Atomic Structure
Structure of the Atom
TEST YOUR KNOWLEDGE!
Before starting the chapter, let’s see how much you know!
The atom is the smallest particle of any matter. Everything is made up of atoms. Atoms have three main parts:
- Protons, positively charged particles, make up part of the nucleus, which is in the center of the atom.
- Neutrons, uncharged particles, make up the other part of the nucleus.
- Electrons, negatively charged particles, orbit around the nucleus like the planets orbit the sun in our solar system.
IMAGE HOTSPOT
Click on the “+” symbols in the image to find more information.
Valence Shells
You may remember from chemistry class that electrons rotate around the nucleus of the atom. An atom has a certain number of shells that surround the nucleus. Each shell has a maximum number of electrons that rotate in its orbit.
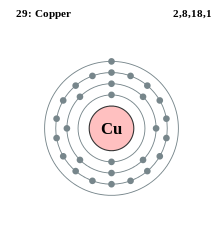
1st shell – 2 electrons
2nd shell – 8 electrons
3rd shell – 18 electrons
4th shell – 32 electrons
5th shell – 50 electrons
While it is very cool to know this, it is not necessary for us as electricians to memorize these numbers. What does concern us is the outermost shell. This is called the valence shell and can only contain up to a maximum of 8 valence electrons. It is this shell that determines if a substance is a conductor, insulator, or semiconductor.
Conductors have 1–3 valence electrons.
Semiconductors have 4 valence electrons.
Insulators have 5–8 valence electrons.

