Unit 8: Cellular Respiration and Energy Metabolism
Unit outline
Part 1: Overview of Metabolic Reactions
Part 2: Carbohydrate Metabolism
Part 5: Metabolic States of the Body
Practice Questions
Learning Objectives
At the end of this unit, you should be able to:
I. Describe the overall connection between nutrients, metabolism and homeostasis.
II. Describe the process of cellular respiration in general terms.
III. Describe the roles of ATP, NAD, and FAD in energy metabolism in the cell.
IV. Describe the process of glycolysis.
V. Describe the formation of acetyl coenzyme A from pyruvic acid.
VI. Explain the role of the Krebs cycle in cellular respiration.
VII. Describe the role of the electron transport chain in cellular respiration.
VIII. Describe the major steps in the generation of ATP by chemiosmosis.
IX. Summarize the ATP produced from the breakdown of a single glucose molecule.
X. Describe the importance of oxygen (O2) in cellular respiration and compare aerobic respiration with lactic acid fermentation.
XI. Describe the importance of carbohydrates, lipids and proteins in energy storage and energy availability, and their use during starvation conditions.
XII. Describe the importance of glucose in cellular respiration and ATP production.
XIII. Describe the role of lipids and amino acids in ATP production.
XIV. Describe the role of ketone bodies in energy metabolism.
XV. Describe the relationship between gluconeogenesis, lipid metabolism, and protein catabolism.
XVI. Describe the fate of amino acids that are metabolized for ATP production.
XVII. Explain the importance of appropriate nutrient intake for maintaining homeostasis of the body.
Learning Objectives and Guiding Questions
At the end of this unit, you should be able to complete all the following tasks, including answering the guiding questions associated with each task.
I. Describe the overall connection between nutrients, metabolism and homeostasis.
II. Describe the process of cellular respiration in general terms.
- Define the term “cellular respiration”.
- What is the main biological function of cellular respiration?
- Determine and write out the overall chemical equation for aerobic cellular respiration.
III. Describe the roles of ATP, NAD, and FAD in energy metabolism in the cell.
- Use complete sentences to describe how cells produce:
- ATP
- NADH
- FADH2
- Use complete sentences to describe the biological purpose of a cell producing:
-
- ATP
- NADH
- FADH2
IV. Describe the process of glycolysis.
V. Describe the formation of acetyl coenzyme A from pyruvic acid.
VI. Explain the role of the Krebs cycle in cellular respiration.
VII. Describe the role of the electron transport chain in cellular respiration.
VIII. Describe the major steps in the generation of ATP by chemiosmosis.
- Write a single-sentence summary of the chemical events that occur during each of the following processes:
- Glycolysis
- Pyruvic acid oxidation
- The Krebs (citric acid) cycle
- The electron transport chain
- Substrate-level phosphorylation
- Oxidative phosphorylation
- Specify the molecules that are required, consumed, and produced during each of the following processes:
- Glycolysis
- Pyruvic acid oxidation
- The Krebs (citric acid) cycle
- The electron transport chain
- Starting with the arrival of NADH and FADH2 at the electron transport chain, thoroughly describe how the electron transport chain is used to generate ATP.
IX. Summarize the ATP produced from the breakdown of a single glucose molecule.
- At which point(s) during aerobic cellular respiration of one glucose molecule are ATP molecules produced by each of the following processes, and how many ATP molecules are produced by each process?
-
- Substrate-level phosphorylation
- Oxidative phosphorylation
X. Describe the importance of oxygen (O2) in cellular respiration and compare aerobic respiration and lactic acid fermentation.
- For what single main function is oxygen required during cellular respiration?
- In the absence of oxygen, how many molecules of ATP can be produced from a single glucose molecule?
- Explain why, in the absence of oxygen, the continued generation of ATP from glucose requires the conversion of pyruvic acid to lactic acid.
XI. Describe the importance of carbohydrates, lipids and proteins in energy storage and energy availability, and their use during starvation conditions.
- Describe and explain the use of carbohydrates, lipids, and proteins for ATP production when in:
- An absorptive (fed) state.
- A postabsorptive (fasting) state.
- Starvation conditions.
- Protein molecules contain approximately the same amount of energy per gram as carbohydrates and are found extensively throughout the human body. Explain why it is physiologically important that proteins are used as major sources of chemical energy only after other energy-containing molecules (i.e., carbohydrates and lipids) have been depleted.
XII. Describe the importance of glucose in cellular respiration and ATP production.
- Which specific nutrient molecule are all human body cells normally capable of breaking down to generate ATP?
XIII. Describe the role of lipids and amino acids in ATP production.
- What other nutrient molecules are at least some human body cells capable of breaking down to generate ATP? For each of these nutrient molecules, which body cell types can (or cannot) break it down?
XIV. Describe the role of ketone bodies in energy metabolism.
- What types of molecules can be used to produce ketone bodies?
- Under what conditions should ketone bodies be produced?
- What function do ketone bodies serve in the human body?
XV. Describe the relationship between gluconeogenesis, lipid metabolism, and protein catabolism.
XVI. Describe the fate of amino acids that are metabolized for ATP production.
- Name and describe with a one-sentence summary the mechanism(s) that are used to allow body cells to continue generating ATP in the event that:
- Blood glucose levels decline
- Glycogen stores in the body decline
- Lipid stores in the body decline
- Oxygen is unavailable
- Explain the functional reason why, under conditions of low oxygen availability, lactic acid (or lactate) must be produced to allow glycolysis to continue.
- Clearly define each of the following terms:
- Glycolysis
- Glycogenesis
- Gluconeogenesis
- Glycogenolysis
- Describe the process in the human body by which some of the energy present in lipid molecules can be used to generate ATP.
- In which organ(s) and/or cell type(s) can this process occur?
- Which major steps are involved?
- Can any of the intermediate molecules be transported to other tissues in a form that will allow the receiving tissues to generate ATP in the absence of glucose?
- At what stage(s) of cellular respiration can the breakdown products of lipid molecules be used?
- Describe the process in the human body by which some of the energy present in amino acids can be used to generate ATP.
-
- In which organ(s) and/or cell type(s) can this process occur?
- Which major step are involved?
- At what stage(s) of cellular respiration can the breakdown products of amino acids be used?
- In breaking down amino acids, what potentially toxic chemical is produced that is not produced when a lipid or carbohydrate is broken down? What is the fate of this product?
- What are the potentially detrimental physiological consequences of breaking down amino acids, rather than glucose, to produce ATP?
XVII. Explain the importance of appropriate nutrient intake for maintaining homeostasis of the body.
- List the classes of nutrients that can be broken down to release energy that can be used to produce ATP.
- For each of the following chemicals, describe its function in metabolism and name the specific nutrient(s) that must be ingested to produce it:
-
- Pyruvate dehydrogenase
- Nicotinamide adenine dinucleotide (NAD+)
- Flavin adenine dinucleotide (FAD)
- Coenzyme A
Part 1: Overview of Metabolic Reactions
Metabolism
Metabolic processes are constantly taking place in the body. Metabolism is the sum of all of the chemical reactions that are involved in catabolism and anabolism. The reactions governing the breakdown of food to obtain energy are called catabolic reactions. In catabolic reactions, large organic molecules are broken down to smaller molecules, releasing the energy contained in the chemical bonds. Some of this energy is used to form adenosine triphosphate (ATP). Conversely, anabolic reactions use the energy released by catabolic reactions to synthesize larger molecules from smaller ones, such as when the body forms proteins by stringing together amino acids. Both sets of reactions are critical to maintaining life.
The energy obtained from ATP drives all bodily functions, such as contracting muscles, maintaining the electrical potential of nerve cells, and absorbing food in the gastrointestinal tract. The metabolic reactions that produce ATP come from various sources (Figure 1).
Because catabolic reactions produce energy and anabolic reactions use energy, ideally, energy usage would balance the energy released. If the net energy change is positive (catabolic reactions release more energy than the anabolic reactions use), then the body stores the excess energy by building fat molecules for long-term storage. On the other hand, if the net energy change is negative (catabolic reactions release less energy than anabolic reactions use), the body uses stored energy to compensate for the deficiency of energy released by catabolism.
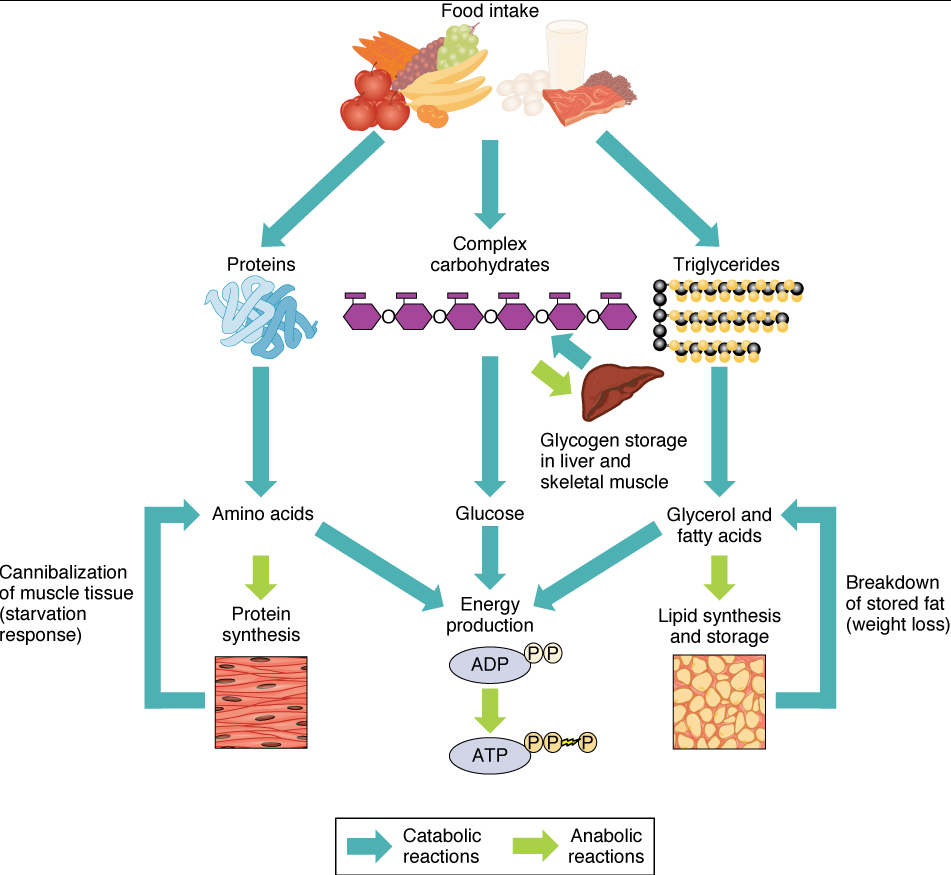
Of the four major macromolecular groups (carbohydrates, lipids, proteins, and nucleic acids) that are processed by digestion, carbohydrates are the most common source of energy to fuel the body. They take the form of either complex carbohydrates, polysaccharides like starch and glycogen, or simple sugars (monosaccharides) like glucose and fructose. Among the monosaccharides, glucose is the most common fuel for ATP production in cells, and as such, there are a number of endocrine control mechanisms to regulate glucose concentration in the bloodstream. Excess glucose is either stored as an energy reserve in the liver and skeletal muscles as the complex polymer glycogen, or it is converted into fat (triglyceride) in adipose cells (adipocytes).
Among the lipids (fats), triglycerides are most often used for energy via a metabolic process called β-oxidation. About one-half of excess fat is stored in adipocytes that accumulate in the subcutaneous tissue under the skin, whereas the rest is stored in adipocytes in other tissues and organs.
Proteins, which are polymers, can be broken down into their monomers, individual amino acids. Amino acids can be used as building blocks of new proteins or broken down further for the production of ATP. When one is chronically starving, this use of amino acids to obtain energy can lead to a wasting away of the body as more and more proteins are broken down.
Nucleic acids are present in most of the foods you eat. During digestion, nucleic acids including DNA and various RNAs are broken down into their constituent nucleotides. These nucleotides are readily absorbed and transported throughout the body to be used by individual cells during nucleic acid metabolism.
Nutrition and Diet
The carbohydrates, lipids, and proteins in the foods you eat are used for energy to power molecular, cellular, and organ system activities. Importantly, the energy is stored primarily as fats. The quantity and quality of food that is ingested, digested, and absorbed affects the amount of fat that is stored as excess calories. A dietary nutrient is a substance that must be ingested and is essential for growth and the maintenance of life. Diet—both what you eat and how much you eat—has a dramatic impact on your health. Eating too much or too little food can lead to serious medical issues, including cardiovascular disease, cancer, and diabetes, among others. Combine an unhealthy diet with unhealthy environmental conditions, such as smoking, and the potential medical complications increase significantly.
Part 2: Carbohydrate Metabolism
Carbohydrates are organic molecules composed of carbon, hydrogen, and oxygen atoms. The family of carbohydrates includes both sugars (i.e. monosaccharides and disaccharides) and polysaccharides. Glucose and fructose are examples of sugars, and starch, glycogen, and cellulose are all examples of polysaccharides. Polysaccharides are made of multiple monosaccharide molecules. Polysaccharides serve as energy storage (e.g., starch and glycogen) and as structural components (e.g., chitin in insects and cellulose in plants).
During digestion, carbohydrates are broken down into simple, soluble sugars that can be transported across the intestinal wall into the circulatory system to be transported throughout the body. Carbohydrate digestion begins in the mouth with the action of salivary amylase on starches and ends with monosaccharides being absorbed across the epithelium of the small intestine. Once the absorbed monosaccharides are transported to the tissues, the process of cellular respiration begins (Figure 2). This section will focus first on glycolysis, a process where the monosaccharide glucose is oxidized, releasing the energy stored in its bonds to produce ATP.
Glycolysis
Glucose is the body’s most readily available source of energy. After digestive processes break polysaccharides down into monosaccharides, including glucose, the monosaccharides are transported across the wall of the small intestine and into the circulatory system, which transports them to the liver. In the liver, hepatocytes either pass the glucose on through the circulatory system or store excess glucose as glycogen. Cells in the body take up the circulating glucose in response to insulin and, through a series of reactions called glycolysis, transfer some of the energy in glucose to a new bond between adenosine diphosphate (ADP) and a third phosphate group to form adenosine triphosphate (ATP) (Figure 3). The last step in glycolysis produces the product pyruvate.
Glycolysis can be expressed as the following equation:
Glucose + 2ATP + 2NAD+ + 4ADP + 2Pi → 2 Pyruvate + 4ATP + 2NADH + 2H+
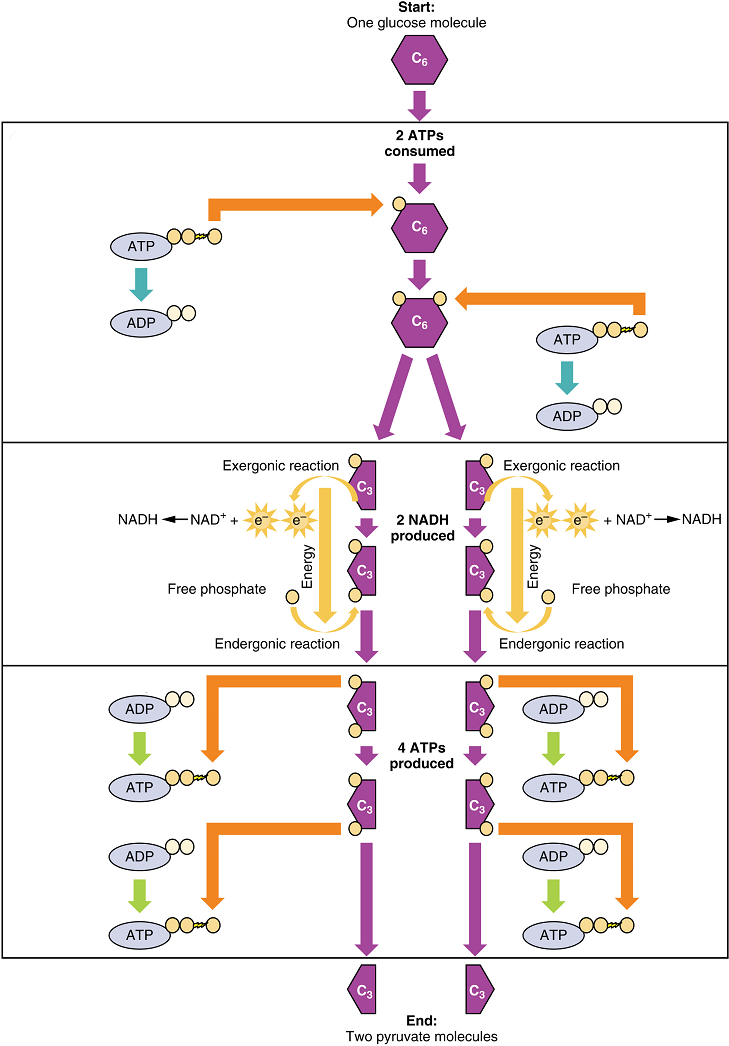
This equation states that glucose – in combination with ATP (a source of chemical energy), nicotinamide adenine dinucleotide (NAD+, a coenzyme that serves as an electron acceptor), and inorganic phosphate – breaks down into two pyruvate molecules, generating four ATP molecules – for a net yield of two ATP – and two energy-containing NADH coenzyme molecules (resulting from adding a hydrogen atom and an extra electron to NAD+). The NADH that is produced in this process will be used later to produce ATP in the mitochondria. Importantly, by the end of this process, one glucose molecule generates two pyruvate molecules, two high-energy ATP molecules, and two electron-carrying NADH molecules.
Glycolysis can be divided into two phases: energy consuming (also called chemical priming) and energy yielding. The first phase is the energy-consuming phase, so it requires two ATP molecules to start the reaction for each molecule of glucose. At the end of this phase, the six-carbon sugar is split to form two phosphorylated three-carbon sugars, glyceraldehyde-3-phosphate (G3P) and dihydroxyacetone phosphate (DHAP). DHAP is then converted into glyceraldehyde-3-phosphate.
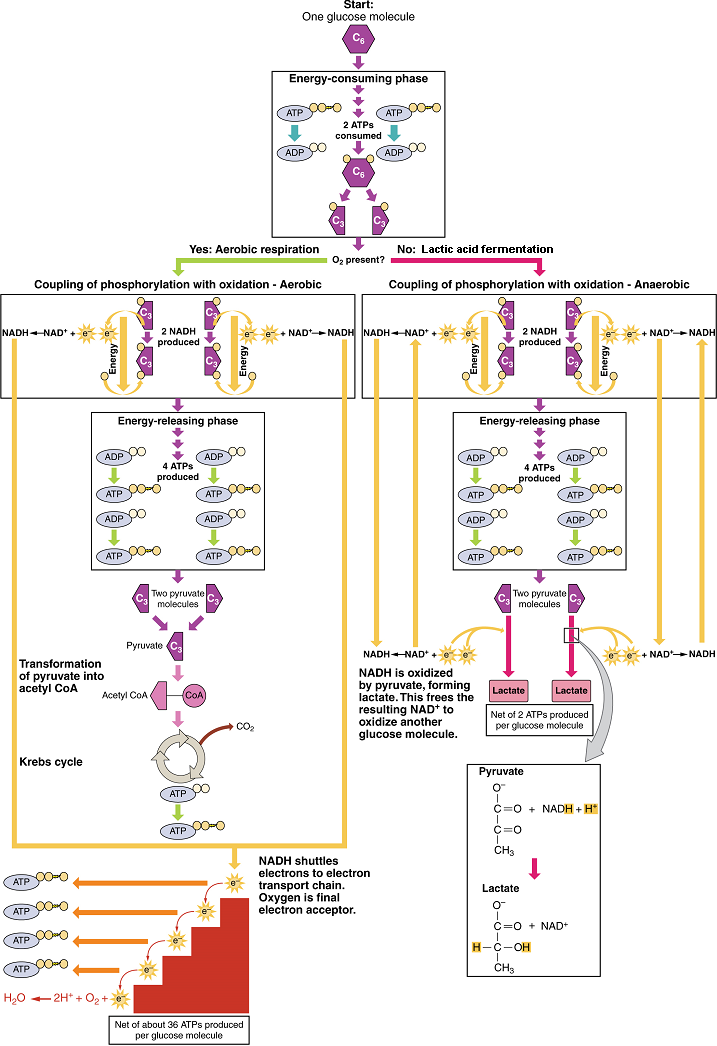
The second phase of glycolysis, the energy-yielding phase, harvests the energy contained in G3P, which is further phosphorylated and oxidized. During this step an electron is released that is then picked up by NAD+ to create an NADH molecule. NADH is a high-energy molecule, like ATP, but unlike ATP, it is not used as energy currency by the cell. Because there are two glyceraldehyde-3-phosphate molecules, two NADH molecules are synthesized during this step. In a series of reactions leading to pyruvate, the two phosphate groups are then transferred from the molecule to which they are attached to two ADPs to form two ATPs by the process of substrate-level phosphorylation (direct phosphorylation). Thus, glycolysis uses two ATPs but generates four ATPs, yielding a net gain of two ATPs and two molecules of pyruvate. In the presence of oxygen, pyruvate continues on to the Krebs cycle (also called the citric acid cycle or tricarboxylic acid cycle (TCA), where additional energy is extracted and passed on, converted into lactic acid by fermentation; or used later for the synthesis of glucose through gluconeogenesis.
Anaerobic Conditions: When oxygen (O2) is limited or absent, pyruvate enters an anaerobic pathway. In these reactions, pyruvate can be converted into lactic acid. This pathway serves to oxidize NADH into the NAD+ needed by glycolysis. In this reaction, pyruvate replaces oxygen as the final electron acceptor. It accepts the electrons from the NADH produced from glycolysis, regenerating NAD+, and is reduced to form lactic acid. This lactic acid fermentation occurs in most cells of the body when oxygen is limited or mitochondria are absent or nonfunctional. For example, because erythrocytes (red blood cells) lack mitochondria, they must produce their ATP from lactic acid fermentation. This is an effective pathway of ATP production for short periods of time, ranging from seconds to a few minutes. The lactic acid produced diffuses into the plasma and is carried to the liver, where it is converted back into pyruvate or glucose. Similarly, when a person exercises, muscles use ATP faster than oxygen can be delivered to them. They depend on glycolysis and lactic acid production for rapid ATP production.
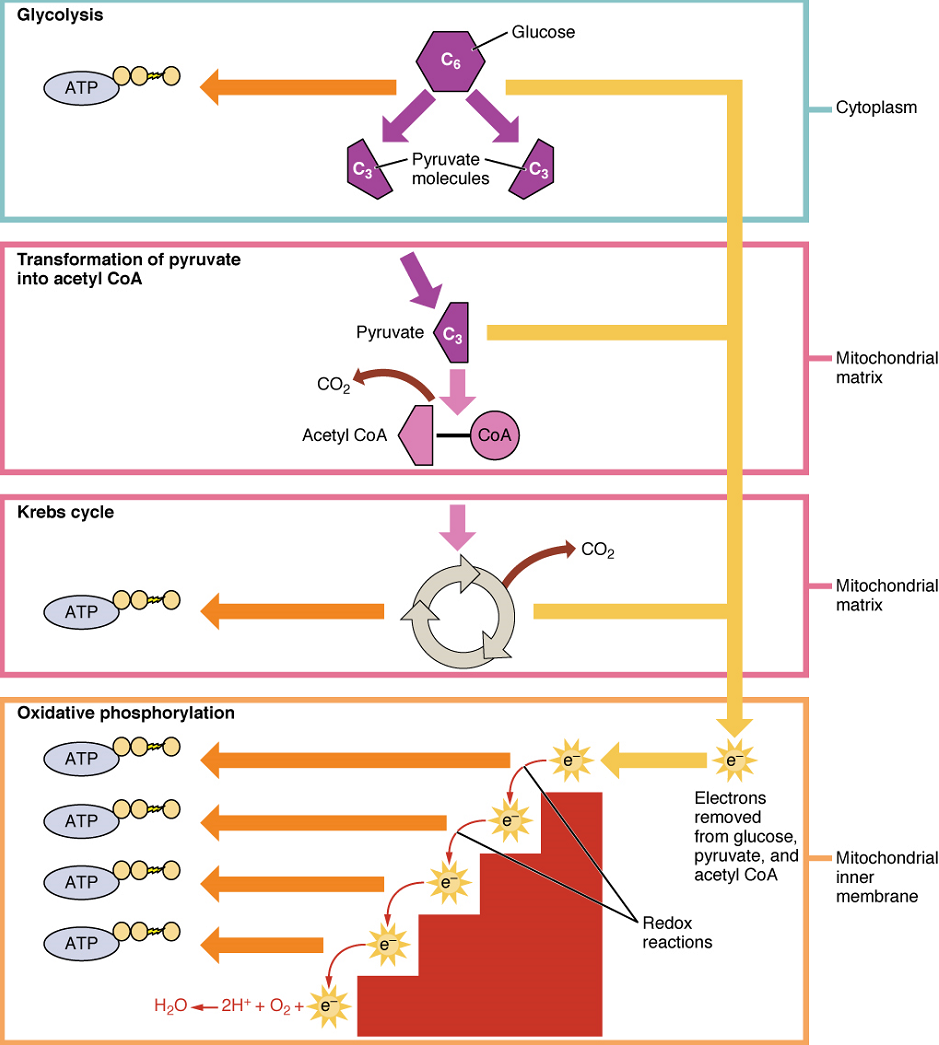
Aerobic Respiration: In the presence of oxygen, pyruvate can enter the Krebs cycle where additional energy is extracted as electrons are transferred from the pyruvate to the acceptors NAD+ and flavin adenine dinucleotide (FAD), with carbon dioxide released as a waste product (Figure 4). The NADH and FADH2 (resulting from the addition of two hydrogen atoms to FAD) pass electrons on to the electron transport chain, which uses the transferred energy to produce ATP by oxidative phosphorylation. As the last step in the electron transport chain, oxygen is the terminal electron acceptor, combining with electrons and hydrogen ions to produce water inside the mitochondria.
Krebs Cycle (Citric Acid Cycle or Tricarboxylic Acid Cycle)
The pyruvate molecules generated during glycolysis are transported across the mitochondrial membrane into the inner mitochondrial matrix, where they are metabolized by enzymes in a pathway called the Krebs cycle (Figure 5). The Krebs cycle is also commonly called the citric acid cycle or the tricarboxylic acid (TCA) cycle. During the Krebs cycle, high-energy molecules, including ATP, NADH, and FADH2, are created. NADH and FADH2 then pass electrons through the electron transport chain in the mitochondria to generate more ATP molecules.
The three-carbon pyruvate molecule generated during glycolysis moves from the cytoplasm into the mitochondrial matrix, where it is converted into a two-carbon acetyl group and bound to coenzyme A to form an acetyl coenzyme A (acetyl CoA) molecule. This reaction is an oxidative decarboxylation that releases carbon dioxide and transfers two electrons to NAD+ to form NADH. Acetyl CoA enters the Krebs cycle by combining with a four-carbon molecule, oxaloacetate, to form the six-carbon molecule citrate, or citric acid, at the same time releasing the coenzyme A molecule.
The six-carbon citrate molecule is then converted to a five-carbon molecule and then a four-carbon molecule, ending with oxaloacetate, the beginning of the cycle. Along the way, each citrate molecule will produce one ATP, one FADH2, and three NADH. The FADH2 and NADH will enter the oxidative phosphorylation system located in the inner mitochondrial membrane. In addition, the Krebs cycle supplies the starting materials to process and break down proteins and fats.
Oxidative Phosphorylation
Oxidative phosphorylation is made up of two closely tied components, the electron transport chain and chemiosmosis. The electron transport chain (ETC) uses the NADH and FADH2 produced by the Krebs cycle to generate a proton gradient. Electrons from NADH and FADH2 are transferred through protein complexes embedded in the inner mitochondrial membrane by a series of enzymatic reactions. The electron transport chain consists of a series of four enzyme complexes (Complex I – Complex IV) and two mobile electron shuttles (ubiquinone and Cytochrome c), which act as electron carriers and proton pumps used to transfer H+ ions into the space between the inner and outer mitochondrial membranes (Figure 6). The ETC couples the transfer of electrons between a donor (like NADH) and an electron acceptor (O2) with the transfer of protons (H+ ions) across the inner mitochondrial membrane. In the presence of oxygen, energy is passed, stepwise, through the electron carriers to collect gradually the energy needed to attach a phosphate to ADP and produce ATP. The role of molecular oxygen, O2, is as the terminal electron acceptor for the ETC. This means that once the electrons have passed through the entire ETC, they must be passed to another, separate molecule. These electrons, O2, and H+ ions from the matrix combine to form new water molecules. This is the basis for your need to breathe in oxygen. Without oxygen, electron flow through the ETC ceases.
The electrons released from NADH and FADH2 are passed along the chain by each of the carriers, which are reduced when they receive the electron and oxidized when passing it on to the next carrier. Each of these reactions releases a small amount of energy, which is used to pump H+ ions across the inner membrane. The accumulation of these protons in the space between the membranes creates a proton gradient with respect to the mitochondrial matrix.
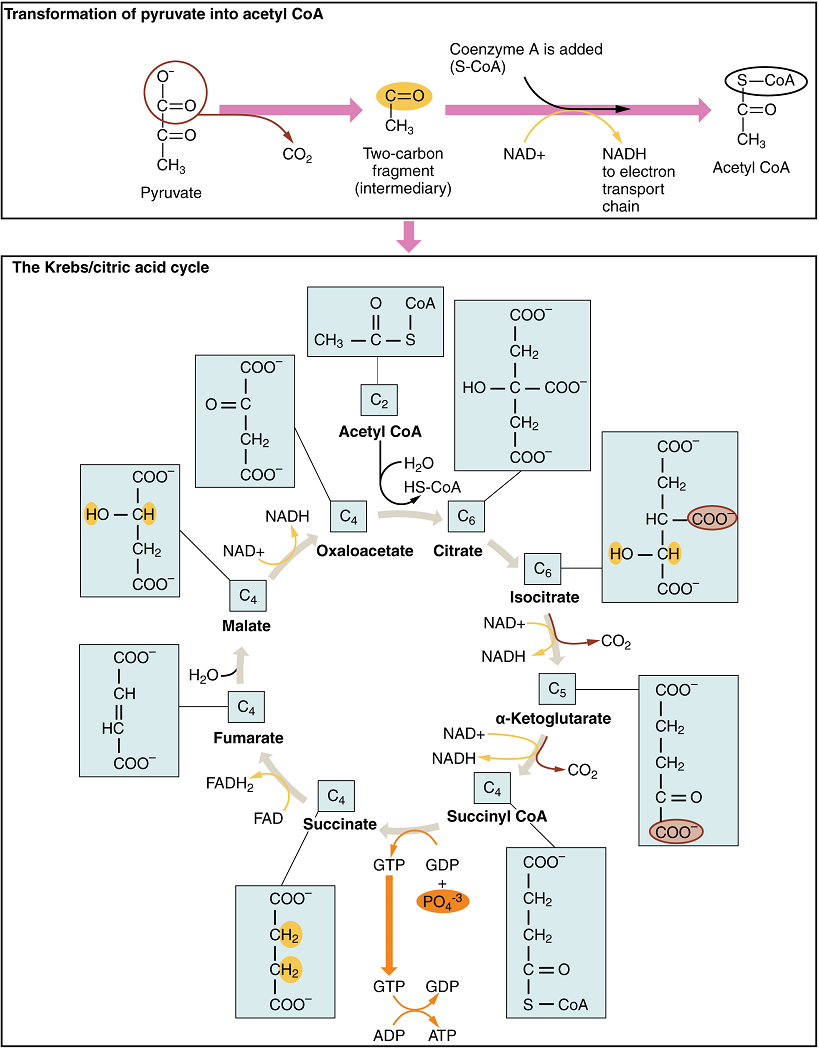
In chemiosmosis, the energy stored in the proton gradient generated by the electron transport chain is used to generate ATP. Embedded in the inner mitochondrial membrane is an amazing protein pore complex called ATP synthase. Effectively, it is a turbine that is powered by the flow of H+ ions across the inner membrane down a gradient and into the mitochondrial matrix. As the H+ ions traverse the complex, the shaft of the complex rotates. This rotation enables other portions of ATP synthase to encourage ADP and Pi to create ATP.
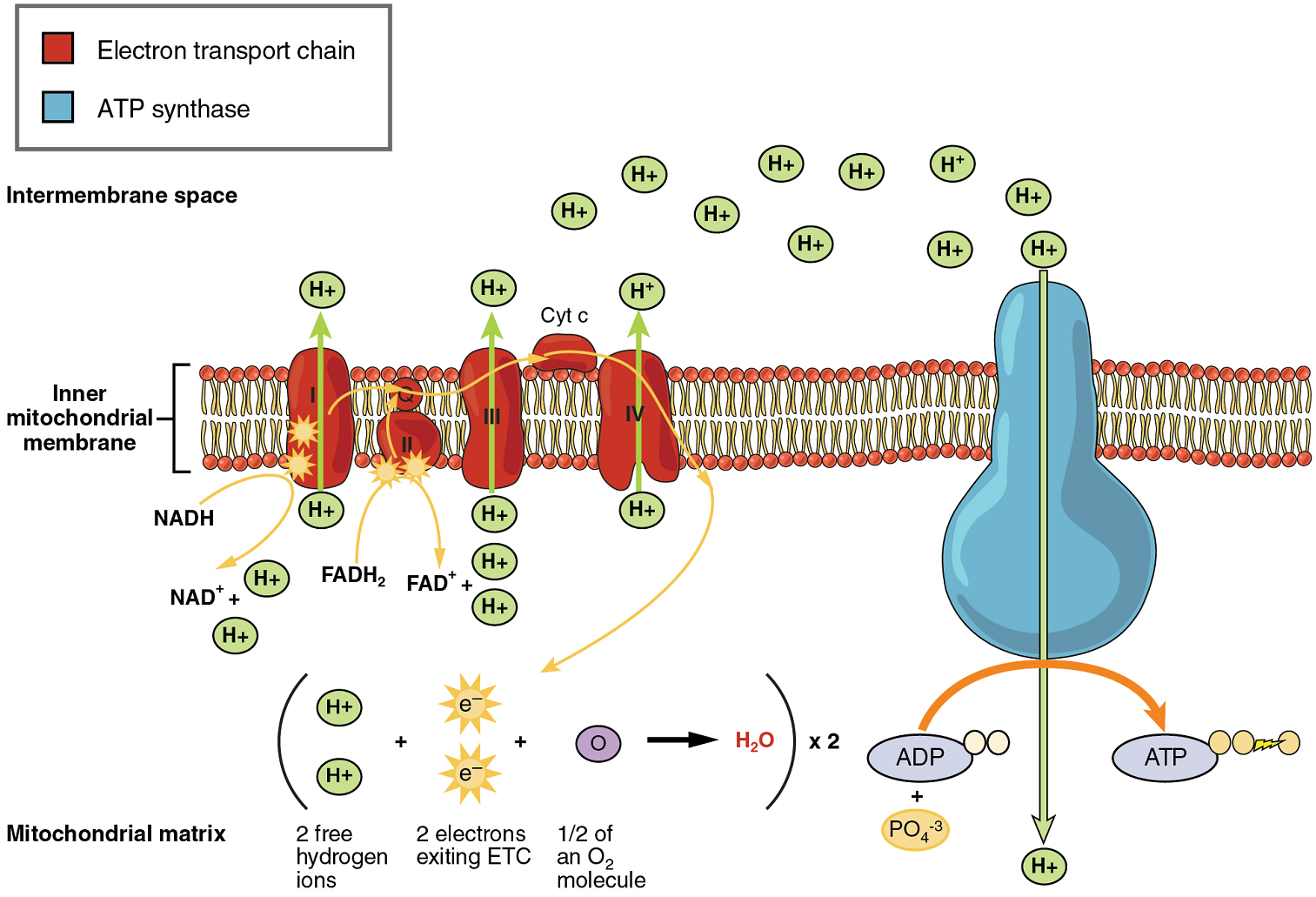
In accounting for the total number of ATP produced per glucose molecule through aerobic respiration, it is important to remember the following points:
A net of two ATP are produced through glycolysis (four produced and two consumed during the energy-consuming stage).
In all phases after glycolysis, the number of ATP, NADH, and FADH2 produced must be multiplied by two to reflect how each glucose molecule produces two pyruvate molecules.
In the ETC, about 2.5 ATP are produced for every oxidized NADH. However, only about 1.5 ATP are produced for every oxidized FADH2. The electrons from FADH2 produce less ATP, because they start at a lower point in the ETC (Complex II) compared to the electrons from NADH (Complex I) (see Figure 6)
Therefore, for every glucose molecule that enters aerobic respiration, a possible net total of 32 ATPs are produced (Figure 7). This total represents the maximum potential ATP production per glucose molecule from aerobic cellular respiration.
Gluconeogenesis
Gluconeogenesis is the synthesis of new glucose molecules from pyruvate, lactate, glycerol, or some amino acids. This process takes place primarily in the liver during periods of low glucose, that is, under conditions of fasting, starvation, and low carbohydrate diets. So, the question can be raised as to why the body would create something it has just spent a fair amount of effort to break down? Certain key organs, including the brain, can use only glucose as an energy source; therefore, it is essential that the body maintain a minimum blood glucose concentration. When the blood glucose concentration falls below that certain point, new glucose is synthesized by the liver to raise the blood concentration to normal.
As will be discussed as part of lipolysis, fats can be broken down into glycerol, which can be phosphorylated to form dihydroxyacetone phosphate or DHAP. DHAP can either enter the glycolytic pathway or be used by the liver as a substrate for gluconeogenesis.
Part 3: Lipid Metabolism
Fats (or triglycerides) within the body are ingested as food or synthesized by adipocytes or hepatocytes from carbohydrate precursors (Figure 8). Lipid metabolism entails the oxidation of fatty acids to either generate energy or synthesize new lipids from smaller constituent molecules. Lipid metabolism is associated with carbohydrate metabolism, as products of glucose (such as acetyl CoA) can be converted into lipids.
Lipolysis
To obtain energy from fat, triglycerides must first be broken down by hydrolysis into their two principal components, fatty acids and glycerol. This process, called lipolysis, takes place in the cytoplasm of adipocytes. Subsequently, the fatty acids and glycerol are released into the bloodstream, to be taken up by tissues such as the muscle, heart and liver. The resulting fatty acids are oxidized by β-oxidation into acetyl CoA, which is used by the Krebs cycle. The glycerol that is released from triglycerides after lipolysis directly enters the glycolysis pathway as DHAP. Because one triglyceride molecule yields three fatty acid molecules with as much as 16 or more carbons in each one, fat molecules yield more energy than carbohydrates and are an important source of energy for the human body. Triglycerides yield more than twice the energy per unit mass when compared to carbohydrates and proteins. Therefore, when glucose levels are low, triglycerides can be converted into acetyl CoA molecules and used to generate ATP through aerobic respiration.
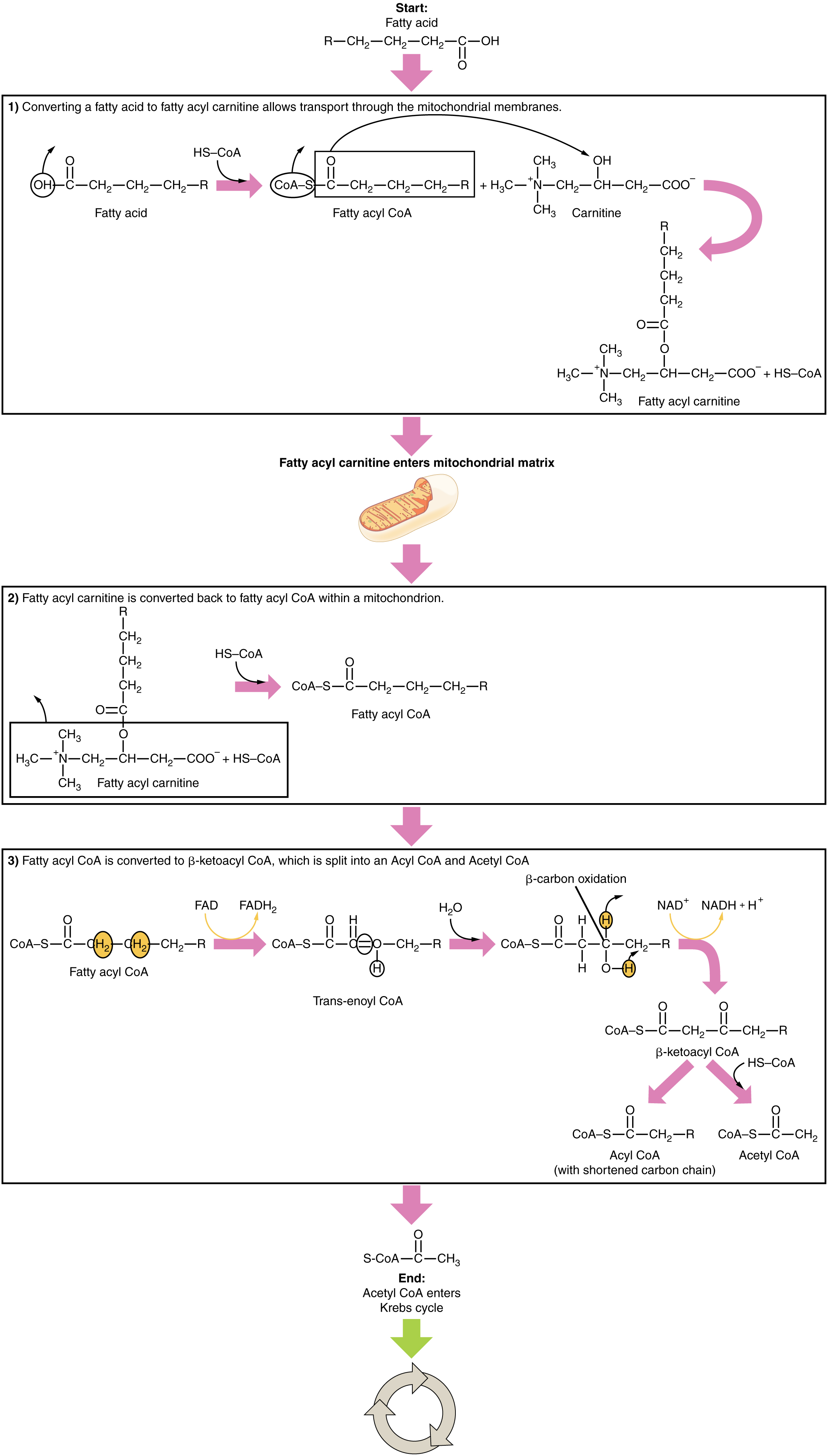
The breakdown of fatty acids begins in the cytoplasm, where fatty acids are converted into fatty acyl CoA molecules. This fatty acyl CoA is transported to the mitochondrial matrix, where it is broken down and oxidized to acetyl CoA in a process called fatty acid oxidation or beta (β)-oxidation (Figure 9). The newly formed acetyl CoA enters the Krebs cycle and is used to produce ATP in the same way as acetyl CoA derived from pyruvate.
Ketogenesis
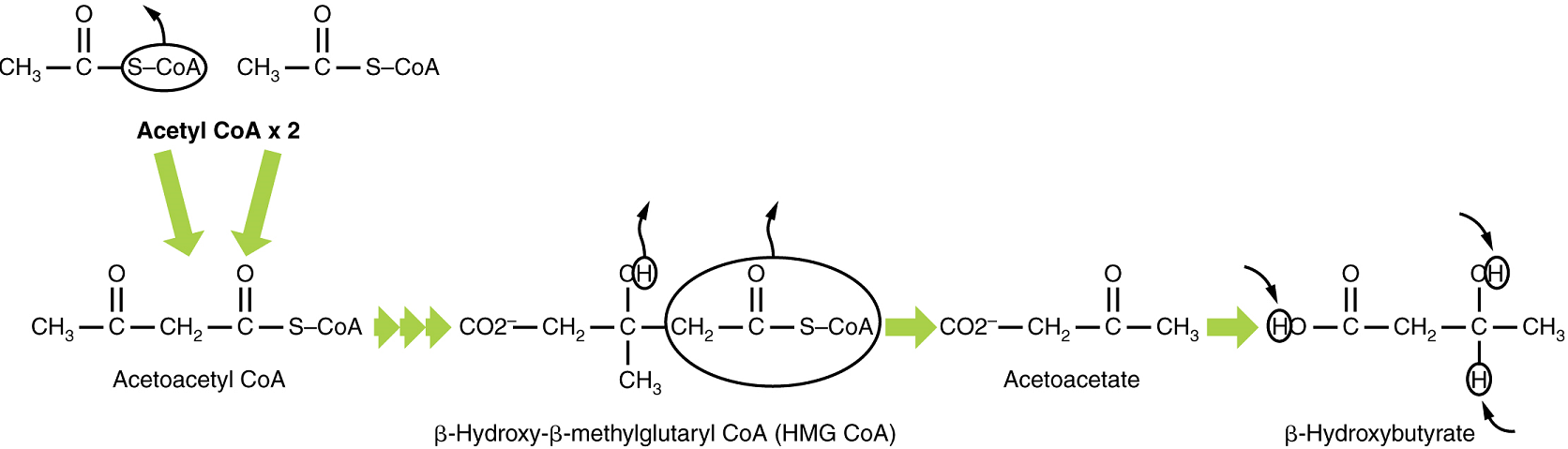
If excessive acetyl CoA is created from the oxidation of fatty acids and the Krebs cycle is overloaded and cannot handle it, the acetyl CoA, in the liver, is diverted to create ketone bodies (Figure 10).
Two of these ketone bodies (β-hydroxybutyrate and acetoacetate, and their acid forms β-hydroxybutyric acid and acetoacetatic acid) can serve as a fuel source if glucose levels are too low in the body. Ketone bodies serve as fuel in times of prolonged starvation or when patients suffer from uncontrolled diabetes and cannot utilize most of the circulating glucose. The third ketone body, acetone, is removed by exhalation. One symptom of ketogenesis is that the patient’s breath smells sweet like alcohol. This effect provides one way of telling if a diabetic is properly controlling the disease.
Ketone Body Oxidation
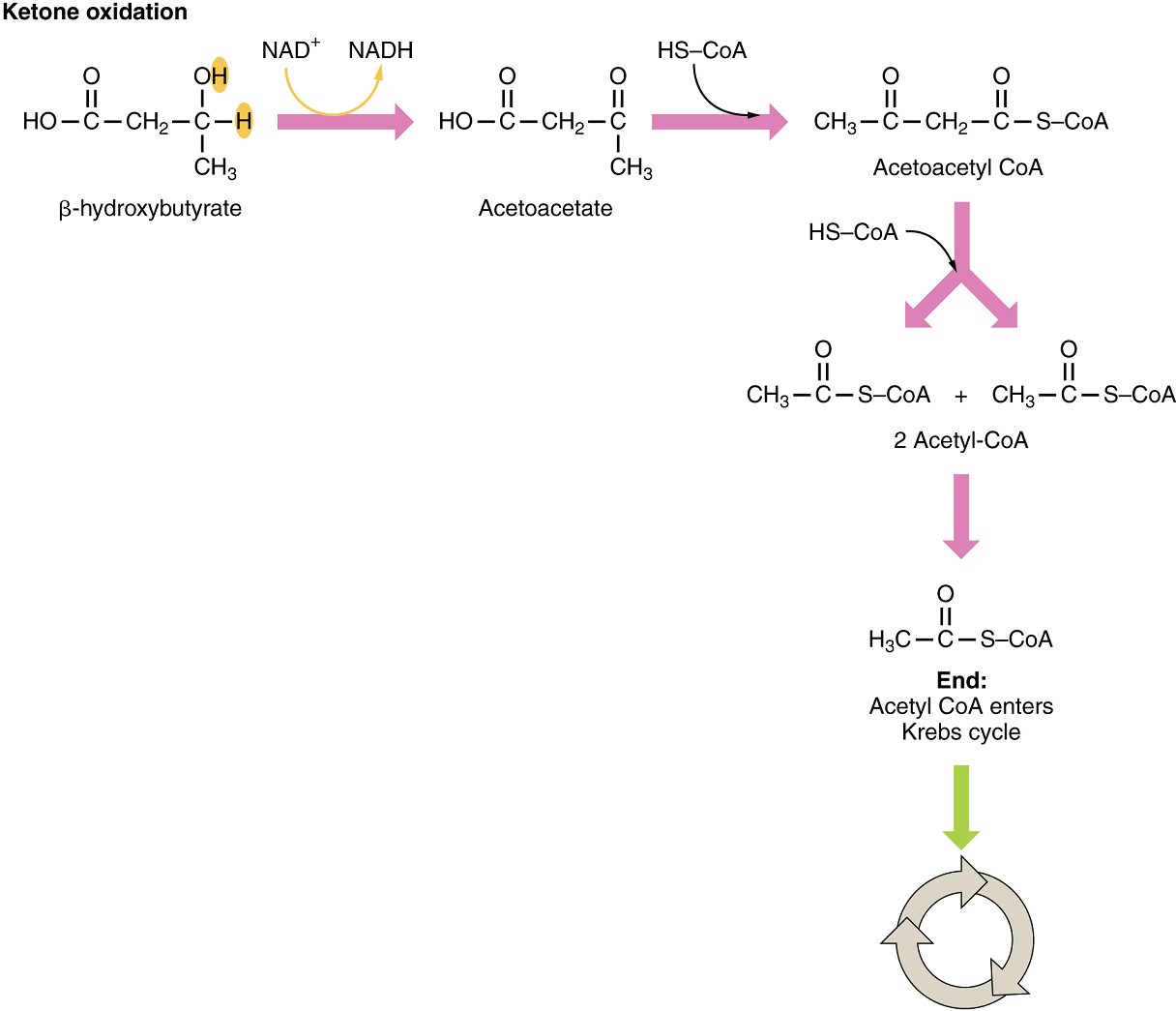
Organs that have classically been thought to be dependent solely on glucose, such as the brain, can actually use ketone bodies as an alternative energy source. This keeps the brain and other organs, such as the heart, functioning when glucose is limited. Since both β-hydroxybutyric acid and acetoacetatic acid are acids, their presence in blood, can cause acidosis (ketoacidosis), a dangerous condition in diabetics.
In these organs, ketone bodies are converted to two acetyl CoA molecules each. These acetyl CoA molecules are then processed through the Krebs cycle to generate energy (Figure 11).
Lipogenesis
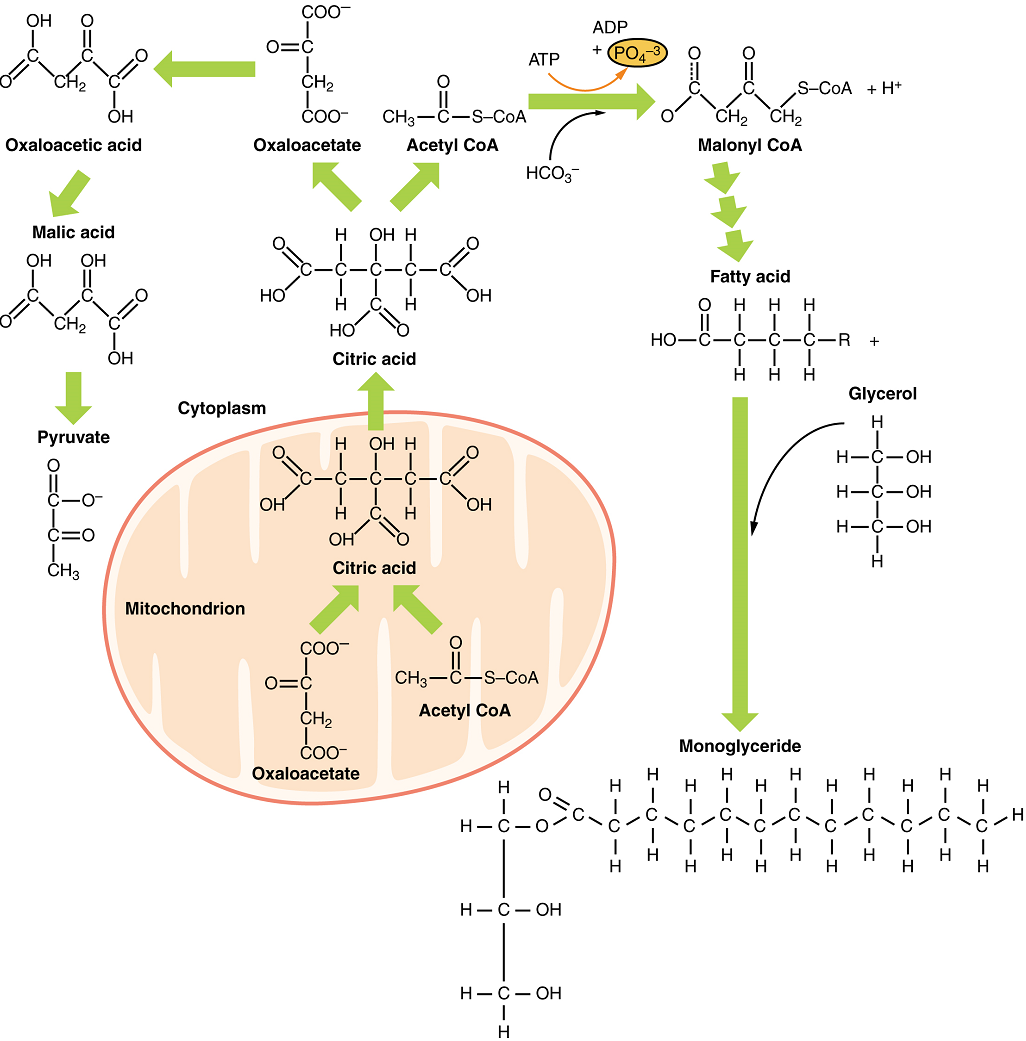
When glucose levels are plentiful, the excess acetyl CoA generated by glycolysis and pyruvate oxidation can be converted into fatty acids, triglycerides, cholesterol, steroids, and bile salts. This process, called lipogenesis, creates lipids (fat) from the acetyl CoA and takes place in the cytoplasm of adipocytes (fat cells) and hepatocytes (liver cells) (Figure 12). When you eat more glucose or carbohydrates than your body needs, acetyl CoA is turned into fat. Although there are several metabolic sources of acetyl CoA, it is most commonly derived from glycolysis. Acetyl CoA availability is significant, because it initiates lipogenesis. Lipogenesis begins with acetyl CoA and advances by the subsequent addition of two carbon atoms from another acetyl CoA; this process is repeated until fatty acids are the appropriate length. Because this is a bond-creating anabolic process, ATP is consumed. However, the creation of triglycerides and lipids is an efficient way of storing the energy available in carbohydrates. Triglycerides and lipids, high-energy molecules, are stored in adipose tissue until they are needed.
Part 4: Protein Metabolism
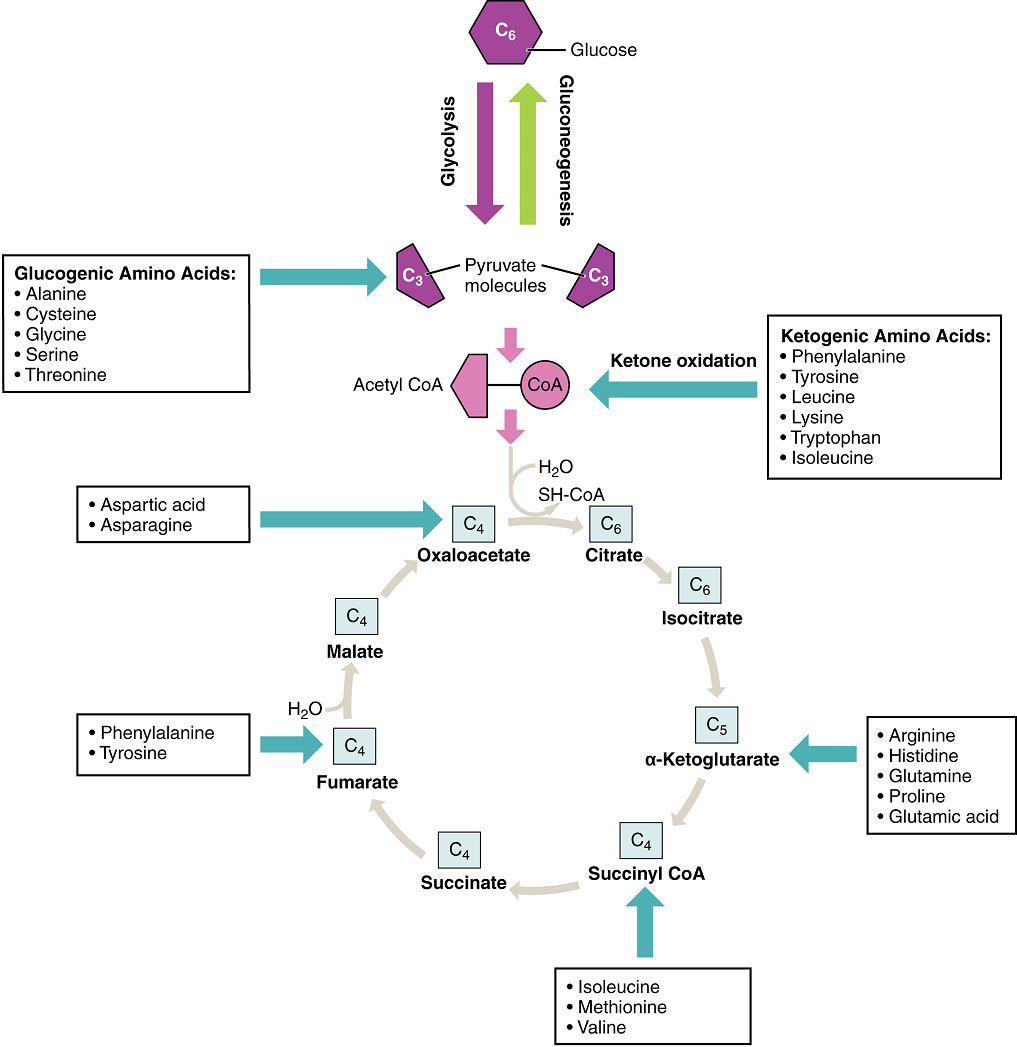
Freely available amino acids are used to create proteins. If amino acids exist in excess, the body has no capacity or mechanism for their storage; thus, they are converted into glucose or ketone bodies. Amino acid breakdown results in hydrocarbons, which are converted to glucose through gluconeogenesis, and nitrogenous waste, due to the removal of the amino group via deamination (i.e. ammonium, NH4+). However, high concentrations of nitrogen are toxic. The urea cycle, a liver process, converts ammonium into urea, facilitating the excretion of excess nitrogen from the body.
In the urea cycle, ammonium is combined with CO2, resulting in urea and water. The urea is eliminated through the kidneys in the urine.
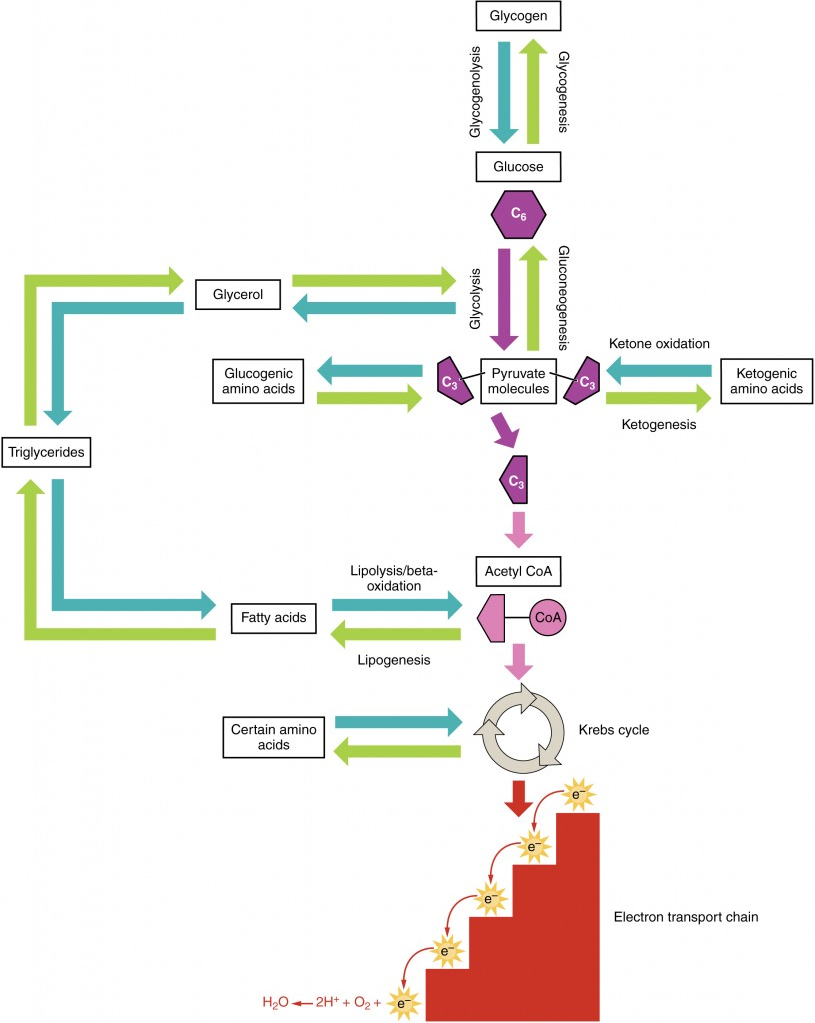
Amino acids can also be used as a source of energy, especially in times of starvation. Because the processing of amino acids results in the creation of metabolic intermediates, including pyruvate, acetyl CoA, acetoacyl CoA, oxaloacetate, and α-ketoglutarate, amino acids can serve as a source of energy production through the Krebs cycle (Figure 13). Figure 14 summarizes the pathways of catabolism and anabolism for carbohydrates, lipids, and proteins.
Part 5: Metabolic States of the Body
You eat periodically throughout the day; however, your organs, especially the brain, need a continuous supply of glucose. How does the body meet this constant demand for energy? Your body processes the food you eat both to use immediately and, importantly, to store as energy for later demands. If there were no method in place to store excess energy, you would need to eat constantly in order to meet energy demands. Distinct mechanisms are in place to facilitate energy storage, and to make stored energy available during times of fasting and starvation.
The Absorptive State
The absorptive state, or the fed state, occurs after a meal when your body is digesting the food and absorbing the nutrients (anabolism exceeds catabolism) (Figure 15). Digestion begins the moment you put food into your mouth, as the food is broken down into its constituent parts to be absorbed through the intestine. The digestion of carbohydrates begins in the mouth, whereas the digestion of proteins and fats begins in the stomach and small intestine. The constituent parts of these carbohydrates, fats, and proteins are transported across the intestinal wall and enter the bloodstream (sugars and amino acids) or the lymphatic system (fats). From the intestines, these systems transport them to the liver, adipose tissue, or muscle cells that will process and use, or store, the energy.
Depending on the amounts and types of nutrients ingested, the absorptive state can linger for up to 4 hours. The ingestion of food and the rise of glucose concentrations in the bloodstream stimulate pancreatic beta cells to release insulin into the bloodstream, where it initiates the absorption of blood glucose by liver hepatocytes, and by adipose and muscle cells. Insulin also stimulates glycogenesis, the storage of glucose as glycogen, in the liver and muscle cells where it can be used for later energy needs of the body. Insulin also promotes the synthesis of protein in muscle. As you will see, muscle protein can be catabolized and used as fuel in times of starvation.
If energy is exerted shortly after eating, the dietary fats and sugars that were just ingested will be processed and used immediately for energy. If not, the excess glucose is stored as glycogen in the liver and muscle cells, or as fat in adipose tissue; excess dietary fat is also stored as triglycerides in adipose tissues.
The Postabsorptive State
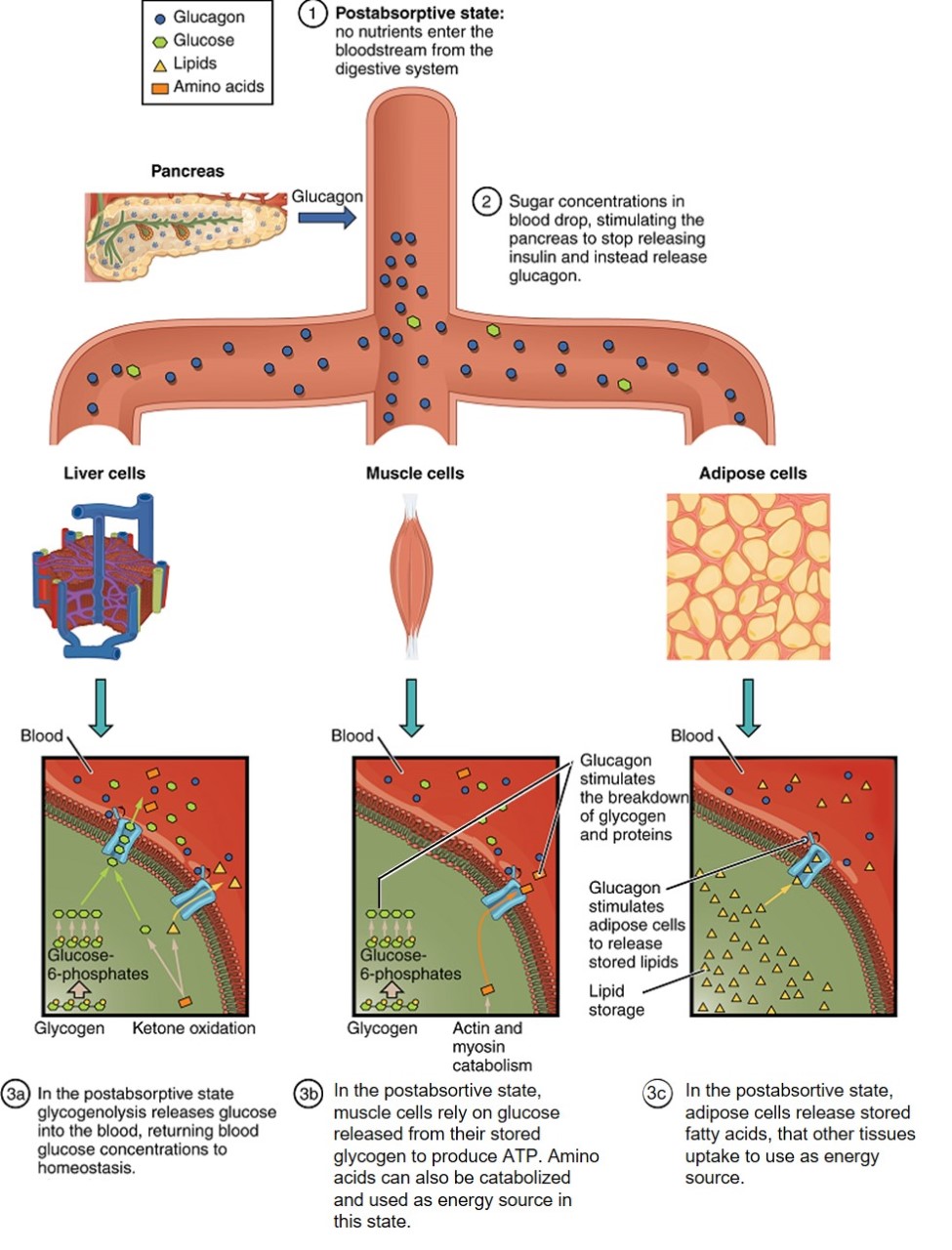
The postabsorptive state, or the fasting state, occurs when the food has been digested, absorbed, and stored (Figure 16). You commonly fast overnight, but skipping meals during the day puts your body in the postabsorptive state as well. During this state, the body must rely initially on stored glycogen. Glucose levels in the blood begin to drop as it is absorbed and used by the cells. In response to the decrease in glucose, insulin levels also drop. Glycogen and triglyceride storage slows. However, due to the demands of the tissues and organs, blood glucose levels must be maintained in the normal range of 80–120 mg/dL. In response to a drop in blood glucose concentration, the hormone glucagon is released from the alpha cells of the pancreas. Glucagon acts upon the liver cells, where it inhibits glycogenesis and stimulates glycogenolysis, the breakdown of stored glycogen back into glucose. The glucose is released from the liver to be used by the peripheral tissues and the brain. As a result, blood glucose levels begin to rise. The stored glycogen in a well-fed human typically is sufficient to meet the energy needs of the body for several hours. Gluconeogenesis, the production of glucose from non-carbohydrates, will also begin in the liver to replace the glucose that has been used by the peripheral tissues.
Starvation
When the body is deprived of nourishment for an extended period of time, it goes into “survival mode.” The first priority for survival is to provide enough glucose or fuel for the brain. The second priority is the conservation of amino acids for proteins. Therefore, when glucose is no longer available, the use of ketone bodies as an energy source helps to decrease the demand for glucose, thus minimizing gluconeogenesis in order to maintain body proteins.
Because glucose levels are very low during starvation, glycolysis will shut off in cells that can use alternative fuels. For example, muscles will switch from using glucose to fatty acids as fuel. As previously explained, fatty acids can be converted into acetyl CoA and processed through the Krebs cycle to make ATP. Pyruvate, lactate, and alanine from muscle cells are not converted into acetyl CoA and used in the Krebs cycle, but are exported to the liver to be used in the synthesis of glucose. As starvation continues, and more glucose is needed, glycerol from fatty acids can be liberated and used as a source for gluconeogenesis.
After several days of starvation, ketone bodies become the major source of fuel for the heart and other organs. As starvation continues, fatty acids and triglyceride stores are oxidized to create these molecules. This prevents the continued breakdown of proteins that serve as carbon sources for gluconeogenesis, helping to maintain the proper functioning of the body’s muscles. Once these lipid stores are fully depleted, proteins from muscles are released and broken down for glucose synthesis. This leads to muscle wasting, as the body is forced to cannibalize the tissue for survival. Overall survival is dependent on the amount of fat and protein stored in the body.
Part 1 Overview of Metabolic Reactions
Part 2 Carbohydrate Metabolism
Part 3 Lipid Metabolism
Part 4 Protein Metabolism
Part 5 Metabolic States of the Body
Chemical reaction that breaks down more complex organic molecules.
Nucleotide containing ribose and an adenine base that is essential in energy transfer.
Reactions that build smaller molecules into larger molecules.
Class of nonpolar organic compounds built from hydrocarbons and distinguished by the fact that they are not soluble in water.
Class of organic compounds that are composed of many amino acids linked together by peptide bonds.
Class or organic molecules consisting of a chain of nucleotides (including DNA and RNA).
Simple carbohydrate consisting of a single carbon-oxygen ring.
Tissue or organ that secretes hormones into the blood and lymph without ducts such that they may be transported to organs distant from the site of secretion.
Monosaccharide commonly used as energy in the body (substrate for glycolysis).
Polysaccharide used as a storage form of glucose in the liver.
Lipid compound composed of a glycerol molecule bonded with three fatty acid chains.
Fatty acid oxidation to form acetyl CoA.
A "building block" of complex organic molecules. Complex organic molecules are usually composed of a number of similar monomers (e.g. amino acids in protein).
Building block of proteins; characterized by an amino and carboxyl functional groups and a variable side-chain.
Class of organic compounds built from sugars, molecules containing carbon, hydrogen, and oxygen in a 1-2-1 ratio.
Digestive enzyme in saliva that acts on starch (a polysaccharide carbohydrate).
Production of ATP from glucose oxidation via glycolysis, the Krebs cycle, and oxidative phosphorylation.
Series of metabolic reactions that breaks down glucose into pyruvate and produces ATP.
Largest gland in the body, overlying the stomach, whose functions include bile production and a number of metabolic and regulatory functions.
Liver cells.
Pancreatic hormone that enhances the cellular uptake and utilization of glucose, thereby decreasing blood glucose levels.
(Also, pyruvate) Three-carbon end product of glycolysis and starting material that is converted into acetyl CoA that enters the citric acid cycle.
Electron acceptor that carries electrons from glycolysis and citric acid cycle to the electron transport chain (or supplies electrons during lactic acid fermentation). NADH is the higher energy form, carrying two electrons.
Process by which a high-energy phosphate is added to a molecule. That molecule is said to be phosphorylated.
Lower energy form of ATP, containing two phosphate groups after the third phosphate group phosphorylated another molecule and transferring energy to it.
Phosphorylation of ATP during glycolysis and the Kreb's Cycle.
Anaerobic pathway by which smaller amounts of ATP can be produced when oxygen is limited or absent. Lactic acid is a byproduct.
Process of glucose synthesis from pyruvate or other molecules.
One of the cellular organelles bound by a double lipid bilayer that function primarily in the production of cellular energy (ATP).
Electron acceptor similar to NAD+, carrying electrons from the Kreb's cycle to the electron transport chain. FADH2 is the reduced, higher energy form with additional electrons.
ATP production pathway in which electrons are passed through a series of oxidation-reduction reactions that forms water and produces a proton gradient.
Process that converts high-energy NADH and FADH2 into ATP.
Space within the inner mitochondrial membrane, containing a viscous fluid where the Krebs cycle takes place.
(Also, citric acid cycle or tricarboxylic (TCA) cycle) converts pyruvate into CO2 and high-energy FADH2, NADH, and ATP molecules.
Starting molecule of the Krebs cycle and end product of β-oxidation.
The diffusion of ions across a semipermeable membrane, down their electrochemical gradient.
Innermost of the double membrane, this is highly folded into cristae, providing a large surface area for chemical reactions such as the electron transport chain.
Protein pore complex on the inner membrane of the mitochondrion that creates ATP.
(Also, lactic acid) a three-carbon acid formed as a product of lactic acid fermentation, and used as a substrate in gluconeogenesis.
Molecule that forms the 'backbone' of many lipids, including triglycerides.
Breakdown of triglycerides into glycerol and fatty acids.
Reactant in an enzymatic reaction.
Chemical reaction in which water is used to break one or more chemical bonds.
Consisting of a carboxyl group (COOH) and an unbranched hydrocarbon chain. A non-polar component of all lipids.
Alternative source of energy when glucose is limited, created when too much acetyl CoA is created during fatty acid oxidation. Consist of acetone, acetoacetic acid and β-hydroxybutyric acid.
Formation of ketone bodies during periods of glucose starvation.
Complication of diabetes wherein the blood becomes acidic because of increased levels of ketones.
A type of lipid containing four rings and a fatty acid tail. Examples include testosterone and cholesterol.
Salts that are released from the liver in response to lipid ingestion and surround the insoluble triglycerides to aid in their conversion to monoglycerides and free fatty acids.
Synthesis of lipids that occurs in the liver or adipose tissues.
Process that converts potentially toxic nitrogen waste into urea that can be eliminated through the kidneys.
Nitrogenous compound formed in the liver from ammonium (which is toxic), a product of protein metabolism.
Metabolic process that builds glycogen molecules from glucose.
Pancreatic hormone that stimulates the catabolism of glycogen to glucose, thereby increasing blood glucose levels.
An amino acid.

