8.5 Polyprotic Acids
Learning Objectives
By the end of this section, you will be able to:
- Extend previously introduced equilibrium concepts to acids and bases that may donate or accept more than one proton
Acids are classified by the number of protons per molecule that they can give up in a reaction. Acids such as HCl, HNO3, and HCN that contain one ionizable hydrogen atom in each molecule are called monoprotic acids. Their reactions with water are:
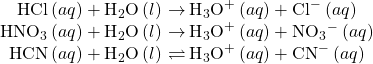
Even though it contains four hydrogen atoms, acetic acid, CH3CO2H, is also monoprotic because only the hydrogen atom from the carboxyl group (COOH) reacts with bases:
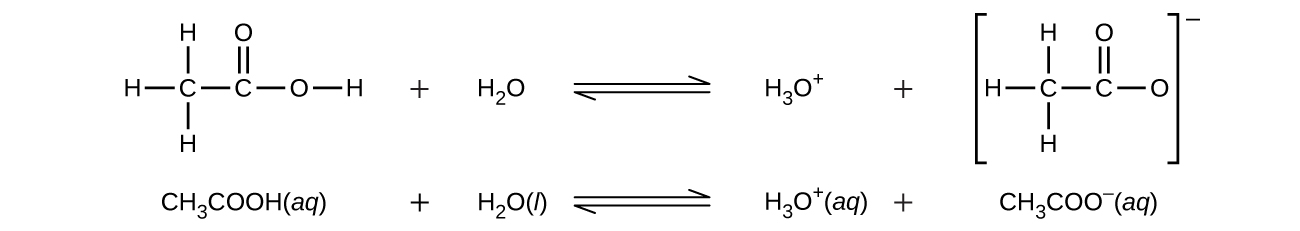
Similarly, monoprotic bases are bases that will accept a single proton. Diprotic acids contain two ionizable hydrogen atoms per molecule; ionization of such acids occurs in two steps. The first ionization always takes place to a greater extent than the second ionization. For example, sulfuric acid, a strong acid, ionizes as follows:
This stepwise ionization process occurs for all polyprotic acids. Carbonic acid, H2CO3, is an example of a weak diprotic acid. The first ionization of carbonic acid yields hydronium ions and bicarbonate ions in small amounts:
![Rendered by QuickLaTeX.com \begin{array}{l}\text{First ionization:}\\ {\text{H}}_{2}{\text{CO}}_{3}\left(aq\right)+{\text{H}}_{2}\text{O}\left(l\right)$\rightleftharpoons${\text{H}}_{3}{\text{O}}^{\text{+}}\left(aq\right)+{\text{HCO}}_{3}{^-}^{\text{}}\left(aq\right)\phantom{\rule{5em}{0ex}}{K}_{{\text{H}}_{2}{\text{CO}}_{3}}=\phantom{\rule{0.2em}{0ex}}\frac{\left[{\text{H}}_{3}{\text{O}}^{\text{+}}\right]\left[{\text{HCO}}_{3}{^-}^{\text{}}\right]}{\left[{\text{H}}_{2}{\text{CO}}_{3}\right]}\phantom{\rule{0.2em}{0ex}}=4.3\phantom{\rule{0.2em}{0ex}}\times\phantom{\rule{0.2em}{0ex}}{10}^{-7}\end{array}](https://pressbooks.bccampus.ca/inorganicchemistrychem250/wp-content/ql-cache/quicklatex.com-45a3a92f548a871f286cceac2873fb64_l3.png)
The bicarbonate ion can also act as an acid. It ionizes and forms hydronium ions and carbonate ions in even smaller quantities:
![Rendered by QuickLaTeX.com \begin{array}{l}\text{Second ionization:}\\ {\text{HCO}}_{3}{^-}^{\text{}}\left(aq\right)+{\text{H}}_{2}\text{O}\left(l\right)$\rightleftharpoons${\text{H}}_{3}{\text{O}}^{\text{+}}\left(aq\right)+{\text{CO}}_{3}{^{2-}}^{}\left(aq\right)\phantom{\rule{5em}{0ex}}{K}_{{\text{HCO}}_{3}{^-}^{\text{}}}=\phantom{\rule{0.2em}{0ex}}\frac{\left[{\text{H}}_{3}{\text{O}}^{\text{+}}\right]\left[{\text{CO}}_{3}{^{2-}}^{}\right]}{\left[{\text{HCO}}_{3}{^-}^{\text{}}\right]}\phantom{\rule{0.2em}{0ex}}=4.7\phantom{\rule{0.2em}{0ex}}\times\phantom{\rule{0.2em}{0ex}}{10}^{-11}\end{array}](https://pressbooks.bccampus.ca/inorganicchemistrychem250/wp-content/ql-cache/quicklatex.com-a9f7f4c9a4784491251c1eb6e22550c6_l3.png)
![]() is larger than
is larger than ![]() by a factor of 104, so H2CO3 is the dominant producer of hydronium ion in the solution. This means that little of the
by a factor of 104, so H2CO3 is the dominant producer of hydronium ion in the solution. This means that little of the ![]() formed by the ionization of H2CO3 ionizes to give hydronium ions (and carbonate ions), and the concentrations of H3O+ and
formed by the ionization of H2CO3 ionizes to give hydronium ions (and carbonate ions), and the concentrations of H3O+ and ![]() are practically equal in a pure aqueous solution of H2CO3.
are practically equal in a pure aqueous solution of H2CO3.
If the first ionization constant of a weak diprotic acid is larger than the second by a factor of at least 20, it is appropriate to treat the first ionization separately and calculate concentrations resulting from it before calculating concentrations of species resulting from subsequent ionization. This approach is demonstrated in the following example exercise.
Activity 8.5.1 – Ionization of a Diprotic Acid
“Carbonated water” contains a palatable amount of dissolved carbon dioxide. The solution is acidic because CO2 reacts with water to form carbonic acid, H2CO3. What are ![]()
![]() and
and ![]() in a saturated solution of CO2 with an initial [H2CO3] = 0.033 M?
in a saturated solution of CO2 with an initial [H2CO3] = 0.033 M?
Solution
As indicated by the ionization constants, H2CO3 is a much stronger acid than ![]() so the stepwise ionization reactions may be treated separately. The first ionization reaction is:
so the stepwise ionization reactions may be treated separately. The first ionization reaction is:
Using provided information, an ICE table for this first step is prepared:
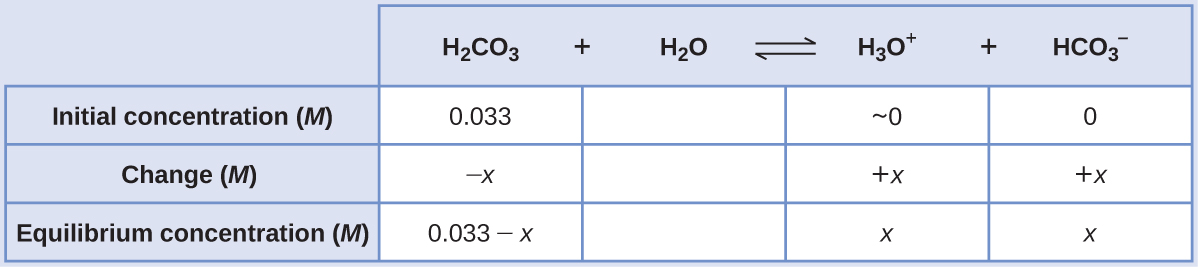
Substituting the equilibrium concentrations into the equilibrium equation gives:
Assuming x << 0.033 and solving the simplified equation yields:
The ICE table defined x as equal to the bicarbonate ion molarity and the hydronium ion molarity:
Using the bicarbonate ion concentration computed above, the second ionization is subjected to a similar equilibrium calculation:
To summarize: at equilibrium [H2CO3] = 0.033 M; ![]() = 1.2
= 1.2 ![]() 10−4;
10−4; ![]()
![]()
Check Your Learning
The concentration of H2S in a saturated aqueous solution at room temperature is approximately 0.1 M. Calculate ![]() [HS−], and [S2−] in the solution:
[HS−], and [S2−] in the solution:
Answer
[H2S] = 0.1 M; ![]() = [HS−] = 0.000094 M; [S2−] = 1
= [HS−] = 0.000094 M; [S2−] = 1 ![]() 10−19M
10−19M
A triprotic acid is an acid that has three ionizable H atoms. Phosphoric acid is one example:
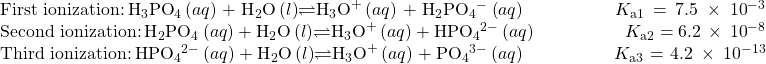
As for the diprotic acid examples, each successive ionization reaction is less extensive than the former, reflected in decreasing values for the stepwise acid ionization constants. This is a general characteristic of polyprotic acids and successive ionization constants often differ by a factor of about 105 to 106.
This set of three dissociation reactions may appear to make calculations of equilibrium concentrations in a solution of H3PO4 complicated. However, because the successive ionization constants differ by a factor of 105 to 106, large differences exist in the small changes in concentration accompanying the ionization reactions. This allows the use of math-simplifying assumptions and processes, as demonstrated in the examples above.
Polyprotic bases are capable of accepting more than one hydrogen ion. The carbonate ion is an example of a diprotic base, because it can accept two protons, as shown below. Similar to the case for polyprotic acids, note the ionization constants decrease with ionization step. Likewise, equilibrium calculations involving polyprotic bases follow the same approaches as those for polyprotic acids.
Key Concepts and Summary
An acid that contains more than one ionizable proton is a polyprotic acid. These acids undergo stepwise ionization reactions involving the transfer of single protons. The ionization constants for polyprotic acids decrease with each subsequent step; these decreases typically are large enough to permit simple equilibrium calculations that treat each step separately.
End of Chapter Exercises
(1) Which of the following concentrations would be practically equal in a calculation of the equilibrium concentrations in a 0.134-M solution of H2CO3, a diprotic acid: ![]() [OH−], [H2CO3],
[OH−], [H2CO3], ![]()
![]() No calculations are needed to answer this question.
No calculations are needed to answer this question.
Solution
[H3O+] and ![]() are practically equal
are practically equal
(2) Calculate the concentration of each species present in a 0.050-M solution of H2S.
(3) Calculate the concentration of each species present in a 0.010-M solution of phthalic acid, C6H4(CO2H)2:
![]()
Solution
[C6H4(CO2H)2]= 7.2 ![]() 10−3M, [C6H4(CO2H)(CO2)−] = [H3O+]= 2.8
10−3M, [C6H4(CO2H)(CO2)−] = [H3O+]= 2.8 ![]() 10−3M,
10−3M, ![]() 3.9
3.9 ![]() 10−6M, [OH−]= 3.6
10−6M, [OH−]= 3.6 ![]() 10−12M
10−12M
(4) Salicylic acid, HOC6H4CO2H, and its derivatives have been used as pain relievers for a long time. Salicylic acid occurs in small amounts in the leaves, bark, and roots of some vegetation (most notably historically in the bark of the willow tree). Extracts of these plants have been used as medications for centuries. The acid was first isolated in the laboratory in 1838.
(4a) Both functional groups of salicylic acid ionize in water, with Ka = 1.0 ![]() 10−3 for the—CO2H group and 4.2
10−3 for the—CO2H group and 4.2 ![]() 10−13 for the −OH group. What is the pH of a saturated solution of the acid (solubility = 1.8 g/L).
10−13 for the −OH group. What is the pH of a saturated solution of the acid (solubility = 1.8 g/L).
(4b) Aspirin was discovered as a result of efforts to produce a derivative of salicylic acid that would not be irritating to the stomach lining. Aspirin is acetylsalicylic acid, CH3CO2C6H4CO2H. The −CO2H functional group is still present, but its acidity is reduced, Ka = 3.0 ![]() 10−4. What is the pH of a solution of aspirin with the same concentration as a saturated solution of salicylic acid (See Part a).
10−4. What is the pH of a solution of aspirin with the same concentration as a saturated solution of salicylic acid (See Part a).
(5) The ion HTe− is an amphiprotic species; it can act as either an acid or a base.
(5a) What is Ka for the acid reaction of HTe− with H2O?
(5b) What is Kb for the reaction in which HTe− functions as a base in water?
(5c) Demonstrate whether or not the second ionization of H2Te can be neglected in the calculation of [HTe−] in a 0.10 M solution of H2Te.
Solution
(a) ![]()
(b) ![]()
(c) ![]()
Solving for x gives 1.5 ![]() 10−11M. Therefore, compared with 0.014 M, this value is negligible (1.1
10−11M. Therefore, compared with 0.014 M, this value is negligible (1.1 ![]() 10−7%).
10−7%).
Glossary
- diprotic acid
- acid containing two ionizable hydrogen atoms per molecule
- diprotic base
- base capable of accepting two protons
- monoprotic acid
- acid containing one ionizable hydrogen atom per molecule
- stepwise ionization
- process in which a polyprotic acid is ionized by losing protons sequentially
- triprotic acid
- acid that contains three ionizable hydrogen atoms per molecule
acid containing two ionizable hydrogen atoms per molecule
base capable of accepting two protons
acid containing one ionizable hydrogen atom per molecule
process in which a polyprotic acid is ionized by losing protons sequentially
acid that contains three ionizable hydrogen atoms per molecule

