Chapter 2 Electric Charge and Electric Field
2.6 Conductors and Insulators
Summary
- Define conductor and insulator, explain the difference, and give examples of each.
- Describe three methods for charging an object.
- Explain what happens to an electric force as you move farther from the source.
- Define polarization.

Some substances, such as metals and salty water, allow charges to move through them with relative ease. Some of the electrons in metals and similar conductors are not bound to individual atoms or sites in the material. These free electrons can move through the material much as air moves through loose sand. Any substance that has free electrons and allows charge to move relatively freely through it is called a conductor. The moving electrons may collide with fixed atoms and molecules, losing some energy, but they can move in a conductor. Superconductors allow the movement of charge without any loss of energy. Salty water and other similar conducting materials contain free ions that can move through them. An ion is an atom or molecule having a positive or negative (nonzero) total charge. In other words, the total number of electrons is not equal to the total number of protons.
Other substances, such as glass, do not allow charges to move through them. These are called insulators. Electrons and ions in insulators are bound in the structure and cannot move easily—as much as 10 23 times more slowly than in conductors. Pure water and dry table salt are insulators, for example, whereas molten salt and salty water are conductors.
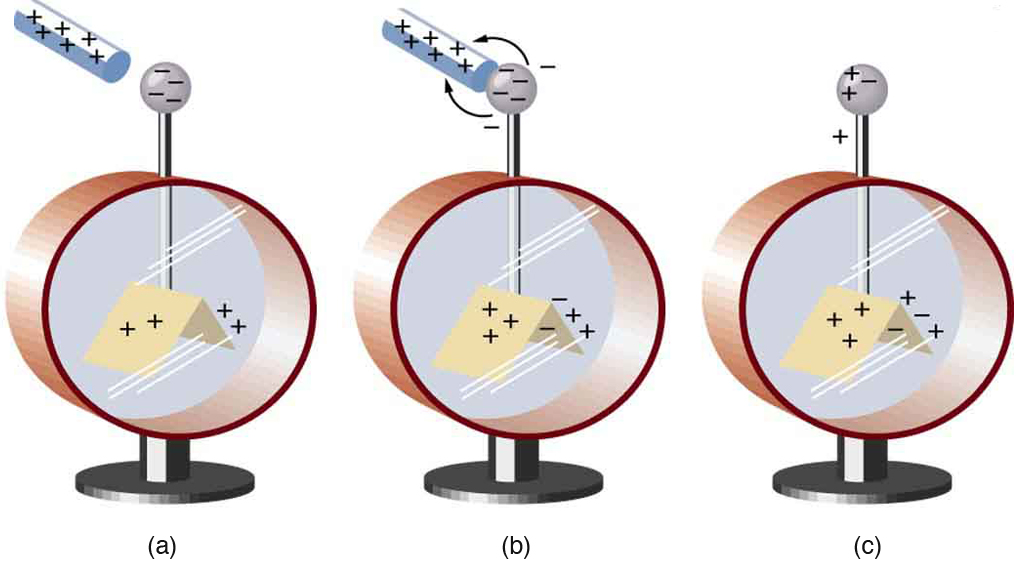
Charging by Contact
Figure 2 shows an electroscope being charged by touching it with a positively charged glass rod. Because the glass rod is an insulator, it must actually touch the electroscope to transfer charge to or from it. (Note that the extra positive charges reside on the surface of the glass rod as a result of rubbing it with silk before starting the experiment.) Since only electrons move in metals, we see that they are attracted to the top of the electroscope. There, some are transferred to the positive rod by touch, leaving the electroscope with a net positive charge.
Electrostatic repulsion in the leaves of the charged electroscope separates them. The electrostatic force has a horizontal component that results in the leaves moving apart as well as a vertical component that is balanced by the gravitational force. Similarly, the electroscope can be negatively charged by contact with a negatively charged object.
Charging by Induction
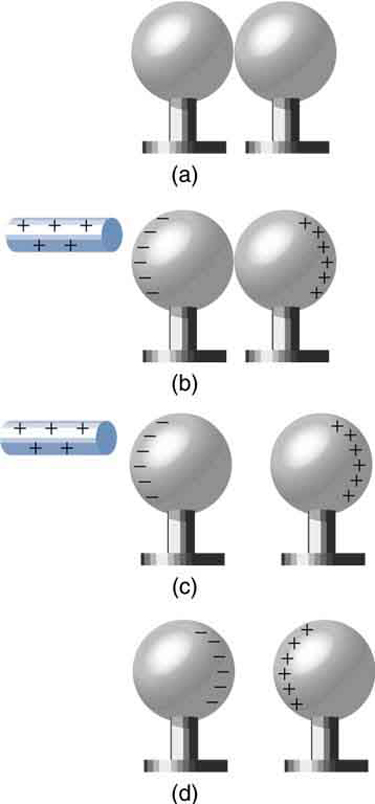
It is not necessary to transfer excess charge directly to an object in order to charge it. Figure 3 shows a method of induction wherein a charge is created in a nearby object, without direct contact. Here we see two neutral metal spheres in contact with one another but insulated from the rest of the world. A positively charged rod is brought near one of them, attracting negative charge to that side, leaving the other sphere positively charged.
This is an example of induced polarization of neutral objects. Polarization is the separation of charges in an object that remains neutral. If the spheres are now separated (before the rod is pulled away), each sphere will have a net charge. Note that the object closest to the charged rod receives an opposite charge when charged by induction. Note also that no charge is removed from the charged rod, so that this process can be repeated without depleting the supply of excess charge.
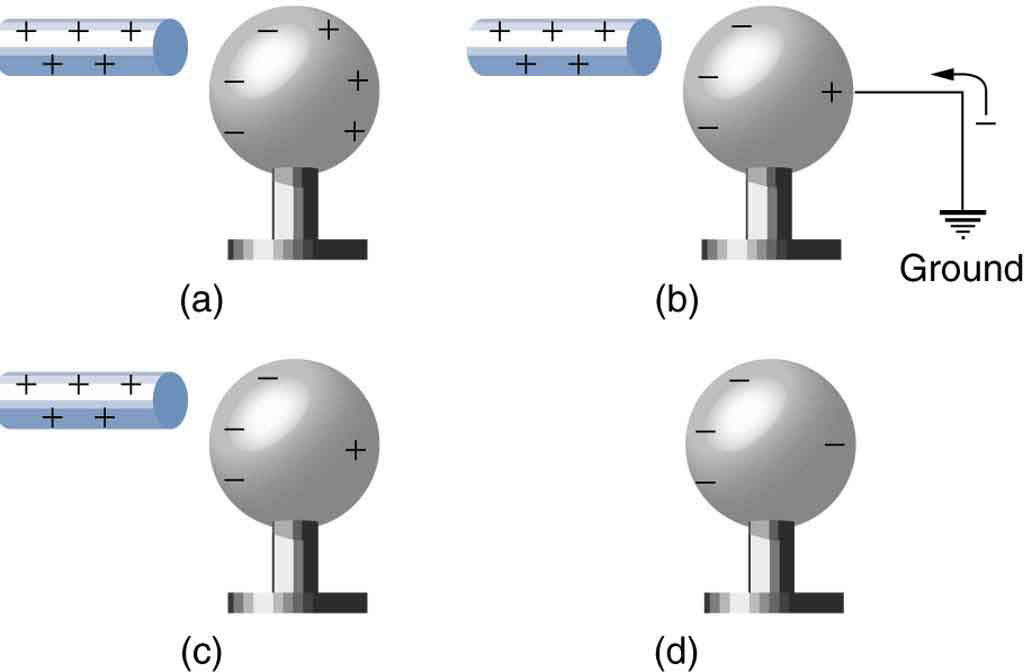
Another method of charging by induction is shown in Figure 4. The neutral metal sphere is polarized when a charged rod is brought near it. The sphere is then grounded, meaning that a conducting wire is run from the sphere to the ground. Since the earth is large and most ground is a good conductor, it can supply or accept excess charge easily. In this case, electrons are attracted to the sphere through a wire called the ground wire, because it supplies a conducting path to the ground. The ground connection is broken before the charged rod is removed, leaving the sphere with an excess charge opposite to that of the rod. Again, an opposite charge is achieved when charging by induction and the charged rod loses none of its excess charge.
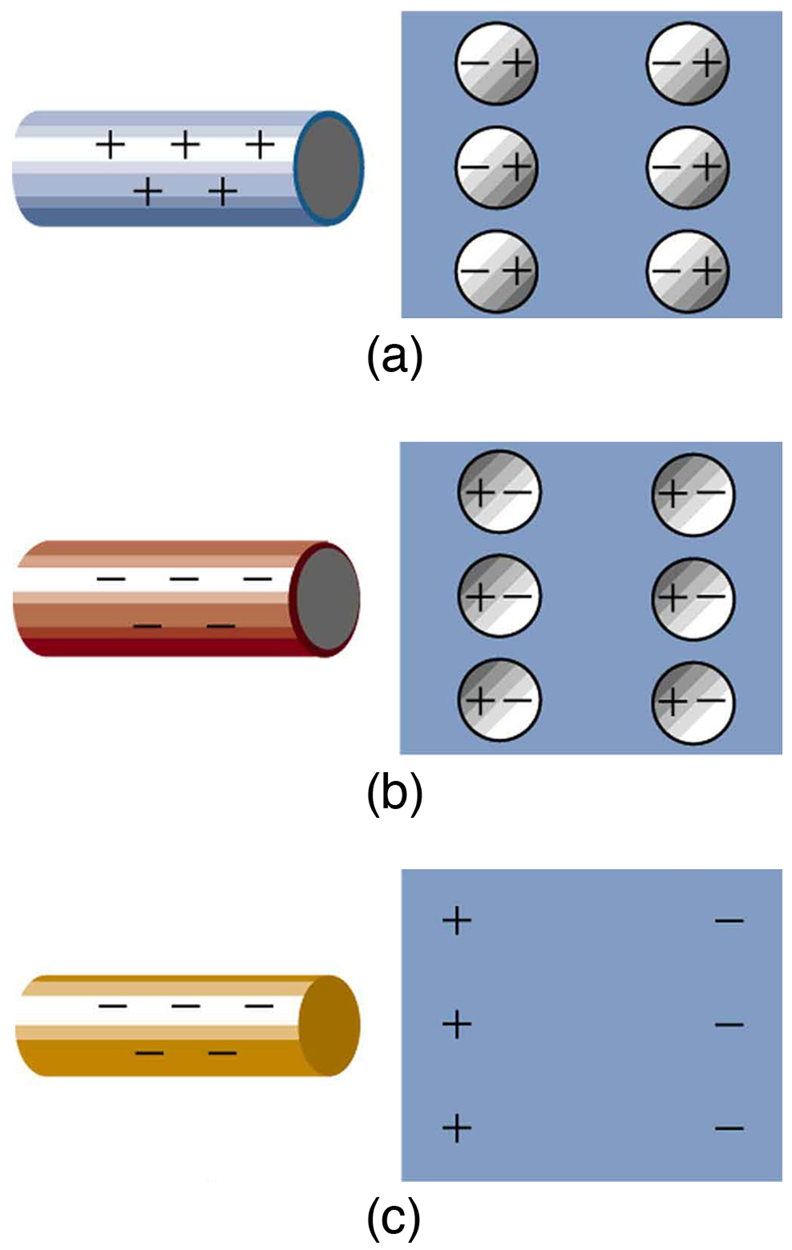
Neutral objects can be attracted to any charged object. The pieces of straw attracted to polished amber are neutral, for example. If you run a plastic comb through your hair, the charged comb can pick up neutral pieces of paper. Figure 5 shows how the polarization of atoms and molecules in neutral objects results in their attraction to a charged object.
When a charged rod is brought near a neutral substance, an insulator in this case, the distribution of charge in atoms and molecules is shifted slightly. Opposite charge is attracted nearer the external charged rod, while like charge is repelled. Since the electrostatic force decreases with distance, the repulsion of like charges is weaker than the attraction of unlike charges, and so there is a net attraction. Thus a positively charged glass rod attracts neutral pieces of paper, as will a negatively charged rubber rod. Some molecules, like water, are polar molecules. Polar molecules have a natural or inherent separation of charge, although they are neutral overall. Polar molecules are particularly affected by other charged objects and show greater polarization effects than molecules with naturally uniform charge distributions.
PhET Explorations: John Travoltage
Make sparks fly with John Travoltage. Wiggle Johnnie’s foot and he picks up charges from the carpet. Bring his hand close to the door knob and get rid of the excess charge.

Section Summary
- Polarization is the separation of positive and negative charges in a neutral object.
- A conductor is a substance that allows charge to flow freely through its atomic structure.
- An insulator holds charge within its atomic structure.
- Objects with like charges repel each other, while those with unlike charges attract each other.
- A conducting object is said to be grounded if it is connected to the Earth through a conductor. Grounding allows transfer of charge to and from the earth’s large reservoir.
- Objects can be charged by contact with another charged object and obtain the same sign charge.
- If an object is temporarily grounded, it can be charged by induction, and obtains the opposite sign charge.
- Polarized objects have their positive and negative charges concentrated in different areas, giving them a non-symmetrical charge.
- Polar molecules have an inherent separation of charge.
Conceptual Questions
1: An eccentric inventor attempts to levitate by first placing a large negative charge on himself and then putting a large positive charge on the ceiling of his workshop. Instead, while attempting to place a large negative charge on himself, his clothes fly off. Explain.
2: If you have charged an electroscope by contact with a positively charged object, describe how you could use it to determine the charge of other objects. Specifically, what would the leaves of the electroscope do if other charged objects were brought near its knob?
3: When a glass rod is rubbed with silk, it becomes positive and the silk becomes negative—yet both attract dust. Does the dust have a third type of charge that is attracted to both positive and negative? Explain.
4: Why does a car always attract dust right after it is polished? (Note that car wax and car tires are insulators.)
5: Describe how a positively charged object can be used to give another object a negative charge. What is the name of this process?
6: What is grounding? What effect does it have on a charged conductor? On a charged insulator?
Problems & Exercises
1: Suppose a speck of dust in an electrostatic precipitator has 1.0000 x 10 12 protons in it and has a net charge of –5.00 nC (a very large charge for a small speck). How many electrons does it have?
2: An amoeba has a net charge of + 0.300 pC and it has 1.00 x 1016 protons. (a) How many fewer electrons are there than protons? (b) If you paired them up, what fraction of the protons would have no electrons?
3: A 50.0 g ball of copper has a net charge of 2.00 microcoloumbs. What fraction of the copper’s electrons has been removed? (Each copper atom has 29 protons, and copper has an atomic mass of 63.5.)
4: What net charge would you place on a 100 gram piece of sulphur if you put an extra electron on 1 in 1012 of its atoms? (Sulphur has an atomic mass of 32.1.)
5: How many coulombs of positive charge are there in 4.00 kg of plutonium, given its atomic mass is 244 and that each plutonium atom has 94 protons?
Glossary
- free electron
- an electron that is free to move away from its atomic orbit
- conductor
- a material that allows electrons to move separately from their atomic orbits
- insulator
- a material that holds electrons securely within their atomic orbits
- grounded
- when a conductor is connected to the Earth, allowing charge to freely flow to and from Earth’s unlimited reservoir
- induction
- the process by which an electrically charged object brought near a neutral object creates a charge in that object
- polarization
- slight shifting of positive and negative charges to opposite sides of an atom or molecule
- electrostatic repulsion
- the phenomenon of two objects with like charges repelling each other
Solutions
Check Your Understanding
Water molecules are polarized, giving them slightly positive and slightly negative sides. This makes water even more susceptible to a charged rod’s attraction. As the water flows downward, due to the force of gravity, the charged conductor exerts a net attraction to the opposite charges in the stream of water, pulling it closer.
Problems & Exercises
- proton’s total charge = +160 nC. Net charge is =5 nC so electron’s total charge = -165 nC 165 nC = 1.03 x1012 electrons.
- Net charge of + 0.300 pC = 3.00 x 10-11 C means there is a “missing” charge of – 3.00 x 10-11 C (- 3.00 x 10-11 C ) ( 1 electron / – 1.602 x10-19 C ) = 1.87 x106 electrons. Fraction difference = (1.87 x106 electrons) / (1.00 x 1016 protons) = 1.87 x 10-10
- 1 copper atom mass = 63.5 amu x 1.66×10-27 kg / amu = 1.05 x 10-25 kg 50.0 grams = 0.0500 kg so 50.0 grams of copper = 4.76 x 1023 atoms of copper so if electrically neutral charge of electrons would be 2.21 x 10 6 C But it has a net charge of 2 x 10-6 C Fraction = ( 2 x 10-6 C ) / ( 2.21 x 106 C ) = 9.09 x 10 -13
- Sulphur has an atomic weight of 32.1 so 1 atom mass = 32.1 amu = (32.1 amu) (1.66×10-27 kg / amu) = 5.33 x 10-26 kg In 100 grams of sulphur there are (0.100 kg)(1 atom / 5.33×10-26 kg) = 1.88 x 10 24 atoms If 1 in 1012 atoms have one extra electron then there are ( 1.88 x 10 24 atoms ) / (1 x1012 ) = 1.88 x 1012 extra electrons. Those extra electrons have a net charge of ( 1.88 x 1012 electrons ) ( 1.602 x 10 -19 C/electron) = 3.00 x 10-7 C .
- 9.88 x 1024 atoms = 9.28 x 10 26 protons = 1.48x 108 C


