are a total of 5 thickeners in the CCD circuit. The aqueous solution flow rate associated with the feed slurry to CCD washing is 1000 m3/h. The feed slurry has 35% solids; the feed solution nickel concentration is 1 g/L. The solids content associated with the underflows from each thickener is 42% by weight. The wash water flow rate is 1605 m3/h. Determine the following quantities and also report appropriate units. Report your answers in a table.
Media Attributions
- Screenshot 2026-05-01 045101
- Screenshot 2026-05-01 045113
- Screenshot 2026-05-01 045047

