Atomic Structure
Electrical Charge
If we were to take a balloon, rub it on our hair, and slowly pull the balloon away, our hair would seem to not want to disconnect from the balloon.
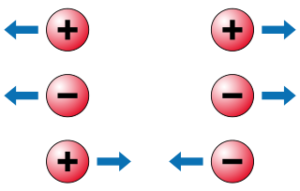
The balloon, being rubbed against our hair, is passing electrons from its valence shell to our hair. This causes our hair to have more electrons than protons, making it negatively charged. The balloon, having lost some of its electrons, has more protons than electrons, making it positively charged. In the illustration presented in Figure 2, you can see how different charges react to one another.
An electrical charge is the electrical characteristic of matter that is caused by its deficiency or excess of electrons.
KEY TAKEAWAY
Three important rules to remember about charges:
- Positive charges repel each other.
- Negative charges repel each other.
- Opposite charges attract.