Water is a versatile solvent
Water is the solvent for most of life’s chemical reactions. A solution is a uniform mixture of liquid and substances dissolved in that liquid. A solution consists of a solvent or liquid and a solute or substance dissolved in the solvent.
Solution = solute(s) + solvent
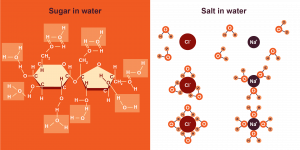
Water is a polar molecule and a polar solvent. This means that water will dissolve other polar molecules as well as charged molecules. One example of a polar molecule is table sugar or sucrose (C12H22O11). Sucrose stirred into water dissolves easily because both sucrose and water are polar molecules and the partial charges on the atoms of those molecules interact with one another favourably. The covalent bonds within sucrose are not easily weakened by water and remain intact when sucrose dissolves.
Charged molecules, such as table salt or sodium chloride (NaCI) also interact favourably with water. The partial negative charges on the oxygen atoms of water molecules attract the positively charged sodium ions; the partial positive charges on the hydrogen atoms of water molecules attract the negatively charged chloride ions. Because the partial charges of water can disrupt the electrostatic interactions between ions, ionic bonds are easily weakened by water. Therefore, water molecules tend to disrupt ionic bonds between ions and surround ions in solution, forming a hydration sphere.
Substances that interact favourably with water are called hydrophilic. Polar and charged solutes are hydrophilic and tend to dissolve easily in water.
In contrast, nonpolar molecules bear no partial charges or full charges to interact favourably with partial charges on water molecules. Therefore, nonpolar molecules are called hydrophobic. Nonpolar solutes do not dissolve in water easily.
Chemistry in the clinic:
Colloids are suspensions of substances in a solvent-like liquid. Colloids differ from solutions by the size of the particle in the liquid. If the substance contains particles that measure between 1 nm and 1 μm (~ size of a bacterial cell), then the mixture is a colloid and not a solution. If the particles are smaller than 1 nm, then the mixture is a solution. Blood plasma, the liquid component of blood, is a colloid that contains relatively large proteins that are required for clotting. You will learn about the consequences of the colloidal nature of blood in a future lecture on the vascular system and hemodynamics.
1 nm = 1 x 10-9 m
1 μm = 1 x 10-6 m
Derived from Greek: “hydros” = water; “philos” = love; water-loving
Derived from Greek: “hydros” = water; “phobos” = fear; water-fearing

