Reproduction
Unit 10: Reproduction and Development
Unit outline
Part 1: Anatomy and Physiology of the Male Reproductive System
- Scrotum
- Testes
- Structure of formed sperm
- Sperm transport
- The penis
- Testosterone
- Function of testosterone
Part 2: Anatomy and Physiology of the Female Reproductive System
- External female genitalia
- Vagina
- Ovaries
- The ovarian cycle
- Hormonal control of the ovarian cycle
- The uterine tubes
- The uterus and cervix
- The menstrual cycle
- The breasts
Part 3: Fertilization
- Transit of sperm
- Contact between sperm and oocyte
- The zygote
Part 4: Embryonic Development
- Pre-implantation embryonic development
- Implantation
- Embryonic membranes
- Germ layers
- Development of the placenta
Part 5: Fetal Development
- The fetal circulatory system
Part 6: Maternal Changes During Pregnancy, Labor, and Birth
- Effects of hormones
- Physiology of labor
- Stages of childbirth
Part 7: Adjustments of the Infant at Birth and Postnatal Stages
- Reproductive adjustments
- Circulatory adjustments
Part 8: Lactation
- Structure of lactating breast
- The process of lactation
- Changes in composition of breast milk
Learning Objectives
At the end of this unit, you should be able to:
I. Describe the location, structure and function(s) of the components of the male reproductive system.
II. Describe the location, structure and function(s) of the components of the female reproductive system.
III. Describe the roles of FSH, LH, and testosterone or estrogens in the male and female reproductive systems
IV. Describe the mechanism, stages, and anatomical pathway of sperm release and ovum release
V. Describe the ovarian cycle as well as the role of the hypothalamus and anterior pituitary gland in this cycle.
VI. Describe the uterine (menstrual) cycle as well as the names and sources of any hormones involved.
VII. Correlate the ovarian and uterine (menstrual) cycles and explain their integrated hormonal regulation.
VIII. Describe the roles of the corpus luteum and placenta if pregnancy occurs.
IX. Distinguish between morula and blastocyst.
X. Identify the three primary germ layers and which body parts they develop into.
XI. Describe the formation and functions of each of the four extra-embryonic membranes.
XII. Describe the formation, structure, and functions of the placenta and umbilical cord.
XIII. Explain the mechanisms underlying the production of fraternal twins and identical twins.
XIV. Describe the endocrine regulation (estrogen, progesterone, prolactin, oxytocin) and maintenance of pregnancy, labor, and lactation.
XV. Name the three stages of labor and describe the events that occur during each stage.
XVI. Describe fetal circulation and the changes that occur in fetal circulation following delivery.
XVII. Describe the respiratory system of the neonate with respect to the importance of surfactant, fluid in the lungs, and initiation of ventilation.
Learning Objectives and Guiding Questions
At the end of this unit, you should be able to complete all the following tasks, including answering the guiding questions associated with each task.
I. Describe the location, structure and function(s) of each of the following components of the male reproductive system:
- Testes
- Epididymis
- Vas deferens
- Ejaculatory duct
- Urethra
- Seminal vesicles (seminal glands)
- Prostate gland
- Penis
- Bulbourethral gland
- Scrotum
- Seminiferous tubules
II. Describe the location, structure and function(s) of each of the following components of the female reproductive system:
- Ovaries
- Uterine tubes
- Uterus
- Cervix
- Clitoris
- Vagina
- Endometrium
- Myometrium
- Perimetrium
- Bartholin’s glands
III. Describe the roles of FSH, LH, and testosterone or estrogens in the male and female reproductive systems
IV. Describe the mechanism, stages, and anatomical pathway of sperm release and ovum release
V. Describe the ovarian cycle. Include in your description the name of the three stages of the cycle and the events that occur during each stage, as well as the role of the hypothalamus and anterior pituitary gland in this cycle.
VI. Describe the uterine (menstrual) cycle. Include in your description the name of the three stages of the cycle and the events that occur during each stage, as well as the names and sources of any hormones involved.
VII. Correlate the ovarian and uterine (menstrual) cycles and explain their integrated hormonal regulation by drawing a fully-annotated diagram showing the hormonal interactions between the ovarian and uterine cycles.
VIII. Describe the roles of the corpus luteum and placenta if pregnancy occurs.
IX. Distinguish between morula and blastocyst.
X. Identify the three primary germ layers and which body parts they develop into.
- Name the primary germ layer from which each of the following structures develops:
- Nervous system
- Sensory organs
- Epidermis
- Hair
- Nails
- Skeleton
- Muscles
- Connective tissue
- Heart
- Blood vessels
- Kidneys
- Gastrointestinal tract lining
- Liver
- Pancreas
- Lungs
XI. Describe the formation and functions of each of the four extra-embryonic membranes.
XII. Describe the formation, structure, and functions of the placenta and umbilical cord.
XIII. Explain the mechanisms underlying the production of fraternal twins and identical twins.
XIV. Describe the endocrine regulation (estrogen, progesterone, prolactin, oxytocin) and maintenance of pregnancy, labor, and lactation.
XV. Name the three stages of labor and describe the events that occur during each stage.
XVI. Describe fetal circulation and the changes that occur in fetal circulation following delivery.
- Name all the vessels or structures found in the fetal circulation but not the adult, and for each one name the structure they develop into after birth.
XVII. Describe the respiratory system of the neonate with respect to the importance of surfactant, fluid in the lungs, and initiation of ventilation.
Small, uncoordinated, and slick with amniotic fluid, a newborn encounters the world outside of her mother’s womb. We do not often consider that a child’s birth is proof of the healthy functioning of both her mother’s and father’s reproductive systems. Moreover, her parents’ endocrine systems had to secrete the appropriate regulating hormones to induce the production and release of unique male and female gametes, reproductive cells containing the parents’ genetic material (one set of 23 chromosomes). Her parent’s reproductive behavior had to facilitate the transfer of male gametes—the sperm—to the female reproductive tract at just the right time to encounter the female gamete, an oocyte (egg). Finally, combination of the gametes (fertilization) had to occur, followed by implantation and development. In this unit, you will explore the male and female reproductive systems, whose healthy functioning can culminate in the powerful sound of a newborn’s first cry.
Part 1: Anatomy and Physiology of the Male Reproductive System
Unique for its role in human reproduction, a gamete is a specialized sex cell carrying 23 chromosomes—one half the number in body cells. At fertilization, the chromosomes in one male gamete, called a sperm (or spermatozoon), combine with the chromosomes in one female gamete, called an oocyte. The function of the male reproductive system (Figure 1) is to produce sperm and transfer them to the female reproductive tract. The paired testes are a crucial component in this process, as they produce both sperm and androgens, the hormones that support male reproductive physiology. In humans, the most important male androgen is testosterone. Several accessory organs and ducts aid the process of sperm maturation and transport the sperm and other seminal components to the penis, which delivers sperm to the female reproductive tract. In this section, we examine each of these different structures, and discuss the process of sperm production and transport.

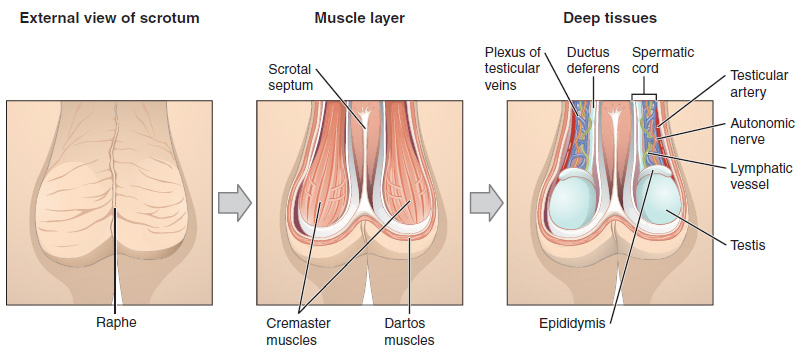
Scrotum: The testes are located in a skin-covered, highly pigmented, muscular sac called the scrotum that extends from the body behind the penis (Figure 1). This location is important in sperm production, which occurs within the testes, and proceeds more efficiently when the testes are kept 2 to 4°C below core body temperature.
Testes: The testes (singular = testis) are the male gonads. They produce both sperm and androgens, such as testosterone, and are active throughout the reproductive lifespan of the male.
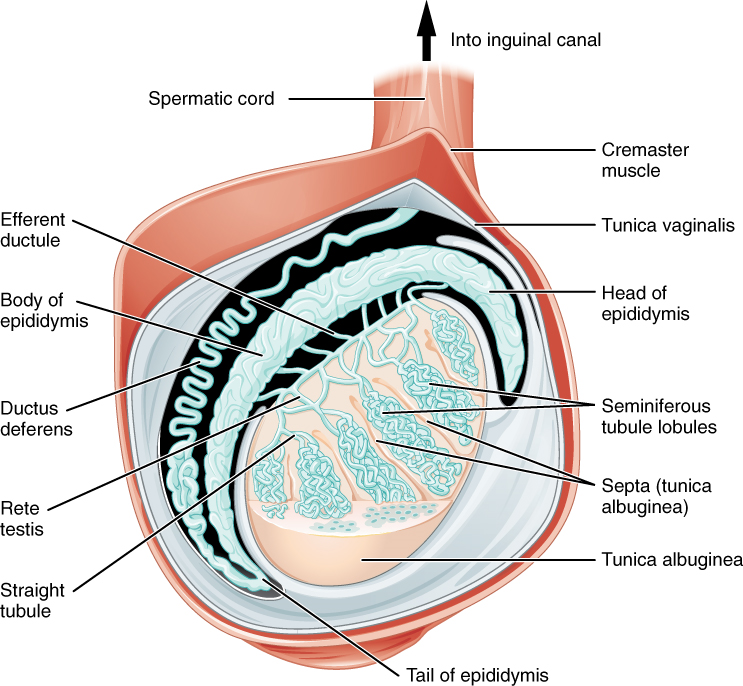
Paired ovals, the testes are each approximately 4 to 5 cm in length and are housed within the scrotum (Figure 2). Within the testes, sperm develop in structures called seminiferous tubules. During the seventh month of the developmental period of a male fetus, each testis moves through the abdominal musculature to descend into the scrotal cavity. This is called the “descent of the testis.”
The tightly coiled seminiferous tubules form the bulk of each testis. They are composed of developing sperm cells surrounding a lumen, the hollow center of the tubule, where formed sperm are released into the duct system of the testis (Figure 3). Specifically, from the lumens of the seminiferous tubules, sperm move into the straight tubules then into a fine meshwork of tubules and then leave the testis itself.
Inside the seminiferous tubules are supporting cells and developing sperm cells called germ cells. Germ cell development progresses from the basement membrane—at the perimeter of the tubule—toward the lumen.
1. Sertoli Cells: Surrounding all stages of the developing sperm cells are elongate, branching Sertoli cells. Sertoli cells secrete signaling molecules that promote sperm production and can control whether germ cells live or die.
2. Germ Cells: The least mature cells line the basement membrane inside the tubule. These divide to produce primary and secondary spermatocytes, then spermatids, which finally produce formed sperm. This process is called spermatogenesis. The process begins at puberty, after which time sperm are produced constantly throughout a man’s life. Eventually, the sperm are released into the lumen and are moved along a series of ducts in the testis toward a structure called the epididymis for the next step of sperm maturation.
Structure of Formed Sperm: Sperm are smaller than most cells in the body; in fact, the volume of a sperm cell is 85,000 times less than that of the female gamete. Approximately 100 to 300 million sperm are produced each day, whereas women typically ovulate only one oocyte per month as is true for most cells in the body, the structure of sperm cells speaks to their function. Sperm have a distinctive head, mid-piece, and tail region (Figure 4). The head of the sperm contains the extremely compact haploid (half the genetic content of a diploid somatic cell) nucleus with very little cytoplasm. These qualities contribute to the overall small size of the sperm (the head is only 5 μm long). A structure called the acrosome covers most of the head of the sperm cell as a “cap” that is filled with lysosomal enzymes important for preparing sperm to participate in fertilization. Tightly packed mitochondria fill the midpiece of the sperm. ATP produced by these mitochondria will power the flagellum, which extends from the neck and the mid-piece through the tail of the sperm, enabling it to move the entire sperm cell. The central strand of the flagellum, the axial filament, is formed from one centriole inside the maturing sperm cell during the final stages of spermatogenesis.
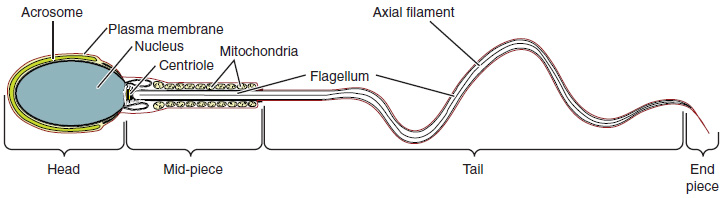
Sperm Transport: To fertilize an egg, sperm must be moved from the seminiferous tubules in the testes, through the epididymis, and—later during ejaculation—along the length of the penis and out into the female reproductive tract.
1. Role of the Epididymis: From the lumen of the seminiferous tubules, the immotile sperm are surrounded by testicular fluid and moved to the epididymis (plural = epididymides), a coiled tube attached to the testis where newly formed sperm continue to mature (Figure 3). Though the epididymis does not take up much room in its tightly coiled state, it would be approximately 6 m (20 feet) long if straightened. It takes an average of 12 days for sperm to move through the coils of the epididymis, with the shortest recorded transit time in humans being one day. Sperm enter the head of the epididymis and are moved along predominantly by the contraction of smooth muscles lining the epididymal tubes. As they are moved along the length of the epididymis, the sperm further mature and acquire the ability to move under their own power. Once inside the female reproductive tract, they will use this ability to move independently toward the unfertilized egg. The more mature sperm are then stored in the tail of the epididymis (the final section) until ejaculation occurs.
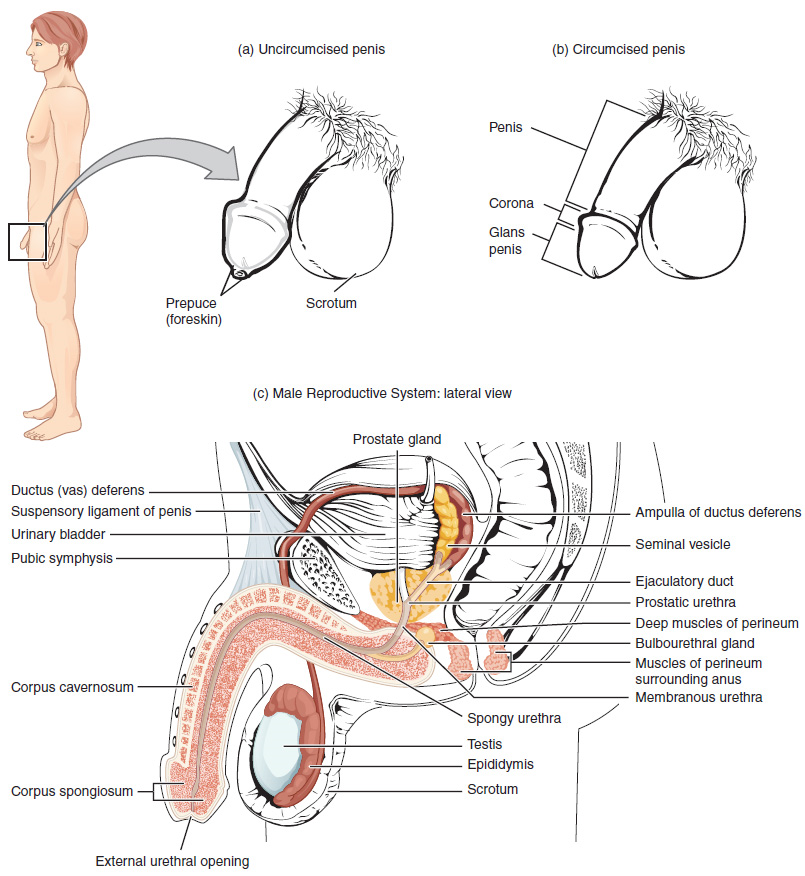
2. Duct System: During ejaculation, sperm exit the tail of the epididymis and are pushed by smooth muscle contraction to the ductus deferens (also called the vas deferens). The ductus deferens is a thick, muscular tube that is bundled together inside the scrotum with connective tissue, blood vessels, and nerves into a structure called the spermatic cord (Figure 1 and Figure 2). Because the ductus deferens is physically accessible within the scrotum, surgical sterilization to interrupt sperm delivery can be performed by cutting and sealing a small section of the ductus (vas) deferens. This procedure is called a vasectomy, and it is an effective form of male birth control. Although it may be possible to reverse a vasectomy, clinicians consider the procedure permanent, and advise men to undergo it only if they are certain they no longer wish to father children.
From each epididymis, each ductus deferens extends superiorly into the abdominal cavity through the inguinal canal in the abdominal wall. From here, the ductus deferens continues posteriorly to the pelvic cavity, ending posterior to the bladder where it dilates in a region called the ampulla (meaning “flask”).
Sperm make up only 5 percent of the final volume of semen, the viscous, whitish-gray fluid that the male ejaculates. The bulk of semen is produced by three critical accessory glands of the male reproductive system: the seminal vesicles, the prostate, and the bulbourethral glands.
3. Seminal Vesicles: As sperm pass through the ampulla of the ductus deferens at ejaculation, they mix with fluid from the associated seminal vesicle (Figure 1). The paired seminal vesicles are glands that contribute approximately 60 percent of the semen volume. Seminal vesicle fluid contains large amounts of fructose, which is used by the sperm mitochondria to generate ATP to allow movement through the female reproductive tract.
The fluid, now containing both sperm and seminal vesicle secretions, next moves into the associated ejaculatory duct, a short structure formed from the ampulla of the ductus deferens and the duct of the seminal vesicle. The paired ejaculatory ducts transport the seminal fluid into the next structure, the prostate gland.
4. Prostate Gland: The centrally located prostate gland sits anterior to the rectum at the base of the bladder surrounding the prostatic urethra, the portion of the urethra that runs within the prostate (Figure 1). About the size of a walnut, the prostate is formed of both muscular and glandular tissues. It excretes a fluid enriched with enzymes and citric acid to the passing seminal fluid—now called semen—that is critical to first coagulate and then decoagulate the semen following ejaculation. The temporary thickening of semen helps retain it within the female reproductive tract, providing time for sperm to utilize the fructose provided by seminal vesicle secretions. When the semen regains its fluid state, sperm can then pass farther into the female reproductive tract.
5. Bulbourethral Glands: The final addition to semen is made by two bulbourethral glands (or Cowper’s glands) that release a thick, salty fluid that lubricates the end of the urethra and the vagina and helps to clean urine residues from the penile urethra. The fluid from these accessory glands is released after the male becomes sexually aroused, and shortly before the release of the semen. It is therefore sometimes called pre-ejaculate. It is important to note that, in addition to the lubricating proteins, it is possible for bulbourethral fluid to pick up sperm already present in the urethra, and therefore it may be able to cause pregnancy.
The Penis: The penis is the male organ of copulation (sexual intercourse). It is flaccid for non-sexual actions, such as urination, and turgid and rod-like with sexual arousal. When erect, the stiffness of the organ allows it to penetrate into the vagina and deposit semen into the female reproductive tract.
The shaft of the penis surrounds the urethra (Figure 1). The end of the penis, called the glans penis, has a high concentration of nerve endings, resulting in very sensitive skin that influences the likelihood of ejaculation (see Figure 1). The skin from the shaft extends down over the glans and forms a collar called the prepuce (or foreskin). The foreskin also contains a dense concentration of nerve endings, and both lubricate and protect the sensitive skin of the glans penis. A surgical procedure called circumcision, often performed for religious or social reasons, removes the prepuce, typically within days of birth.
Testosterone: Testosterone, an androgen, is a steroid hormone produced by Leydig cells. The alternate term for Leydig cells, interstitial cells, reflects their location between the seminiferous tubules in the testes. In male embryos, testosterone is secreted by Leydig cells by the seventh week of development, with peak concentrations reached in the second trimester. This early release of testosterone results in the anatomical differentiation of the male sexual organs. In childhood, testosterone concentrations are low. They increase during puberty, activating characteristic physical changes and initiating spermatogenesis.
Functions of Testosterone: The continued presence of testosterone is necessary to keep the male reproductive system working properly, and Leydig cells produce approximately 6 to 7 mg of testosterone per day. Testicular steroidogenesis (the manufacturing of androgens, including testosterone) results in testosterone concentrations that are 100 times higher in the testes than in the circulation. Maintaining these normal concentrations of testosterone promotes spermatogenesis, whereas low levels of testosterone can lead to infertility. In addition to intratesticular secretion, testosterone is also released into the systemic circulation and plays an important role in muscle development, bone growth, the development of secondary sex characteristics, and maintaining libido (sex drive) in both males and females. In females, the ovaries secrete small amounts of testosterone, although most is converted to estradiol. A small amount of testosterone is also secreted by the adrenal glands in both sexes.
Part 2: Anatomy and Physiology of the Female Reproductive System
The female reproductive system functions to produce gametes and reproductive hormones, just like the male reproductive system; however, it also has the additional task of supporting the developing fetus and delivering it to the outside world. Unlike its male counterpart, the female reproductive system is located primarily inside the pelvic cavity (Figure 5). Recall that the ovaries are the female gonads. The gamete they produce is called an oocyte. We’ll discuss the production of oocytes in detail shortly. First, let’s look at some of the structures of the female reproductive system.

External Female Genitals: The external female reproductive structures are referred to collectively as the vulva. The mons pubis is a pad of fat found in both males and females that is located at the anterior, over the pubic bone. After puberty, it becomes covered in pubic hair. In females, the labia majora (labia = “lips”; majora = “larger”) are folds of hair-covered skin that begin just posterior to the mons pubis. The thinner and more pigmented labia minora (labia = “lips”; minora = “smaller”) extend medial to the labia majora. Although they naturally vary in shape and size from woman to woman, the labia minora serve to protect the female urethra and the entrance to the female reproductive tract.
The superior, anterior portions of the labia minora come together to encircle the clitoris (or glans clitoris), an organ that originates from the same cells as the glans penis and has abundant nerves that make it important in sexual sensation and orgasm. The hymen is a thin membrane that sometimes partially covers the entrance to the vagina. The vaginal opening is located between the opening of the urethra and the anus. It is flanked by outlets to the Bartholin’s glands (or greater vestibular glands).
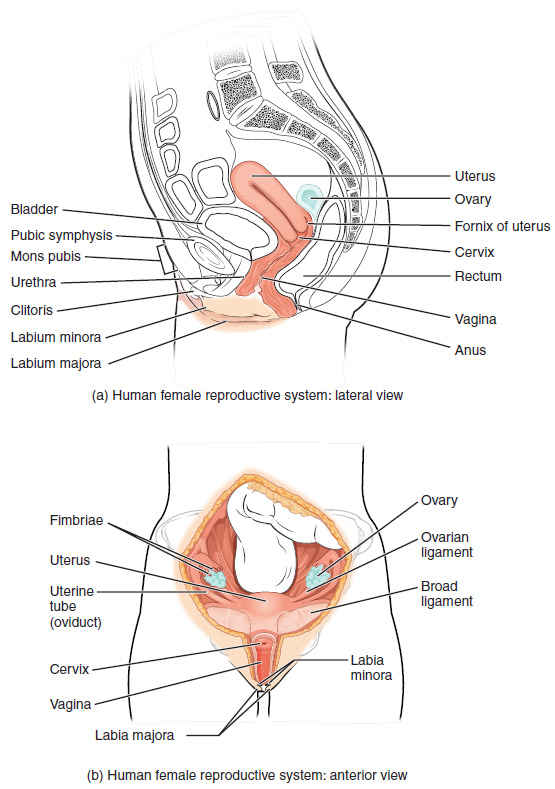
Vagina: The vagina is a muscular canal (approximately 10 cm long) that serves as the entrance to the reproductive tract (Figure 5). It also serves as the exit from the uterus during menses and childbirth. The outer walls of the anterior and posterior vagina are formed into longitudinal columns, or ridges, and the superior portion of the vagina—called the fornix—meets the protruding uterine cervix. The walls of the vagina are lined with an outer fibrous adventitia, a middle layer of smooth muscle, and an inner mucous membrane with transverse folds called rugae. Together, the middle and inner layers allow the expansion of the vagina to accommodate intercourse and childbirth. The Bartholin’s glands and the lesser vestibular glands (located near the clitoris) secrete mucus, which keeps the vestibular area moist.
The vagina is home to a normal population of microorganisms that help to protect against infection by pathogenic bacteria, yeast, or other organisms that can enter the vagina. In a healthy woman, the most predominant type of vaginal bacteria is from the genus Lactobacillus. This family of beneficial bacterial flora secretes lactic acid, and thus protects the vagina by maintaining an acidic pH (below 4.5). Potential pathogens are less likely to survive in these acidic conditions. Lactic acid, in combination with other vaginal secretions, makes the vagina a self-cleansing organ.
Ovaries: The ovaries are the female gonads (Figure 5). Paired ovals, they are each about 2 to 3 cm in length, about the size of an almond. The ovaries are located within the pelvic cavity, and are supported by ligaments.
The bulk of the adult ovary is composed of a tissue framework. Oocytes develop within the outer layer of this stroma, each surrounded by supporting cells. This grouping of an oocyte and its supporting cells is called a follicle. The growth and development of ovarian follicles will be described shortly.
The Ovarian Cycle: The ovarian cycle is a set of predictable changes in a female’s oocytes and ovarian follicles. During a woman’s reproductive years, it is a roughly 28-day cycle that can be correlated with, but is not the same as, the menstrual cycle (discussed shortly). The cycle includes two interrelated processes: oogenesis (the production of female gametes) and folliculogenesis (the growth and development of ovarian follicles).
1. Oogenesis: Gametogenesis in females is called oogenesis. The process begins with the ovarian stem cells, or oogonia. Oogonia form primary oocytes in the fetal ovary prior to birth. These primary oocytes are then arrested in this stage, only to resume it years later, beginning at puberty and continuing until the woman is near menopause (the cessation of a woman’s reproductive functions). The number of primary oocytes present in the ovaries declines from one to two million in an infant, to approximately 400,000 at puberty, to zero by the end of menopause.
The initiation of ovulation—the release of an oocyte from the ovary—marks the transition from puberty into reproductive maturity for women. From then on, throughout a woman’s reproductive years, ovulation occurs approximately once every 28 days, triggered by a surge of LH just prior to ovulation.
The larger amount of cytoplasm contained in the female gamete is used to supply the developing zygote with nutrients during the period between fertilization and implantation into the uterus. Interestingly, sperm contribute only DNA at fertilization, not cytoplasm. Therefore, the cytoplasm and all the cytoplasmic organelles in the developing embryo are of maternal origin. This includes mitochondria, which contain their own DNA. Scientific research in the 1980s determined that mitochondrial DNA is maternally inherited, meaning that you can trace your mitochondrial DNA directly to your mother, her mother, and so on back through your female ancestors.
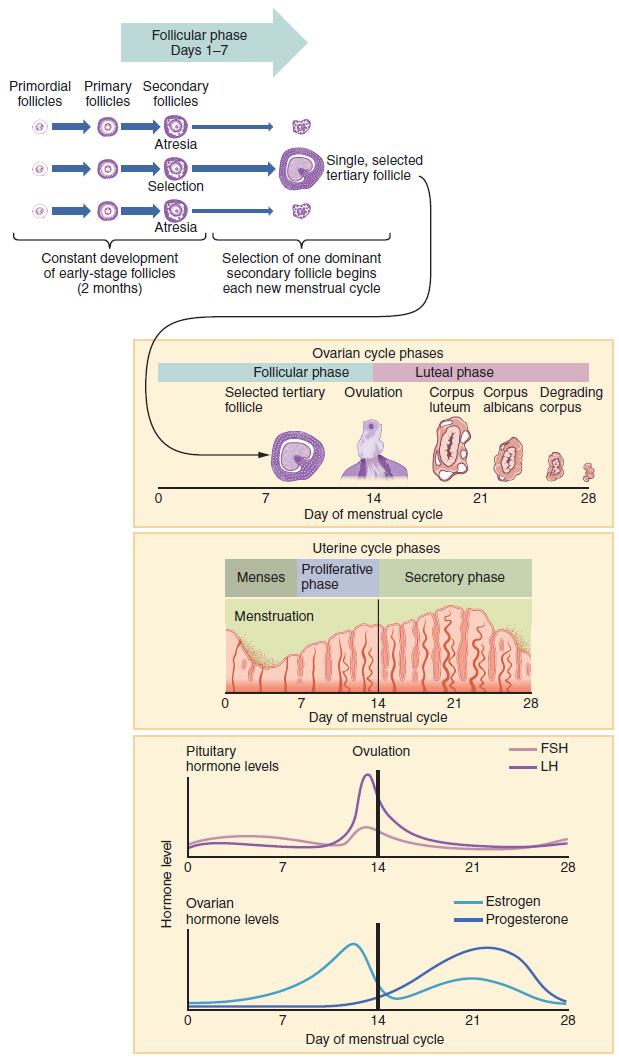
2. Folliculogenesis: Again, ovarian follicles are oocytes and their supporting cells. They grow and develop in a process called folliculogenesis, which typically leads to ovulation of one follicle approximately every 28 days, along with death to multiple other follicles. The death of ovarian follicles is called atresia and can occur at any point during follicular development. Recall that, a female infant at birth will have one to two million oocytes within her ovarian follicles, and that this number declines throughout life until menopause, when no follicles remain. As you will see next, follicles progress from primordial, to primary, to secondary and tertiary stages prior to ovulation—with the oocyte inside the follicle remaining as a primary oocyte until right before ovulation.
Folliculogenesis begins with follicles in a resting state. These small primordial follicles are present in newborn females and are the prevailing follicle type in the adult ovary (Figure 6). Primordial follicles have only a single flat layer of support cells, called granulosa cells, that surround the oocyte, and they can stay in this resting state for years—some until right before menopause.
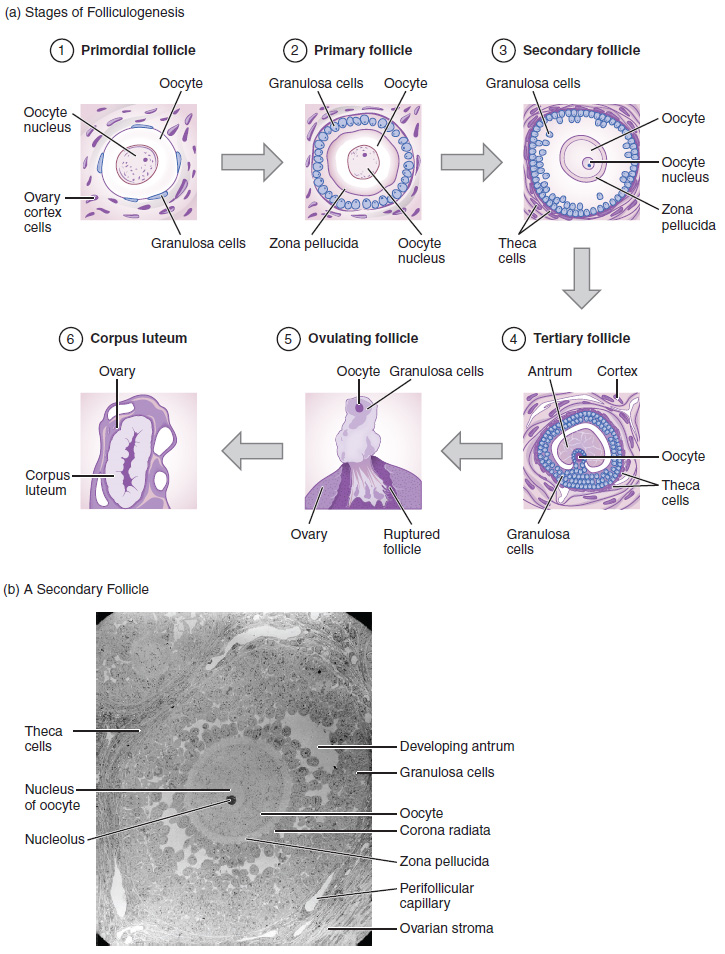
After puberty, a few primordial follicles will respond to a recruitment signal each day and will join a pool of immature growing follicles called primary follicles. Primary follicles start with a single layer of granulosa cells, but the granulosa cells then become active and transition from a flat or squamous shape to a rounded cuboidal shape as they increase in size and proliferate. As the granulosa cells divide, the follicles—now called secondary follicles (Figure 6)—increase in diameter, adding a new outer layer of connective tissue, blood vessels, and theca cells—cells that work with the granulosa cells to produce estrogens.
Within the growing secondary follicle, the primary oocyte now secretes a thin acellular membrane called the zona pellucida that will play a critical role in fertilization. A thick fluid, called follicular fluid, that has formed between the granulosa cells also begins to collect into one large pool, or antrum. Follicles in which the antrum has become large and fully formed are considered tertiary follicles (or antral follicles). Several follicles reach the tertiary stage at the same time, and most of these will undergo atresia. The one that does not die will continue to grow and develop until ovulation, when it will expel its secondary oocyte surrounded by several layers of granulosa cells from the ovary. Keep in mind that most follicles do not make it to this point. In fact, roughly 99 percent of the follicles in the ovary will undergo atresia, which can occur at any stage of folliculogenesis.
Hormonal Control of the Ovarian Cycle: The process of development that we have just described, from primordial follicle to early tertiary follicle, takes approximately two months in humans. The final stages of development of a small cohort of tertiary follicles, ending with ovulation of a secondary oocyte, occur over a course of approximately 28 days. These changes are regulated by many of the same hormones that regulate the male reproductive system, including GnRH, LH, and FSH.
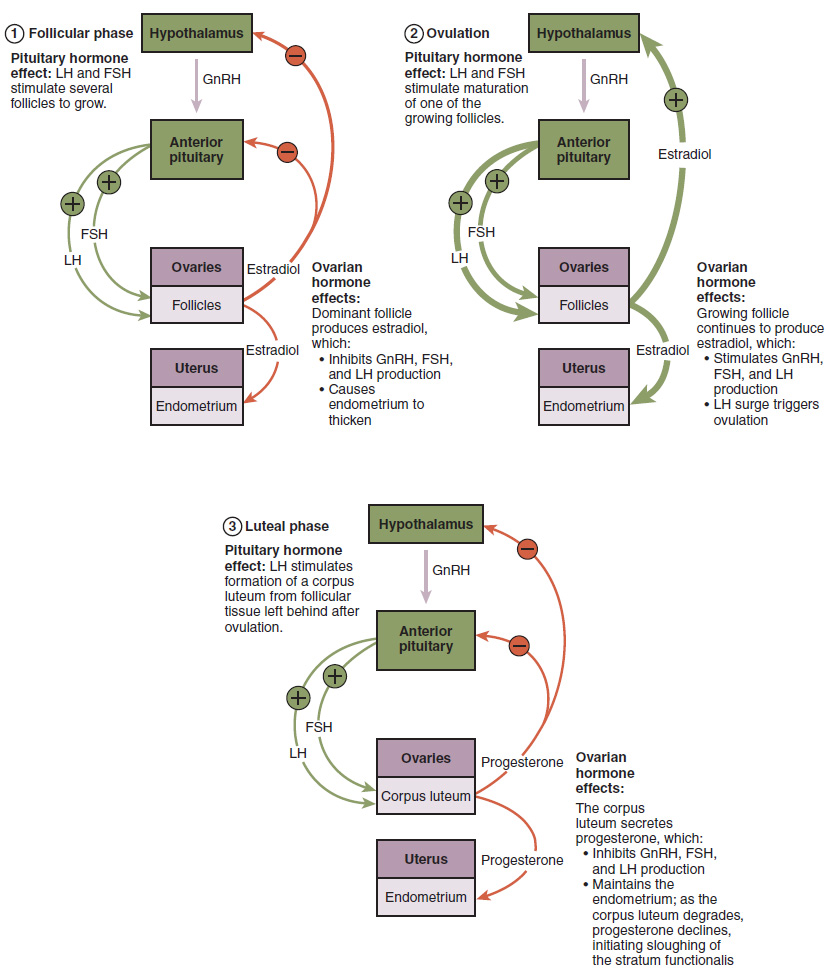
As in men, the hypothalamus produces GnRH, a hormone that signals the anterior pituitary gland to produce the gonadotropins FSH and LH (Figure 7). These gonadotropins leave the pituitary and travel through the bloodstream to the ovaries, where they bind to receptors on the granulosa and theca cells of the follicles. FSH stimulates the follicles to grow (hence its name of follicle-stimulating hormone), and the five or six tertiary follicles expand in diameter. The release of LH also stimulates the granulosa and theca cells of the follicles to produce the sex steroid hormone estradiol, a type of estrogen. This phase of the ovarian cycle, when the tertiary follicles are growing and secreting estrogen, is known as the follicular phase.
The more granulosa and theca cells a follicle has (that is, the larger and more developed it is), the more estrogen it will produce in response to LH stimulation. As a result of these large follicles producing large amounts of estrogen, systemic plasma estrogen concentrations increase. Following a classic negative feedback loop, the high concentrations of estrogen will inhibit the production of GnRH, LH, and FSH by the hypothalamus and pituitary. Because the large tertiary follicles require FSH to grow and survive at this point, this decline in FSH caused by negative feedback leads most of them to die (atresia). Typically, only one follicle, now called the dominant follicle, will survive this reduction in FSH, and this follicle will be the one that releases an oocyte.
When only the one dominant follicle remains in the ovary, it again begins to secrete estrogen. It produces more estrogen than all the developing follicles did together before the negative feedback occurred. It produces so much estrogen that the normal negative feedback does not occur. Instead, these extremely high concentrations of systemic plasma estrogen trigger a regulatory switch in the anterior pituitary that responds by secreting large amounts of LH and FSH into the bloodstream (Figure 7). The positive feedback loop by which more estrogen triggers release of more LH and FSH only occurs at this point in the cycle.
It is this large burst of LH (called the LH surge) that leads to ovulation of the dominant follicle. The LH surge induces many changes in the dominant follicle, including triggering proteases (enzymes that cleave proteins) to break down structural proteins in the ovary wall on the surface of the bulging dominant follicle. This degradation of the wall, combined with pressure from the large, fluid-filled antrum, results in the expulsion of the oocyte surrounded by granulosa cells into the peritoneal cavity. This release is ovulation.
In the next section, you will follow the ovulated oocyte as it travels toward the uterus, but there is one more important event that occurs in the ovarian cycle. The surge of LH also stimulates a change in the granulosa and theca cells that remain in the follicle after the oocyte has been ovulated. This change is called luteinization (recall that the full name of LH is luteinizing hormone), and it transforms the collapsed follicle into a new endocrine structure called the corpus luteum, a term meaning “yellowish body” (Figure 8). Instead of estrogen, the luteinized granulosa and theca cells of the corpus luteum begin to produce large amounts of the sex steroid hormone progesterone, a hormone that is critical for the establishment and maintenance of pregnancy. Progesterone triggers negative feedback at the hypothalamus and pituitary, which keeps GnRH, LH, and FSH secretions low, so no new dominant follicles develop at this time.
The post-ovulatory phase of progesterone secretion is known as the luteal phase of the ovarian cycle. If pregnancy does not occur within 10 to 12 days, the corpus luteum will stop secreting progesterone and degrade into the corpus albicans, a nonfunctional “whitish body” that will disintegrate in the ovary over a period of several months. During this time of reduced progesterone secretion, FSH and LH are once again stimulated, and the follicular phase begins again with a new cohort of early tertiary follicles beginning to grow and secrete estrogen.
The Uterine Tubes: The uterine tubes (also called fallopian tubes or oviducts) serve as the conduit of the oocyte from the ovary to the uterus (Figure 8). Each of the two uterine tubes is close to, but not directly connected to, the ovary and divided into sections. The isthmus is the narrow medial end of each uterine tube that is connected to the uterus. The wide distal infundibulum flares out with slender, finger-like projections called fimbriae. The middle region of the tube, called the ampulla, is where fertilization often occurs. The uterine tubes also contain ciliated cells that beat in the direction of the uterus, producing a current that will be critical to move the oocyte.
Following ovulation, the secondary oocyte surrounded by a few granulosa cells is released into the peritoneal cavity. The nearby uterine tube, either left or right, receives the oocyte. Unlike sperm, oocytes lack flagella, and therefore cannot move on their own. So how do they travel into the uterine tube and toward the uterus? High concentrations of estrogen that occur around the time of ovulation induce contractions of the smooth muscle along the length of the uterine tube and the result is a coordinated movement that sweeps the surface of the ovary and the pelvic cavity. Current flowing toward the uterus is generated by coordinated beating of the cilia that line the lumen of the length of the uterine tube. These cilia beat more strongly in response to the high estrogen concentrations that occur around the time of ovulation. As a result of these mechanisms, the oocyte–granulosa cell complex is pulled into the interior of the tube. Once inside, the muscular contractions and beating cilia move the oocyte slowly toward the uterus. When fertilization does occur, sperm typically meet the egg while it is still moving through the ampulla.
If the oocyte is successfully fertilized, the resulting zygote will begin to divide into two cells, then four, and so on, as it makes its way through the uterine tube and into the uterus. There, it will implant and continue to grow. If the egg is not fertilized, it will simply degrade—either in the uterine tube or in the uterus, where it may be shed with the next menstrual period.
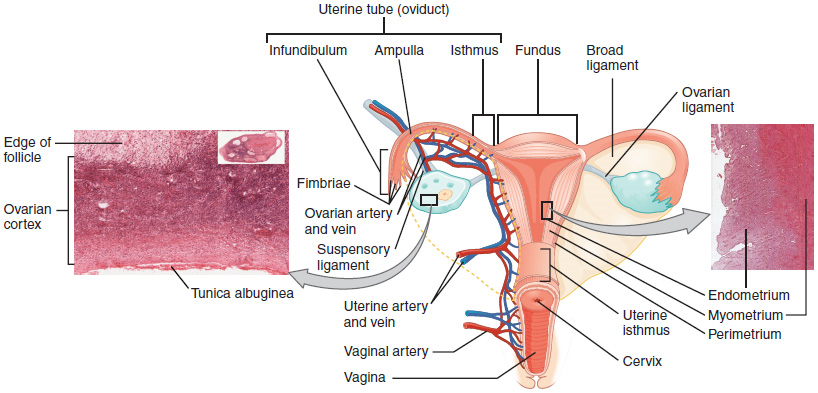
The Uterus and Cervix: The uterus is the muscular organ that nourishes and supports the growing embryo (Figure 8). Its average size is approximately 5 cm wide by 7 cm long (approximately 2 in by 3 in) when a female is not pregnant. It has three sections. The portion of the uterus superior to the opening of the uterine tubes is called the fundus. The middle section of the uterus is called the body of uterus (or corpus). The cervix is the narrow inferior portion of the uterus that projects into the vagina. The cervix produces mucus secretions that become thin and stringy under the influence of high systemic plasma estrogen concentrations, and these secretions can facilitate sperm movement through the reproductive tract. Several ligaments maintain the position of the uterus within the abdominopelvic cavity.
The wall of the uterus is made up of three layers. The most superficial layer is the serous membrane, or perimetrium, which consists of epithelial tissue that covers the exterior portion of the uterus. The middle layer, or myometrium, is a thick layer of smooth muscle responsible for uterine contractions. Most of the uterus is myometrial tissue, and the muscle fibers run horizontally, vertically, and diagonally, allowing the powerful contractions that occur during labor and the less powerful contractions (or cramps) that help to expel menstrual blood during a woman’s period.
The innermost layer of the uterus is called the endometrium. The endometrium contains a connective tissue lining, the lamina propria, which is covered by epithelial tissue that lines the lumen. Structurally, the endometrium consists of two layers: the stratum basalis and the stratum functionalis (the basal and functional layers). The stratum basalis layer is part of the lamina propria and is adjacent to the myometrium; this layer does not shed during menses. In contrast, the thicker stratum functionalis layer contains the glandular portion of the lamina propria and the endothelial tissue that lines the uterine lumen. It is the stratum functionalis that grows and thickens in response to increased levels of estrogen and progesterone. In the luteal phase of the menstrual cycle, special branches off of the uterine artery called spiral arteries supply the thickened stratum functionalis. This inner functional layer provides the proper site of implantation for the fertilized egg, and—should fertilization not occur—it is only the stratum functionalis layer of the endometrium that sheds during menstruation.
Recall that during the follicular phase of the ovarian cycle, the tertiary follicles are growing and secreting estrogen. At the same time, the stratum functionalis of the endometrium is thickening to prepare for a potential implantation. The post-ovulatory increase in progesterone, which characterizes the luteal phase, is key for maintaining a thick stratum functionalis. As long as a functional corpus luteum is present in the ovary, the endometrial lining is prepared for implantation. Indeed, if an embryo implants, signals are sent to the corpus luteum to continue secreting progesterone to maintain the endometrium, and thus maintain the pregnancy. If an embryo does not implant, no signal is sent to the corpus luteum and it degrades, ceasing progesterone production and ending the luteal phase. Without progesterone, the endometrium becomes thinner and, under the influence of prostaglandins, the spiral arteries of the endometrium constrict and rupture, preventing oxygenated blood from reaching the endometrial tissue. As a result, endometrial tissue dies and blood, pieces of the endometrial tissue, and white blood cells are shed through the vagina during menstruation, or the menses.
The Menstrual Cycle: Now that we have discussed the maturation of the cohort of tertiary follicles in the ovary, the build-up and then shedding of the endometrial lining in the uterus, and the function of the uterine tubes and vagina, we can put everything together to talk about the three phases of the menstrual cycle—the series of changes in which the uterine lining is shed, rebuilds, and prepares for implantation.
The timing of the menstrual cycle starts with the first day of menses, referred to as day one of a woman’s period. Cycle length is determined by counting the days between the onset of bleeding in two subsequent cycles. Because the average length of a woman’s menstrual cycle is 28 days, this is the time period used to identify the timing of events in the cycle. However, the length of the menstrual cycle varies among women, and even in the same woman from one cycle to the next, typically from 21 to 32 days.
Just as the hormones produced by the granulosa and theca cells of the ovary “drive” the follicular and luteal phases of the ovarian cycle, they also control the three distinct phases of the menstrual cycle. These are the menses phase, the proliferative phase, and the secretory phase.
1. Menses Phase: The menses phase of the menstrual cycle is the phase during which the lining is shed; that is, the days that the woman menstruates. Although it averages approximately five days, the menses phase can last from 2 to 7 days, or longer. The menses phase occurs during the early days of the follicular phase of the ovarian cycle, when progesterone, FSH, and LH levels are low (Figure 9). Recall that progesterone concentrations decline as a result of the degradation of the corpus luteum, marking the end of the luteal phase. This decline in progesterone triggers the shedding of the stratum functionalis of the endometrium.
2. Proliferative Phase: Once menstrual flow ceases, the endometrium begins to proliferate again, marking the beginning of the proliferative phase of the menstrual cycle (Figure 9). It occurs when the granulosa and theca cells of the tertiary follicles begin to produce increased amounts of estrogen. These rising estrogen concentrations stimulate the endometrial lining to rebuild.
Recall that the high estrogen concentrations will eventually lead to a decrease in FSH as a result of negative feedback, resulting in atresia of all but one of the developing tertiary follicles. The switch to positive feedback—which occurs with the elevated estrogen production from the dominant follicle—then stimulates the LH surge that will trigger ovulation. In a typical 28-day menstrual cycle, ovulation occurs on day 14. Ovulation marks the end of the proliferative phase as well as the end of the follicular phase.
3. Secretory Phase: In addition to prompting the LH surge, high estrogen levels increase the uterine tube contractions that facilitate the pick-up and transfer of the ovulated oocyte. High estrogen levels also slightly decrease the acidity of the vagina, making it more hospitable to sperm. In the ovary, the luteinization of the granulosa cells of the collapsed follicle forms the progesterone-producing corpus luteum, marking the beginning of the luteal phase of the ovarian cycle. In the uterus, progesterone from the corpus luteum begins the secretory phase of the menstrual cycle, in which the endometrial lining prepares for implantation (Figure 9). Over the next 10 to 12 days, the endometrial glands secrete a fluid rich in glycogen. If fertilization has occurred, this fluid will nourish the ball of cells now developing from the zygote. At the same time, the spiral arteries develop to provide blood to the thickened stratum functionalis.
If no pregnancy occurs within approximately 10 to 12 days, the corpus luteum will degrade into the corpus albicans. Levels of both estrogen and progesterone will fall, and the endometrium will grow thinner. Prostaglandins will be secreted that cause constriction of the spiral arteries, reducing oxygen supply. The endometrial tissue will die, resulting in menses—or the first day of the next cycle.
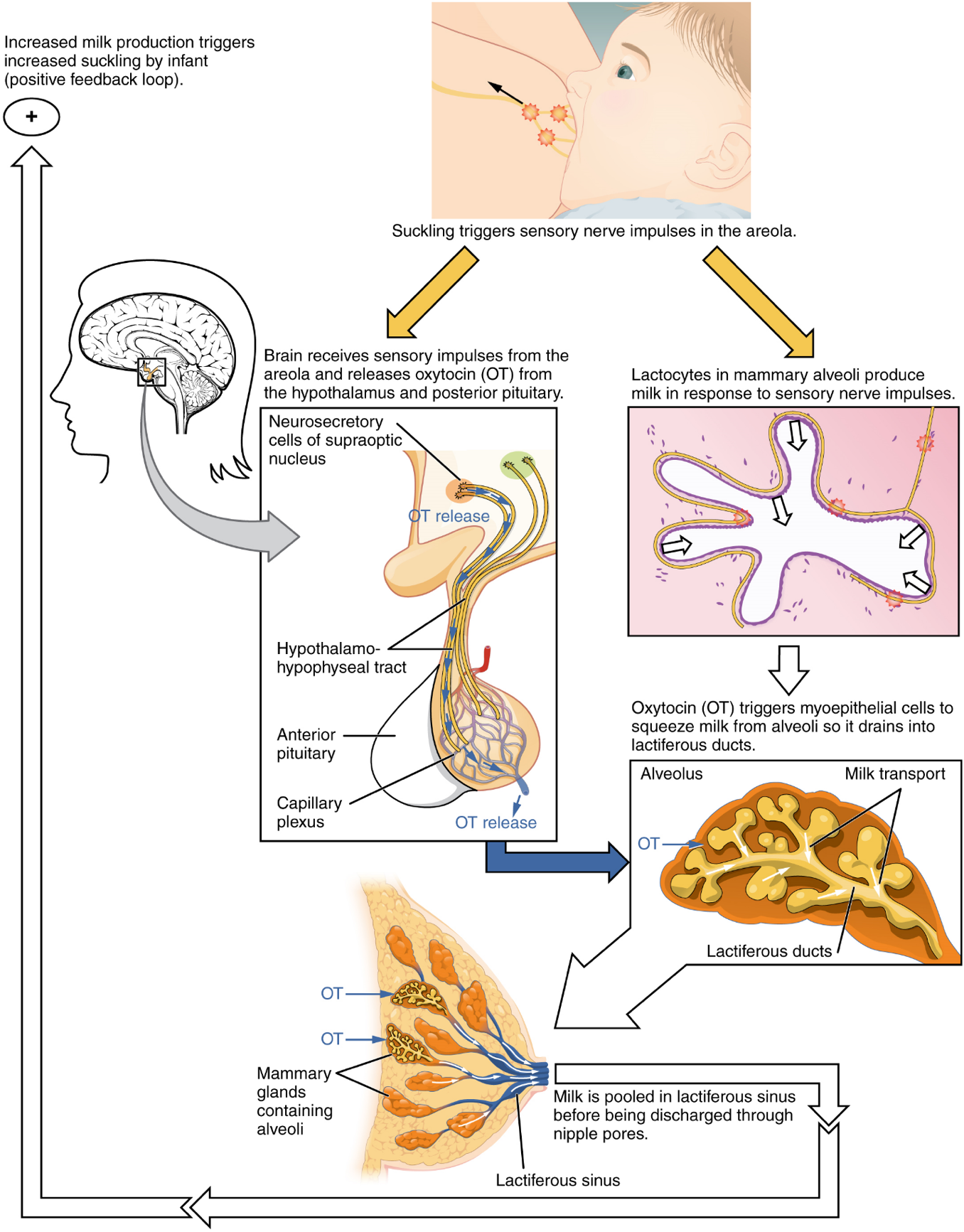
The Breasts: Although the breasts are located far from the other female reproductive organs, they are considered accessory organs of the female reproductive system. The function of the breasts is to supply milk to an infant in a process called lactation. The external features of the breast include a nipple surrounded by a pigmented areola (Figure 10), whose coloration may deepen during pregnancy. The areola is typically circular and can vary in size from 25 to 100 mm in diameter. The areolar region is characterized by small, raised areolar glands that secrete lubricating fluid during lactation to protect the nipple from chafing. When a baby nurses, or draws milk from the breast, the entire areolar region is taken into the mouth.
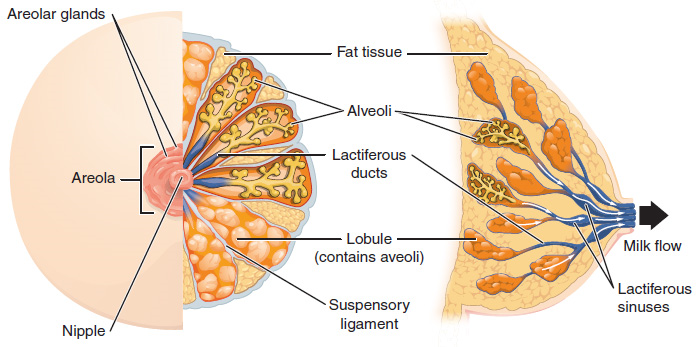
Breast milk is produced by the mammary glands, which are modified sweat glands. The milk itself exits the breast through the nipple via 15 to 20 lactiferous ducts that open on the surface of the nipple. These lactiferous ducts each extend to a lactiferous sinus that connects to a glandular lobe within the breast itself that contains groups of milk-secreting cells in clusters called alveoli (Figure 10). The clusters can change in size depending on the amount of milk in the alveolar lumen. Once milk is made in the alveoli, stimulated myoepithelial cells that surround the alveoli contract to push the milk to the lactiferous sinuses. From here, the baby can draw milk through the lactiferous ducts by suckling. The lobes themselves are surrounded by fat tissue, which determines the size of the breast; breast size differs between individuals and does not affect the amount of milk produced. Supporting the breasts are multiple bands of connective tissue called suspensory ligaments that connect the breast tissue to the dermis of the overlying skin.
Part 3: Fertilization
Fertilization occurs when a sperm and an oocyte (egg) combine and their nuclei fuse. Because each of these reproductive cells is a haploid cell containing half of the genetic material needed to form a human being, their combination forms a diploid cell. This new single cell, called a zygote, contains all the genetic material needed to form a human—half from the mother and half from the father.
Transit of Sperm: Fertilization is a numbers game. During ejaculation, hundreds of millions of sperm (spermatozoa) are released into the vagina. Almost immediately, millions of these sperm are overcome by the acidity of the vagina (approximately pH 3.8), and millions more may be blocked from entering the uterus by thick cervical mucus. Of those that do enter, thousands are destroyed by phagocytic uterine leukocytes. Thus, the race into the uterine tubes, which is the most typical site for sperm to encounter the oocyte, is reduced to a few thousand contenders. Their journey—thought to be facilitated by uterine contractions—usually takes from 30 minutes to 2 hours. If the sperm do not encounter an oocyte immediately, they can survive in the uterine tubes for another 3–5 days. Thus, fertilization can still occur if intercourse takes place a few days before ovulation. In comparison, an oocyte can survive independently for only approximately 24 hours following ovulation. Intercourse more than a day after ovulation will therefore usually not result in fertilization.
During the journey, fluids in the female reproductive tract prepare the sperm for fertilization through a process called capacitation, or priming. The fluids improve the motility of the spermatozoa. They also deplete cholesterol molecules embedded in the membrane of the head of the sperm, thinning the membrane in such a way that will help facilitate the release of the lysosomal (digestive) enzymes needed for the sperm to penetrate the oocyte’s exterior once contact is made. Sperm must undergo the process of capacitation in order to have the “capacity” to fertilize an oocyte. If they reach the oocyte before capacitation is complete, they will be unable to penetrate the oocyte’s thick outer layer of cells.
Contact Between Sperm and Oocyte: Upon ovulation, the oocyte released by the ovary is swept into—and along—the uterine tube. Fertilization must occur in the distal uterine tube because an unfertilized oocyte cannot survive the 72-hour journey to the uterus. As you will recall from your study of the oogenesis, this oocyte (specifically a secondary oocyte) is surrounded by two protective layers. The corona radiata is an outer layer of follicular (granulosa) cells that form around a developing oocyte in the ovary and remain with it upon ovulation. The underlying zona pellucida (pellucid = “transparent”) is a transparent, but thick, glycoprotein membrane that surrounds the cell’s plasma membrane.
As it is swept along the distal uterine tube, the oocyte encounters the surviving capacitated sperm, which stream toward it in response to chemical attractants released by the cells of the corona radiata. To reach the oocyte itself, the sperm must penetrate the two protective layers. The sperm first burrow through the cells of the corona radiata. Then, upon contact with the zona pellucida, the sperm bind to receptors in the zona pellucida. This initiates a process called the acrosomal reaction in which the enzyme-filled “cap” of the sperm, called the acrosome, releases its stored digestive enzymes. These enzymes clear a path through the zona pellucida that allows sperm to reach the oocyte. Finally, a single sperm makes contact with sperm-binding receptors on the oocyte’s plasma membrane (Figure 11). The plasma membrane of that sperm then fuses with the oocyte’s plasma membrane, and the head and mid-piece of the “winning” sperm enter the oocyte interior.
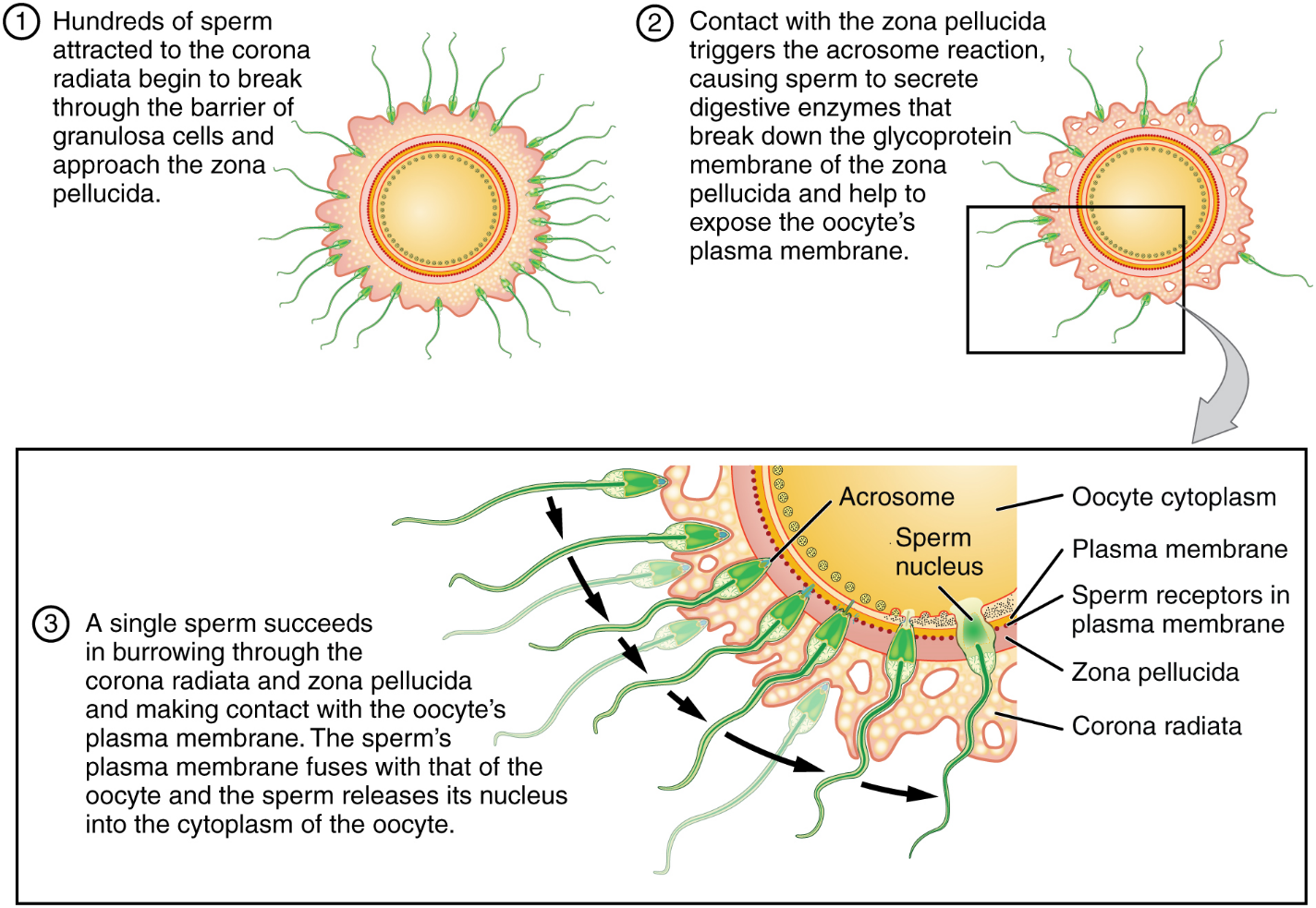
How do sperm penetrate the corona radiata? Some sperm undergo a spontaneous acrosomal reaction, which is an acrosomal reaction not triggered by contact with the zona pellucida. The digestive enzymes released by this reaction digest the extracellular matrix of the corona radiata. As you can see, the first sperm to reach the oocyte is never the one to fertilize it. Rather, hundreds of sperm cells must undergo the acrosomal reaction, each helping to degrade the corona radiata and zona pellucida until a path is created to allow one sperm to contact and fuse with the plasma membrane of the oocyte. If you consider the loss of millions of sperm between entry into the vagina and degradation of the zona pellucida, you can understand why a low sperm count can cause male infertility.
When the first sperm fuses with the oocyte, the oocyte deploys two mechanisms to prevent polyspermy, which is penetration by more than one sperm. This is critical because if more than one sperm were to fertilize the oocyte, the resulting zygote would be a triploid organism with three sets of chromosomes. This is incompatible with life.
The first mechanism is the fast block, which involves a near instantaneous change in sodium ion permeability upon binding of the first sperm, depolarizing the oocyte plasma membrane and preventing the fusion of additional sperm cells. The fast block sets in almost immediately and lasts for about a minute, during which time an influx of calcium ions following sperm penetration triggers the second mechanism, the slow block. In this process, referred to as the cortical reaction, cortical granules sitting immediately below the oocyte plasma membrane fuse with the membrane and release zonal inhibiting proteins and mucopolysaccharides into the space between the plasma membrane and the zona pellucida. Zonal inhibiting proteins cause the release of any other attached sperm and destroy the oocyte’s sperm receptors, thus preventing any more sperm from binding. The mucopolysaccharides then coat the nascent zygote in an impenetrable barrier that, together with hardened zona pellucida, is called a fertilization membrane.
The Zygote: Upon fertilization the two haploid nuclei derived from the sperm and oocyte decondense, expand, and replicate their DNA. The pronuclei then migrate toward each other, their nuclear envelopes disintegrate, and the male- and female-derived genetic material intermingles. This step completes the process of fertilization and results in a single-celled diploid zygote with all the genetic instructions it needs to develop into a human.
Most of the time, a woman releases a single egg during an ovulation cycle. However, in approximately 1 percent of ovulation cycles, two eggs are released and both are fertilized. Two zygotes form, implant, and develop, resulting in the birth of dizygotic (or fraternal) twins. Because dizygotic twins develop from two eggs fertilized by two sperm, they are no more identical than siblings born at different times.
Much less commonly, a zygote can divide into two separate offspring during early development. This results in the birth of monozygotic (or identical) twins. Although the zygote can split as early as the two-cell stage, splitting occurs most commonly during the early blastocyst stage, with roughly 70–100 cells present. These two scenarios are distinct from each other, in that the twin embryos that separated at the two-cell stage will have individual placentas, whereas twin embryos that form from separation at the blastocyst stage will share a placenta and a chorionic cavity.

Part 4: Embryonic Development
Throughout this chapter, we will express embryonic and fetal ages in terms of weeks from fertilization, commonly called conception. The period of time required for full development of a fetus in utero is referred to as gestation (gestare = “to carry” or “to bear”). It can be subdivided into distinct gestational periods. A developing human is referred to as an embryo during weeks 3–8, and a fetus from the ninth week of gestation until birth. In this section, we’ll cover the embryonic stages of development, which are characterized by cell division, migration, and differentiation. By the end of the embryonic period, all the organ systems are structured in rudimentary form, although the organs themselves are either nonfunctional or only semi-functional.
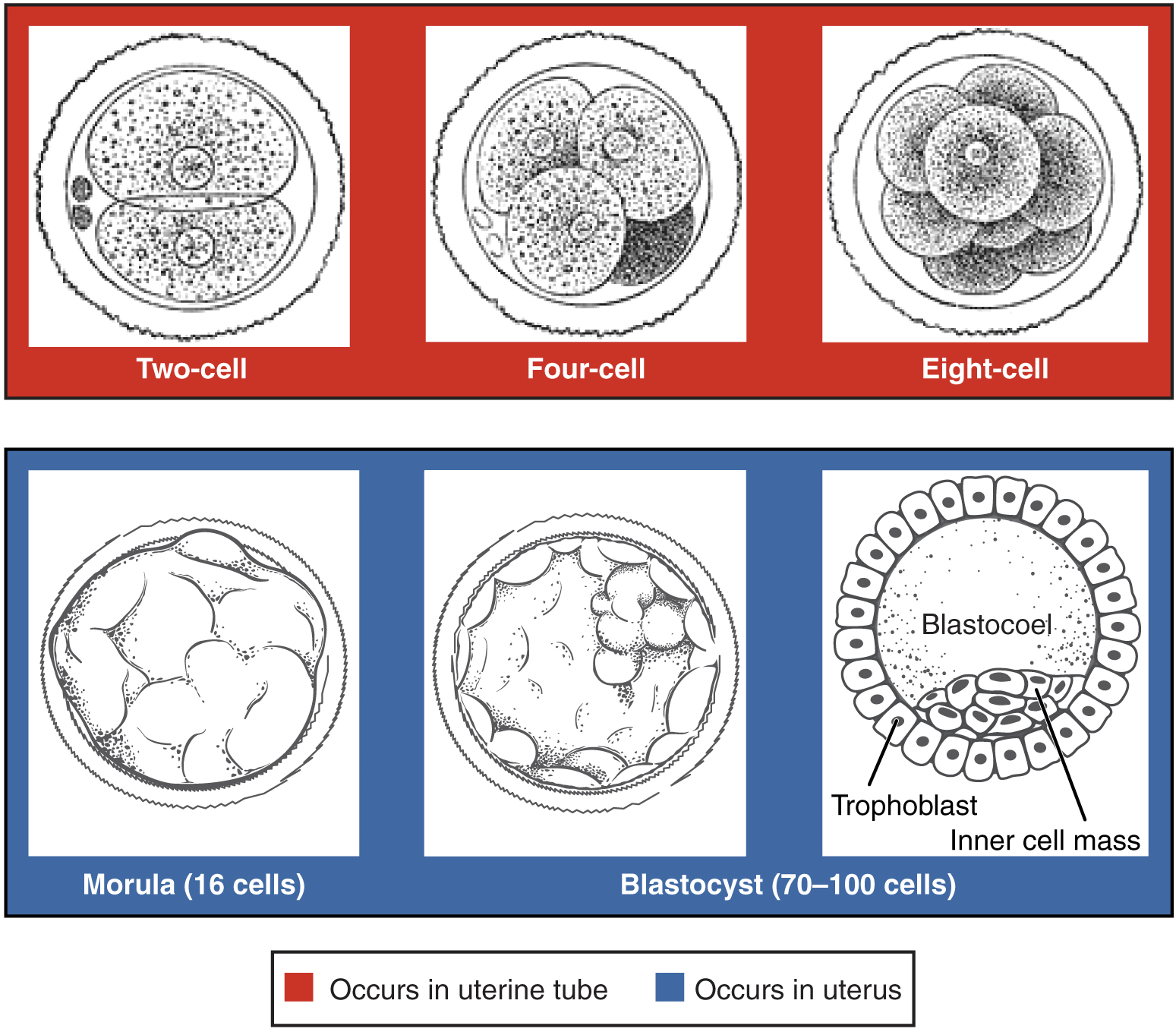
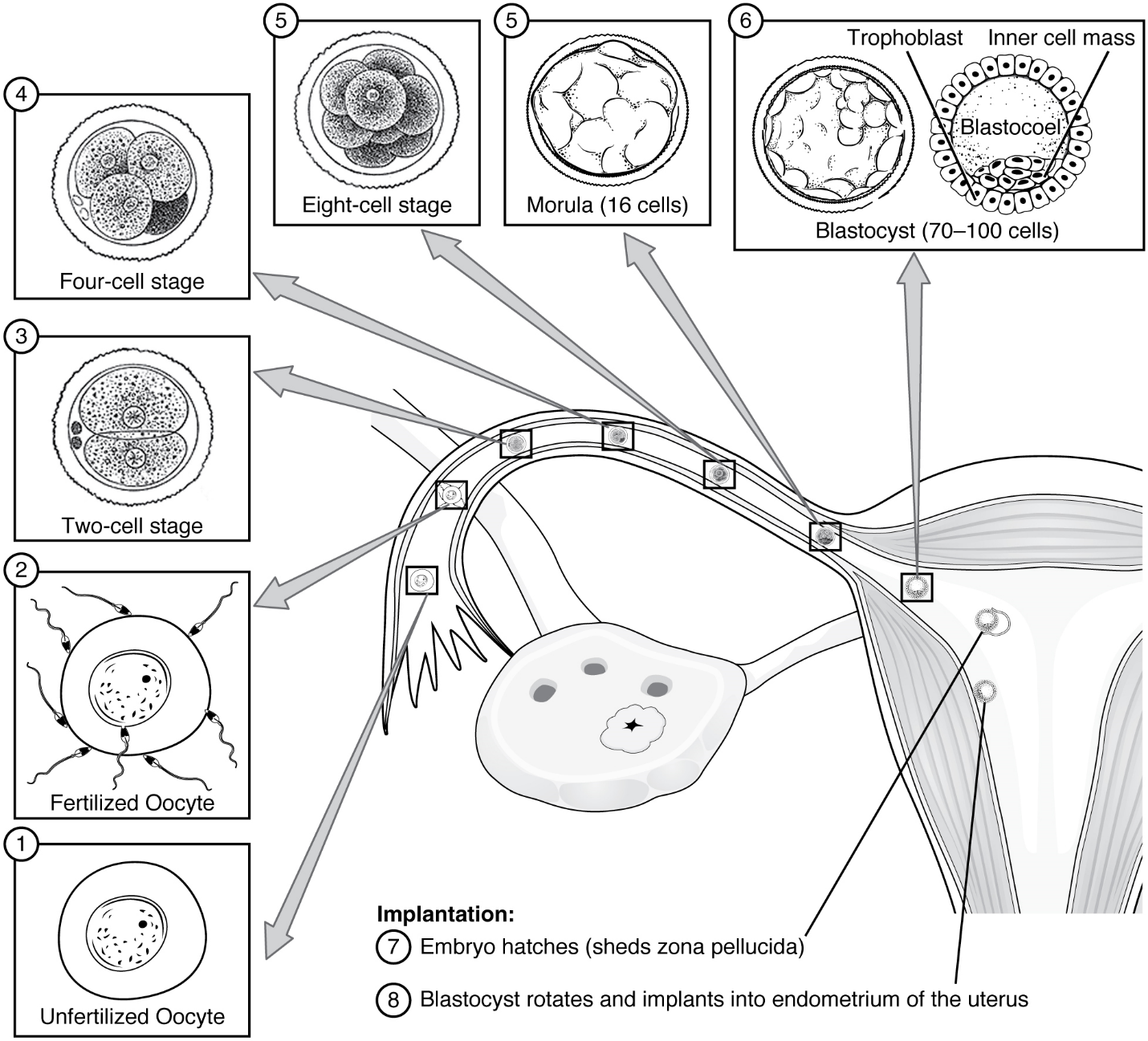
Pre-implantation Embryonic Development: Following fertilization, the zygote and its associated membranes, together referred to as the conceptus, continue to be projected toward the uterus by peristalsis and beating cilia. During its journey to the uterus, the zygote undergoes five or six rapid mitotic cell divisions. Although each cleavage results in more cells, it does not increase the total volume of the conceptus (Figure 12). Each daughter cell produced by cleavage is called a blastomere (blastos = “germ,” in the sense of a seed or sprout).
Approximately 3 days after fertilization, a 16-cell conceptus reaches the uterus. The cells that had been loosely grouped are now compacted and look more like a solid mass. The name given to this structure is the morula (morula = “little mulberry”). Once inside the uterus, the conceptus floats freely for several more days. It continues to divide, creating a ball of approximately 100 cells, and consuming nutritive endometrial secretions called uterine milk while the uterine lining thickens. The ball of now tightly bound cells starts to secrete fluid and organize themselves around a fluid-filled cavity, the blastocoel. At this developmental stage, the conceptus is referred to as a blastocyst. Within this structure, a group of cells forms into an inner cell mass, which is fated to become the embryo. The cells that form the outer shell are called trophoblasts (trophe = “to feed” or “to nourish”). These cells will develop into the chorionic sac and the fetal portion of the placenta (the organ of nutrient, waste, and gas exchange between mother and the developing offspring).
The inner mass of embryonic cells is totipotent during this stage, meaning that each cell has the potential to differentiate into any cell type in the human body. Totipotency lasts for only a few days before the cells’ fates are set as being the precursors to a specific lineage of cells.
As the blastocyst forms, the trophoblast excretes enzymes that begin to degrade the zona pellucida. In a process called “hatching,” the conceptus breaks free of the zona pellucida in preparation for implantation.
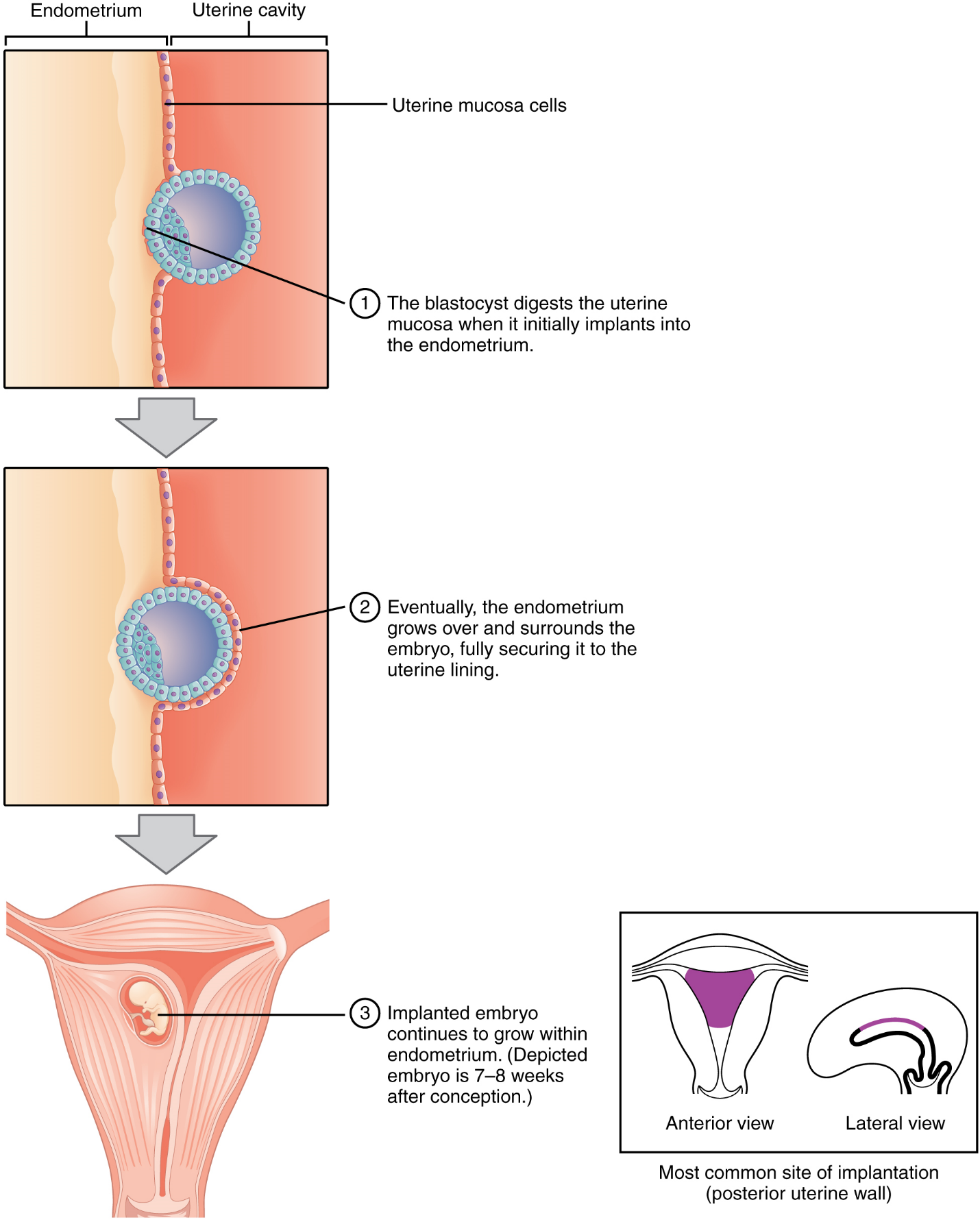
Implantation: At the end of the first week, the blastocyst comes in contact with the uterine wall and adheres to it, embedding itself in the uterine lining via the trophoblast cells. Thus begins the process of implantation, which signals the end of the embryonic stage of development (Figure 13). Implantation can be accompanied by minor bleeding. The blastocyst typically implants in the fundus of the uterus or on the posterior wall. However, if the endometrium is not fully developed and ready to receive the blastocyst, the blastocyst will detach and find a better spot. A significant percentage (50–75 percent) of blastocysts fail to implant; when this occurs, the blastocyst is shed with the endometrium during menses. The high rate of implantation failure is one reason why pregnancy typically requires several ovulation cycles to be achieved.
When implantation succeeds and the blastocyst adheres to the endometrium, the superficial cells of the trophoblast fuse with each other, forming the syncytiotrophoblast, a multinucleated body that digests endometrial cells to firmly secure the blastocyst to the uterine wall. In response, the uterine mucosa rebuilds itself and envelops the blastocyst (Figure 14). The trophoblast secretes human chorionic gonadotropin (hCG), a hormone that directs the corpus luteum to survive, enlarge, and continue producing progesterone and estrogen to suppress menses. These functions of hCG are necessary for creating an environment suitable for the developing embryo. As a result of this increased production, hCG accumulates in the maternal bloodstream and is excreted in the urine. Implantation is complete by the middle of the second week. Just a few days after implantation, the trophoblast has secreted enough hCG for an at-home urine pregnancy test to give a positive result.
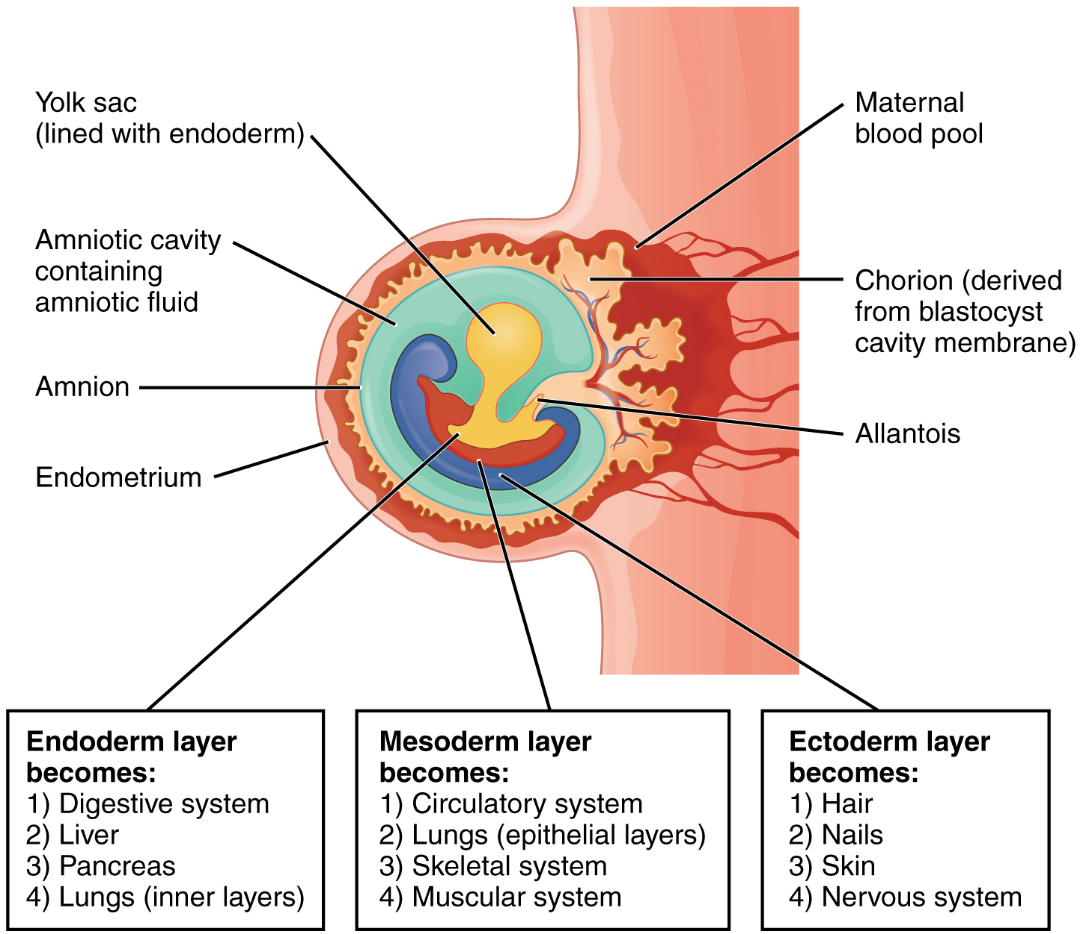
Embryonic Membranes: During the second week of development, with the embryo implanted in the uterus, cells within the blastocyst start to organize into layers. Some grow to form the extra-embryonic membranes needed to support and protect the growing embryo: the amnion, the yolk sac, the allantois, and the chorion (Figure 16). The amnion fills with amniotic fluid and eventually grows to surround the embryo. The yolk sac supplies some nutrients absorbed from the trophoblast and also provides primitive blood circulation to the developing embryo for the second and third week of development. During week 3, a finger-like outpocketing of the yolk sac develops into the allantois, a primitive excretory duct of the embryo that will become part of the urinary bladder. Together, the stalks of the yolk sac and allantois establish the outer structure of the umbilical cord. The last of the extra-embryonic membranes is the chorion, which is the one membrane that surrounds all others. The development of the chorion will be discussed in more detail shortly, as it relates to the growth and development of the placenta.
Germ Layers: As the third week of development begins, the two-layered disc of cells becomes a three-layered disc through the process of gastrulation, during which the cells transition from an undifferentiated to a more differentiated state. Three groups of cells, called germ layers, are formed: the endoderm, mesoderm (middle), and ectoderm.
Each of these germ layers will develop into specific structures in the embryo. Whereas the ectoderm and endoderm form tightly connected epithelial sheets, the mesodermal cells are less organized and exist as a loosely connected cell community. The ectoderm gives rise to cell lineages that differentiate to become the central and peripheral nervous systems, sensory organs, epidermis, hair, and nails. Mesodermal cells ultimately become the skeleton, muscles, connective tissue, heart, blood vessels, and kidneys. The endoderm goes on to form the epithelial lining of the gastrointestinal tract, liver, and pancreas, as well as the lungs (Figure 15).
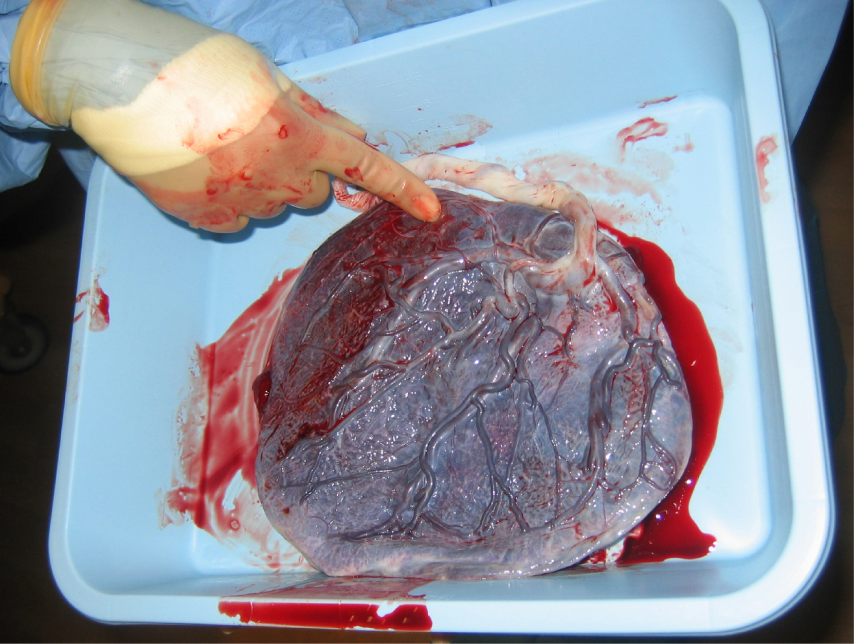
Development of the Placenta: During the first several weeks of development, the cells of the endometrium—referred to as decidual cells—nourish the nascent embryo. During prenatal weeks 4–12, the developing placenta gradually takes over the role of feeding the embryo, and the decidual cells are no longer needed. The mature placenta is composed of tissues derived from the embryo, as well as maternal tissues of the endometrium. The placenta connects to the conceptus via the umbilical cord, which carries deoxygenated blood and wastes from the fetus through two umbilical arteries; nutrients and oxygen are carried from the mother to the fetus through the single umbilical vein.
The maternal portion of the placenta develops from the deepest layer of the endometrium, the decidua basalis. To form the embryonic portion of the placenta, the syncytiotrophoblast and the underlying cells of the trophoblast (cytotrophoblast cells) begin to proliferate along with a layer of extraembryonic mesoderm cells. These form the chorionic membrane, which envelops the entire conceptus as the chorion. The chorionic membrane forms finger-like structures called chorionic villi that burrow into the endometrium like tree roots, making up the fetal portion of the placenta. The cytotrophoblast cells perforate the chorionic villi, burrow farther into the endometrium, and remodel maternal blood vessels to augment maternal blood flow surrounding the villi. Meanwhile, fetal mesenchymal cells derived from the mesoderm fill the villi and differentiate into blood vessels, including the three umbilical blood vessels that connect the embryo to the developing placenta (Figure 16).
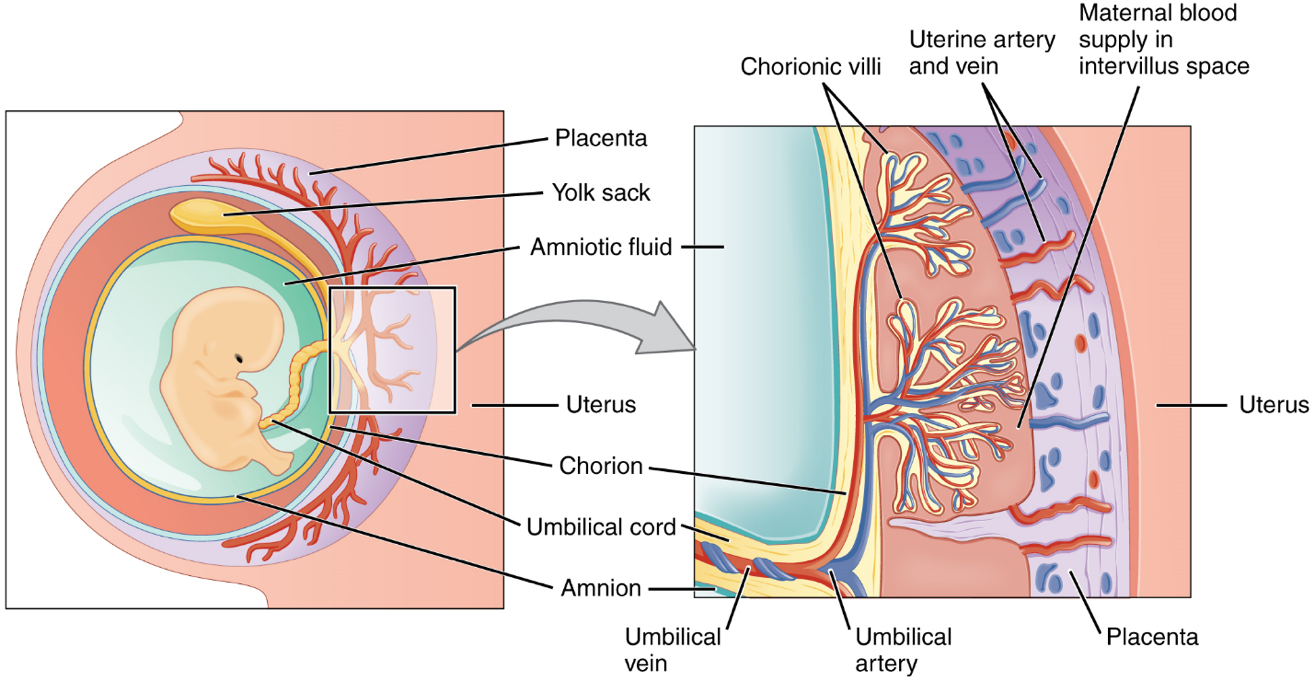
The placenta develops throughout the embryonic period and during the first several weeks of the fetal period; placentation is complete by weeks 14–16. As a fully developed organ, the placenta provides nutrition and excretion, respiration, and endocrine function (Table 1 and Figure 17). It receives blood from the fetus through the umbilical arteries. Capillaries in the chorionic villi filter fetal wastes out of the blood and return clean, oxygenated blood to the fetus through the umbilical vein. Nutrients and oxygen are transferred from maternal blood surrounding the villi through the capillaries and into the fetal bloodstream.
Maternal and fetal blood do not mingle because blood cells cannot move across the placenta. This separation prevents the mother’s cytotoxic T cells from reaching and subsequently destroying the fetus, which bears “non-self” antigens.
| Nutrition & digestion | Respiration | Endocrine function |
|---|---|---|
| Mediates diffusion of maternal glucose, amino acids, fatty acids, vitamins, minerals
Stores nutrients during early pregnancy to accommodate increased fetal demand later in pregnancy Excretes & filters fetal nitrogenous wastes into maternal blood |
Mediates maternal-to-fetal oxygen transport
Mediates fetal-to-maternal carbon dioxide transport |
Secretes hCG, estrogens, and progesterone to maintain the pregnancy and stimulate maternal & fetal development
Mediates transmission of maternal hormones into fetal blood Mediates transmission of fetal hormones into maternal blood |
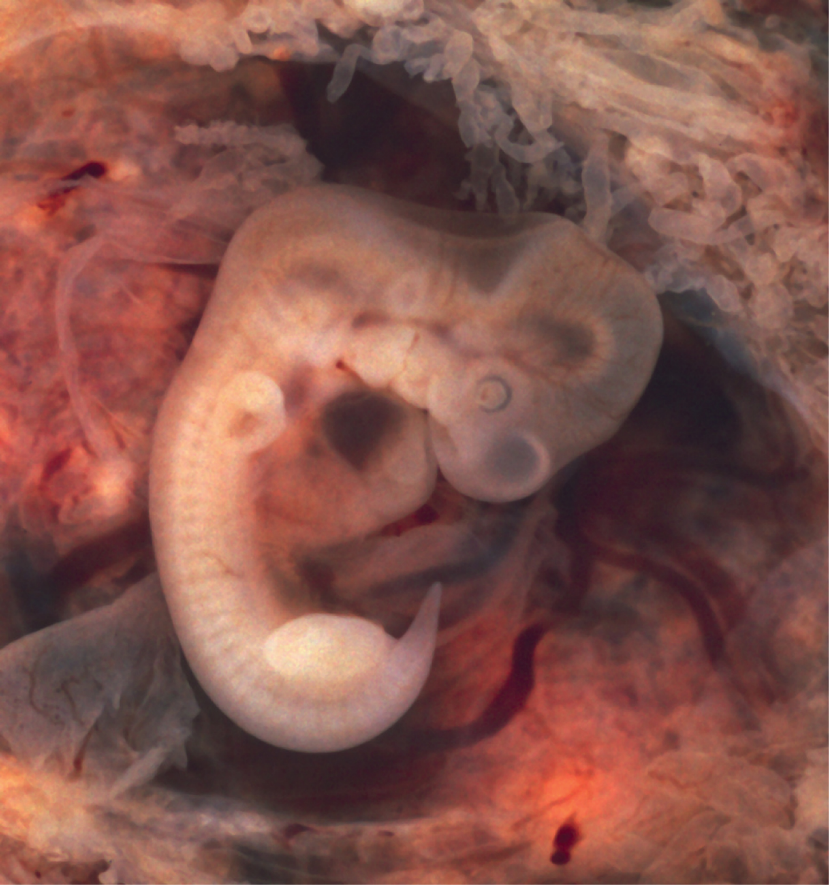
During the sixth week, uncontrolled fetal limb movements begin to occur. The gastrointestinal system develops too rapidly for the embryonic abdomen to accommodate it, and the intestines temporarily loop into the umbilical cord. Paddle-shaped hands and feet develop fingers and toes by the process of apoptosis (programmed cell death), which causes the tissues between the fingers to disintegrate. By week 7, the facial structure is more complex and includes nostrils, outer ears, and lenses (Figure 18). By the eighth week, the head is nearly as large as the rest of the embryo’s body, and all major brain structures are in place. The external genitalia are apparent, but at this point, male and female embryos are indistinguishable. Bone begins to replace cartilage in the embryonic skeleton through the process of ossification. By the end of the embryonic period, the embryo is approximately 3 cm (1.2 in) from crown to rump and weighs approximately 8 g (0.25 oz).

Part 5: Fetal Development
A developing human is called a fetus from the ninth week of gestation until birth. This 30-week period of development is marked by continued cell growth and differentiation, which fully develop the structures and functions of the immature organ systems formed during the embryonic period. The completion of fetal development results in a newborn who, although still immature in many ways, is capable of survival outside the womb.
The Fetal Circulatory System: During prenatal development, the fetal circulatory system is integrated with the placenta via the umbilical cord so that the fetus receives both oxygen and nutrients from the placenta. However, after childbirth, the umbilical cord is severed, and the newborn’s circulatory system must be reconfigured. When the heart first forms in the embryo, it exists as two parallel tubes derived from mesoderm and lined with endothelium, which then fuse together. As the embryo develops into a fetus, the tube-shaped heart folds and further differentiates into the four chambers present in a mature heart. Unlike a mature cardiovascular system, however, the fetal cardiovascular system also includes circulatory shortcuts, or shunts. A shunt is an anatomical (or sometimes surgical) diversion that allows blood flow to bypass immature organs such as the lungs and liver until childbirth.
The placenta provides the fetus with necessary oxygen and nutrients via the umbilical vein. (Remember that veins carry blood toward the heart. In this case, the blood flowing to the fetal heart is oxygenated because it comes from the placenta. The respiratory system is immature and cannot yet oxygenate blood on its own.)
From the umbilical vein, the oxygenated blood flows toward the inferior vena cava, all but bypassing the immature liver, via the ductus venosus shunt (Figure 19). The liver receives just a trickle of blood, which is all that it needs in its immature, semifunctional state. Blood flows from the inferior vena cava to the right atrium, mixing with fetal venous blood along the way.
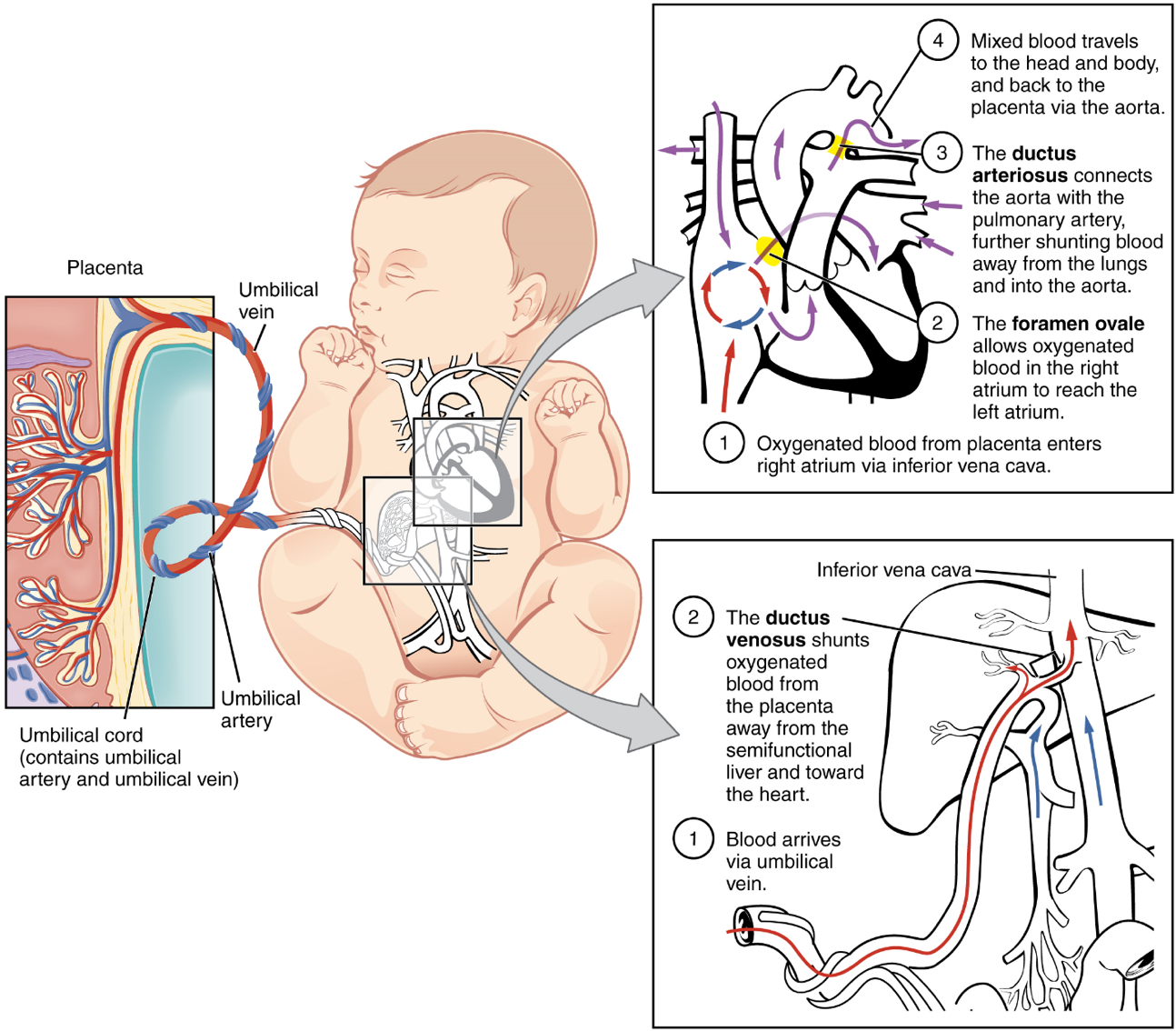
Although the fetal liver is semifunctional, the fetal lungs are nonfunctional. The fetal circulation therefore bypasses the lungs by shifting some of the blood through the foramen ovale, a shunt that directly connects the right and left atria and avoids the pulmonary trunk altogether. Most of the rest of the blood is pumped to the right ventricle, and from there, into the pulmonary trunk, which splits into pulmonary arteries. However, a shunt within the pulmonary artery, the ductus arteriosus, diverts a portion of this blood into the aorta. This ensures that only a small volume of oxygenated blood passes through the immature pulmonary circuit, which has only minor metabolic requirements. Blood vessels of uninflated lungs have high resistance to flow, a condition that encourages blood to flow to the aorta, which presents much lower resistance. The oxygenated blood moves through the foramen ovale into the left atrium, where it mixes with the now deoxygenated blood returning from the pulmonary circuit. This blood then moves into the left ventricle, where it is pumped into the aorta. Some of this blood moves through the coronary arteries into the myocardium, and some moves through the carotid arteries to the brain.
The descending aorta carries partially oxygenated and partially deoxygenated blood into the lower regions of the body. It eventually passes into the umbilical arteries through branches of the internal iliac arteries. The deoxygenated blood collects waste as it circulates through the fetal body and returns to the umbilical cord. Thus, the two umbilical arteries carry blood low in oxygen and high in carbon dioxide and fetal wastes. This blood is filtered through the placenta, where wastes diffuse into the maternal circulation. Oxygen and nutrients from the mother diffuse into the placenta and from there into the fetal blood, and the process repeats.
Prior to birth, the lungs are filled with amniotic fluid, mucus, and surfactant. As the fetus is squeezed through the birth canal, the fetal thoracic cavity is compressed, expelling much of this fluid. Some fluid remains, however, but is rapidly absorbed by the body shortly after birth. The first inhalation occurs within 10 seconds after birth and not only serves as the first inspiration, but also acts to inflate the lungs. Pulmonary surfactant is critical for inflation to occur, as it reduces the surface tension of the alveoli. Preterm birth around 26 weeks frequently results in severe respiratory distress, although with current medical advancements, some babies may survive. Prior to 26 weeks, sufficient pulmonary surfactant is not produced, and the surfaces for gas exchange have not formed adequately; therefore, survival is low.
During approximately weeks 16–20, as the fetus grows and limb movements become more powerful, the mother may begin to feel quickening, or fetal movements. However, space restrictions limit these movements and typically force the growing fetus into the “fetal position,” with the arms crossed and the legs bent at the knees. Sebaceous glands coat the skin with a waxy, protective substance called vernix caseosa that protects and moisturizes the skin and may provide lubrication during childbirth. A silky hair called lanugo also covers the skin during weeks 17–20, but it is shed as the fetus continues to grow.
Developmental weeks 21–30 are characterized by rapid weight gain, which is important for maintaining a stable body temperature after birth. The bone marrow completely takes over erythrocyte synthesis, and the axons of the spinal cord begin to be myelinated, or coated in the electrically insulating glial cell sheaths that are necessary for efficient nervous system functioning. (The process of myelination is not completed until adolescence.) During this period, the fetus grows eyelashes. The eyelids are no longer fused and can be opened and closed. The lungs begin producing surfactant, a substance that reduces surface tension in the lungs and assists proper lung expansion after birth. Inadequate surfactant production in premature newborns may result in respiratory distress syndrome, and as a result, the newborn may require surfactant replacement therapy, supplemental oxygen, or maintenance in a continuous positive airway pressure (CPAP) chamber during their first days or weeks of life. In male fetuses, the testes descend into the scrotum near the end of this period. The fetus at 30 weeks measures 28 cm (11 in) from crown to rump and exhibits the approximate body proportions of a full-term newborn, but still is much leaner.
The fetus continues to lay down subcutaneous fat from week 31 until birth. The added fat fills out the hypodermis, and the skin transitions from red and wrinkled to soft and pink. Lanugo is shed, and the nails grow to the tips of the fingers and toes. Immediately before birth, the average crown-to-rump length is 35.5–40.5 cm (14–16 in), and the fetus weighs approximately 2.5–4 kg (5.5–8.8 lbs). Once born, the newborn is no longer confined to the fetal position, so subsequent measurements are made from head-to-toe instead of from crown-to-rump. At birth, the average length is approximately 51 cm (20 in).
Part 6: Maternal Changes During Pregnancy, Labor, and Birth
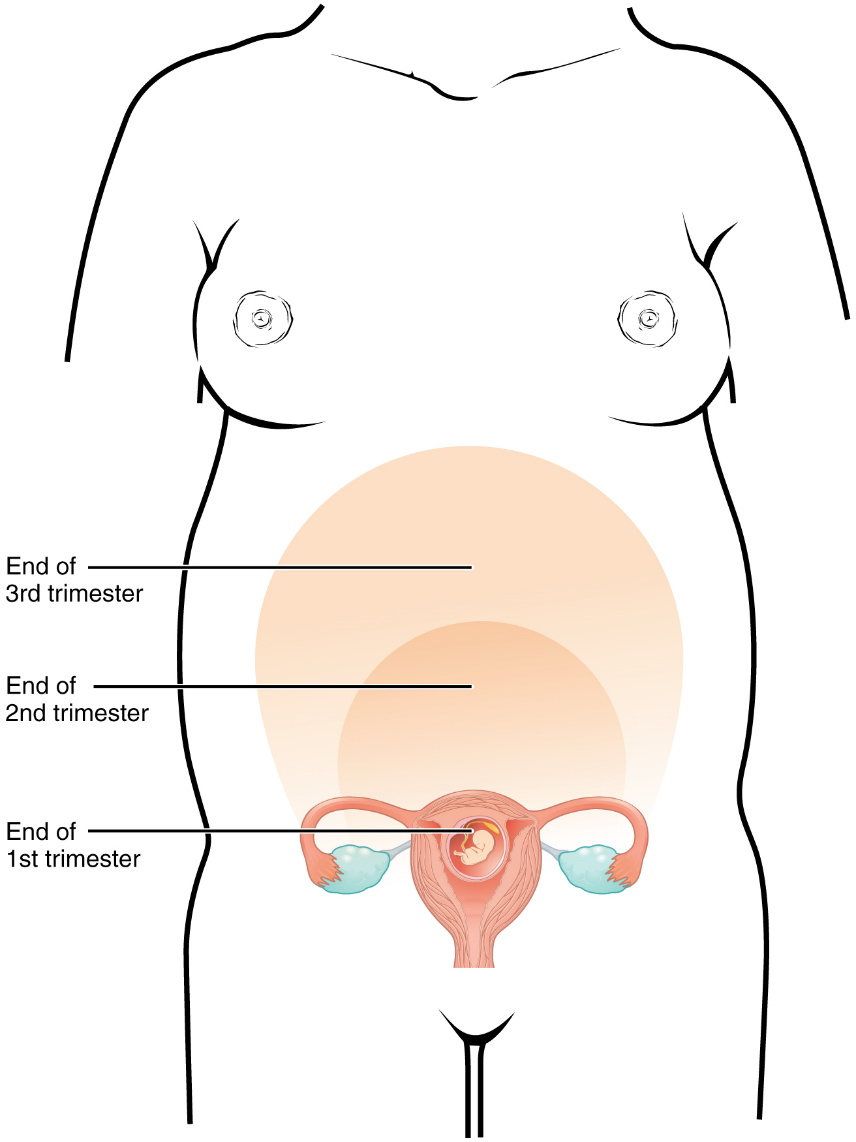
A full-term pregnancy lasts approximately 270 days (approximately 38.5 weeks) from conception to birth. Because it is easier to remember the first day of the last menstrual period (LMP) than to estimate the date of conception, obstetricians set the due date as 284 days (approximately 40.5 weeks) from the LMP. This assumes that conception occurred on day 14 of the woman’s cycle, which is usually a good approximation. The 40 weeks of an average pregnancy are usually discussed in terms of three trimesters, each approximately 13 weeks. During the second and third trimesters, the pre-pregnancy uterus—about the size of a fist—grows dramatically to contain the fetus, causing a number of anatomical changes in the mother (Figure 20).
Effects of Hormones: Virtually all the effects of pregnancy can be attributed in some way to the influence of hormones—particularly estrogens, progesterone, and hCG. During weeks 7–12 from the LMP, the pregnancy hormones are primarily generated by the corpus luteum. Progesterone secreted by the corpus luteum stimulates the production of decidual cells of the endometrium that nourish the blastocyst before placentation. As the placenta develops and the corpus luteum degenerates during weeks 12–17, the placenta gradually takes over as the endocrine organ of pregnancy.
The placenta converts weak androgens secreted by the maternal and fetal adrenal glands to estrogens, which are necessary for pregnancy to progress. Estrogen levels climb throughout the pregnancy, increasing 30-fold by childbirth. Estrogens have the following actions:
- They suppress FSH and LH production, effectively preventing ovulation. (This function is the biological basis of hormonal birth control pills.)
- They induce the growth of fetal tissues and are necessary for the maturation of the fetal lungs and liver.
- They promote fetal viability by regulating progesterone production and triggering fetal synthesis of cortisol, which helps with the maturation of the lungs, liver, and endocrine organs such as the thyroid gland and adrenal gland.
- They stimulate maternal tissue growth, leading to uterine enlargement and mammary duct expansion and branching.
Relaxin, another hormone secreted by the corpus luteum and then by the placenta, helps prepare the mother’s body for childbirth. It increases the elasticity of the symphysis pubis joint and pelvic ligaments, making room for the growing fetus and allowing expansion of the pelvic outlet for childbirth. Relaxin also helps dilate the cervix during labor.
The placenta takes over the synthesis and secretion of progesterone throughout pregnancy as the corpus luteum degenerates. Like estrogen, progesterone suppresses FSH and LH. It also inhibits uterine contractions, protecting the fetus from preterm birth. This hormone decreases in late gestation, allowing uterine contractions to intensify and eventually progress to true labor. The placenta also produces hCG. In addition to promoting survival of the corpus luteum, hCG stimulates the male fetal gonads to secrete testosterone, which is essential for the development of the male reproductive system.
Physiology of Labor: Childbirth, or parturition, typically occurs within a week of a woman’s due date, unless the woman is pregnant with more than one fetus, which usually causes her to go into labor early. As a pregnancy progresses into its final weeks, several physiological changes occur in response to hormones that trigger labor.
First, recall that progesterone inhibits uterine contractions throughout the first several months of pregnancy. As the pregnancy enters its seventh month, progesterone levels plateau and then drop. Estrogen levels, however, continue to rise in the maternal circulation (Figure 21). The increasing ratio of estrogen to progesterone makes the myometrium (the uterine smooth muscle) more sensitive to stimuli that promote contractions (because progesterone no longer inhibits them). Moreover, in the eighth month of pregnancy, fetal cortisol rises, which boosts estrogen secretion by the placenta and further overpowers the uterine-calming effects of progesterone.
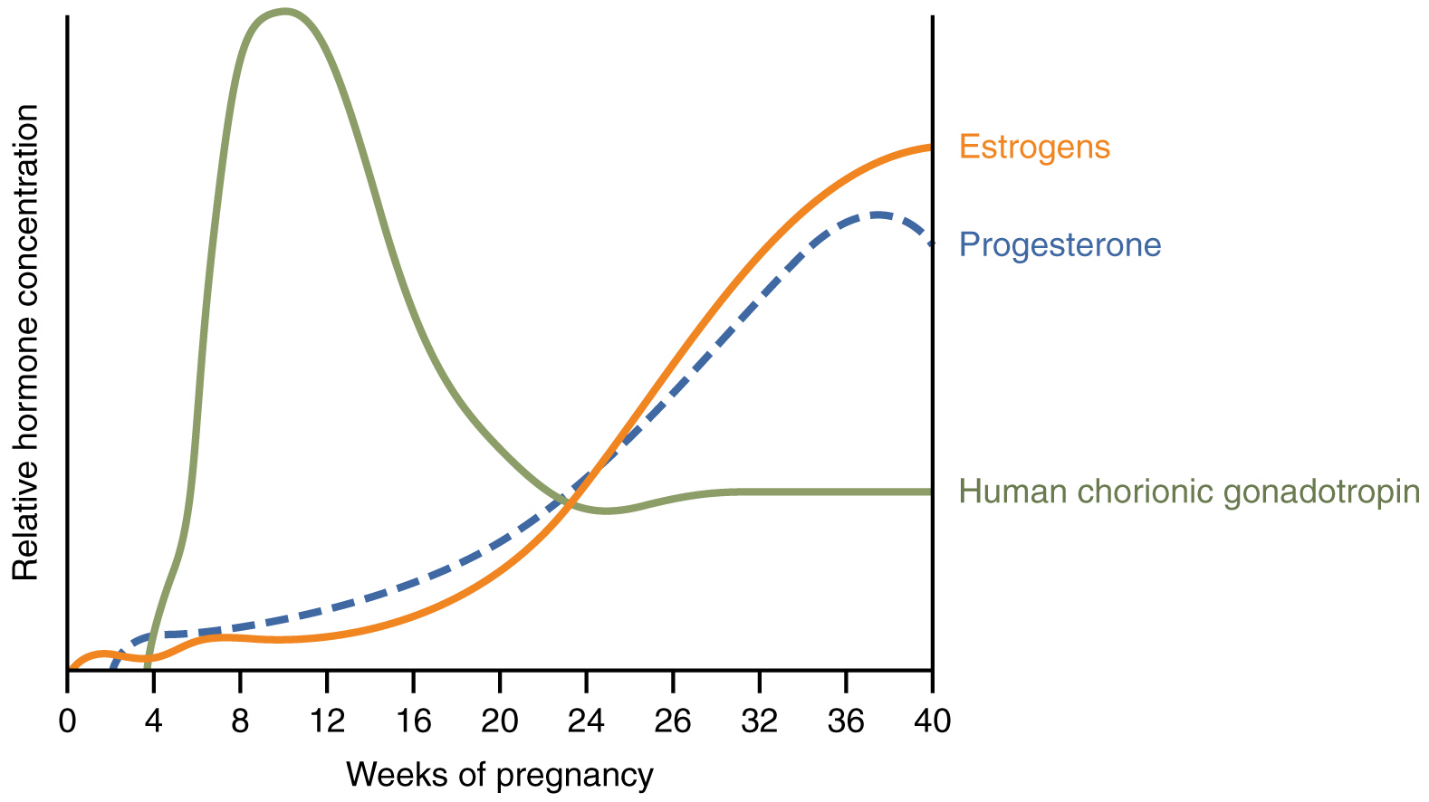
Meanwhile, the posterior pituitary has been boosting its secretion of oxytocin, a hormone that stimulates the contractions of labor. At the same time, the myometrium increases its sensitivity to oxytocin by expressing more receptors for this hormone. As labor nears, oxytocin begins to stimulate stronger, more painful uterine contractions, which—in a positive feedback loop—stimulate the secretion of prostaglandins from fetal membranes. Like oxytocin, prostaglandins also enhance uterine contractile strength.
Finally, stretching of the myometrium and cervix by a full-term fetus in the vertex (head-down) position is regarded as a stimulant to uterine contractions. The sum of these changes initiates the regular contractions known as true labor, which become more powerful and more frequent with time. The pain of labor is attributed to myometrial hypoxia during uterine contractions.
Stages of Childbirth: The process of childbirth can be divided into three stages: cervical dilation, expulsion of the newborn ending with birth, and afterbirth (Figure 22).
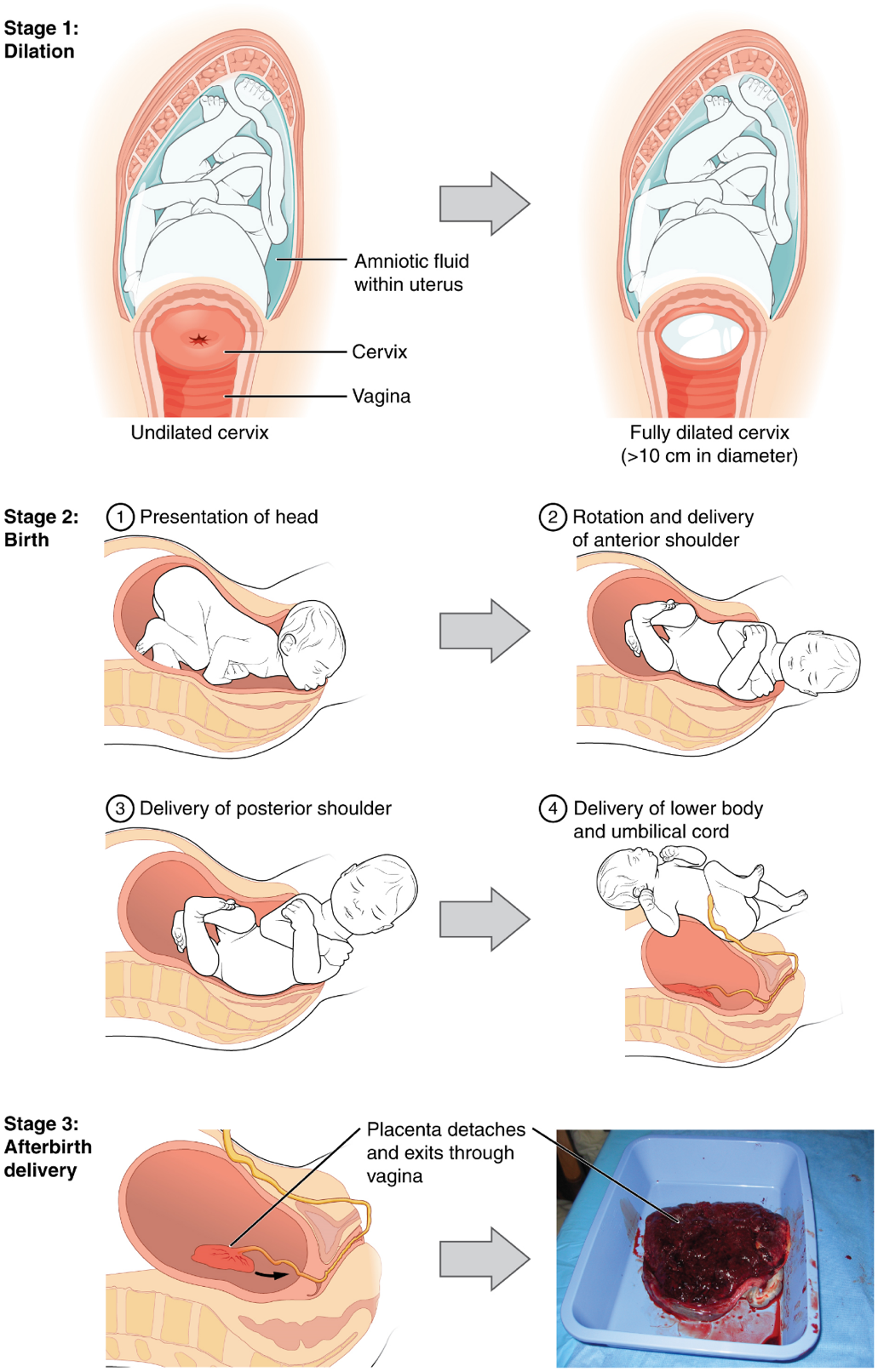
1. Cervical Dilation: For vaginal birth to occur, the cervix must dilate fully to 10 cm in diameter—wide enough to deliver the newborn’s head. The dilation stage is the longest stage of labor and typically takes 6–12 hours. However, it varies widely and may take minutes, hours, or days, depending in part on whether the mother has given birth before; in each subsequent labor, this stage tends to be shorter.
True labor progresses in a positive feedback loop in which uterine contractions stretch the cervix, causing it to dilate and efface, or become thinner. Cervical stretching induces reflexive uterine contractions that dilate and efface the cervix further. In addition, cervical dilation boosts oxytocin secretion from the pituitary, which in turn triggers more powerful uterine contractions. When labor begins, uterine contractions may occur only every 3–30 minutes and last only 20–40 seconds; however, by the end of this stage, contractions may occur as frequently as every 1.5–2 minutes and last for a full minute.
Each contraction sharply reduces oxygenated blood flow to the fetus. For this reason, it is critical that a period of relaxation occur after each contraction. Fetal distress, measured as a sustained decrease or increase in the fetal heart rate, can result from severe contractions that are too powerful or lengthy for oxygenated blood to be restored to the fetus. Such a situation can be cause for an emergency birth with vacuum, forceps, or surgically by Caesarian section.
2. Expulsion Stage: The expulsion stage begins when the fetal head enters the birth canal and ends with birth of the newborn. It typically takes up to 2 hours, but it can last longer or be completed in minutes, depending in part on the orientation of the fetus. The most common presentation and associated with the greatest ease of vaginal birth is when the fetus faces the maternal spinal cord and the smallest part of the head (the posterior aspect called the occiput) exits the birth canal first.
Upon birth of the newborn’s head, an obstetrician will aspirate mucus from the mouth and nose before the newborn’s first breath. Once the head is birthed, the rest of the body usually follows quickly. The umbilical cord is then double-clamped, and a cut is made between the clamps. This completes the second stage of childbirth.
3. Afterbirth: The delivery of the placenta and associated membranes, commonly referred to as the afterbirth, marks the final stage of childbirth. After expulsion of the newborn, the myometrium continues to contract. This movement shears the placenta from the back of the uterine wall. It is then easily delivered through the vagina. Continued uterine contractions then reduce blood loss from the site of the placenta. Delivery of the placenta marks the beginning of the postpartum period—the period of approximately 6 weeks immediately following childbirth during which the mother’s body gradually returns to a non-pregnant state. If the placenta does not birth spontaneously within approximately 30 minutes, it is considered retained, and the obstetrician may attempt manual removal.
Part 7: Adjustments of the Infant at Birth and Postnatal Stages
From a fetal perspective, the process of birth is a crisis. In the womb, the fetus was snuggled in a soft, warm, dark, and quiet world. The placenta provided nutrition and oxygen continuously. Suddenly, the contractions of labor and vaginal childbirth forcibly squeeze the fetus through the birth canal, limiting oxygenated blood flow during contractions and shifting the skull bones to accommodate the small space. After birth, the newborn’s system must make drastic adjustments to a world that is colder, brighter, and louder, and where he or she will experience hunger and thirst. The neonatal period (neo- = “new”; -natal = “birth”) spans the first to the thirtieth day of life outside of the uterus.
Respiratory Adjustments: Although the fetus “practices” breathing by inhaling amniotic fluid in utero, there is no air in the uterus and thus no true opportunity to breathe. (There is also no need to breathe because the placenta supplies the fetus with all the oxygenated blood it needs.) During gestation, the partially collapsed lungs are filled with amniotic fluid and exhibit very little metabolic activity. Several factors stimulate newborns to take their first breath at birth. First, labor contractions temporarily constrict umbilical blood vessels, reducing oxygenated blood flow to the fetus and elevating carbon dioxide levels in the blood. High carbon dioxide levels cause acidosis and stimulate the respiratory center in the brain, triggering the newborn to take a breath.
The first breath typically is taken within 10 seconds of birth, after mucus is aspirated from the infant’s mouth and nose. The first breaths inflate the lungs to nearly full capacity and dramatically decrease lung pressure and resistance to blood flow, causing a major circulatory reconfiguration. Pulmonary alveoli open, and alveolar capillaries fill with blood. Amniotic fluid in the lungs drains or is absorbed, and the lungs immediately take over the task of the placenta, exchanging carbon dioxide for oxygen by the process of respiration.
Circulatory Adjustments
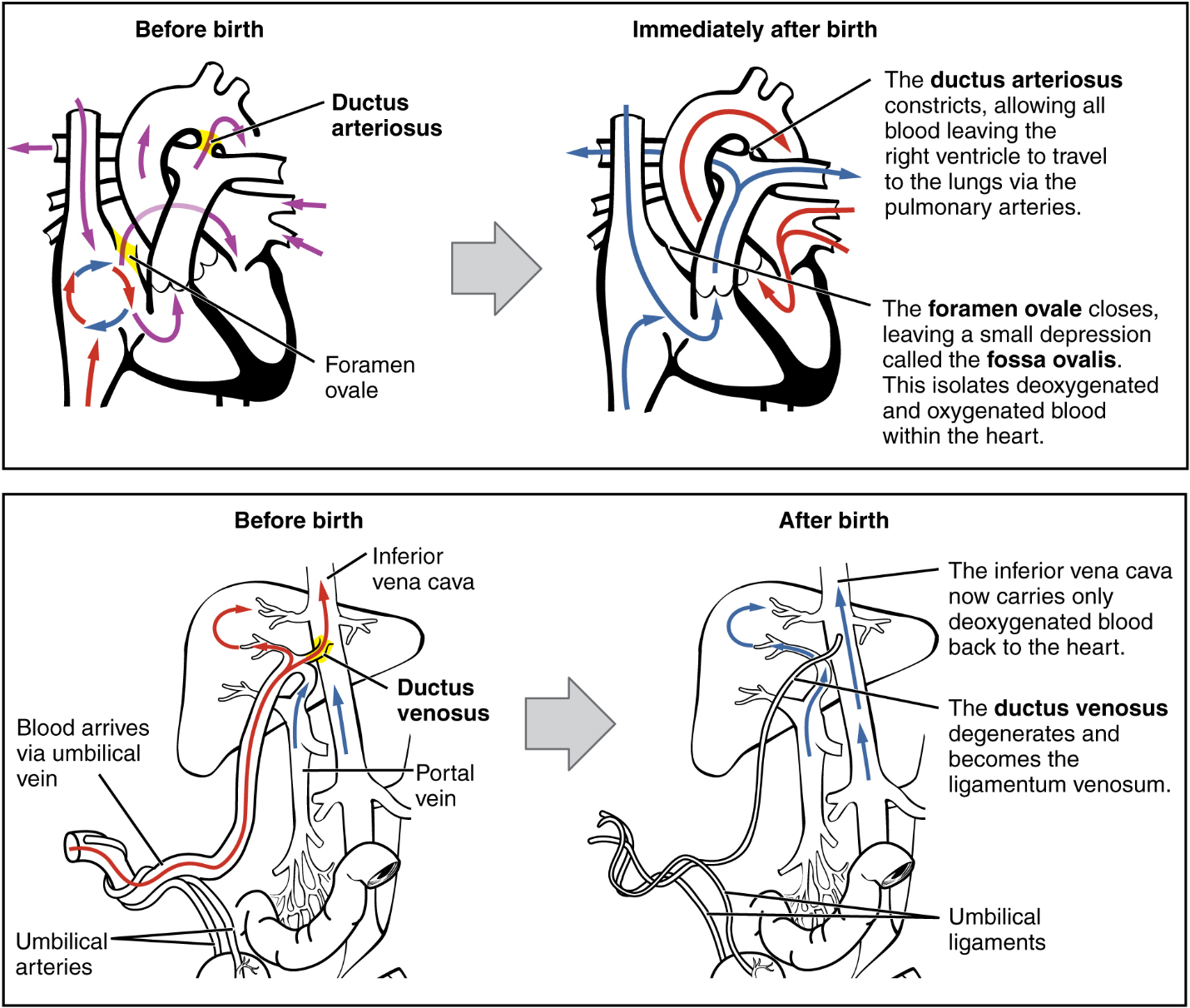
The newborn’s first breath is vital to initiate the transition from the fetal to the neonatal circulatory pattern. Inflation of the lungs decreases blood pressure throughout the pulmonary system, as well as in the right atrium and ventricle. In response to this pressure change, the flow of blood temporarily reverses direction through the foramen ovale, moving from the left to the right atrium, and blocking the shunt with two flaps of tissue. Within 1 year, the tissue flaps usually fuse over the shunt, turning the foramen ovale into the fossa ovalis (Figure 23). The ductus arteriosus constricts as a result of increased oxygen concentration and becomes the ligamentum arteriosum (Figure 23). Closing of the ductus arteriosus ensures that all blood pumped to the pulmonary circuit will be oxygenated by the newly functional neonatal lungs. The ductus venosus degenerates to become the ligamentum venosum beneath the liver.
Part 8: Lactation
Lactation is the process by which milk is synthesized and secreted from the mammary glands of the postpartum female breast in response to an infant sucking at the nipple. Breast milk provides ideal nutrition and passive immunity for the infant, encourages mild uterine contractions to return the uterus to its pre-pregnancy size (i.e., involution), and induces a substantial metabolic increase in the mother, consuming the fat reserves stored during pregnancy.
Structure of the Lactating Breast: Mammary glands are modified sweat glands. The non-pregnant and non-lactating female breast is composed primarily of adipose and collagenous tissue, with mammary glands making up a very minor proportion of breast volume. The mammary gland is composed of milk-transporting lactiferous ducts, which expand and branch extensively during pregnancy in response to estrogen, growth hormone, cortisol, and prolactin. Moreover, in response to progesterone, clusters of breast alveoli bud from the ducts and expand outward toward the chest wall. Breast alveoli are balloon-like structures lined with milk-secreting cuboidal cells, or lactocytes, that are surrounded by a net of contractile myoepithelial cells. Milk is secreted from the lactocytes, fills the alveoli, and is squeezed into the ducts. Clusters of alveoli that drain to a common duct are called lobules; the lactating female has 12–20 lobules organized radially around the nipple. Milk drains from lactiferous ducts into lactiferous sinuses that meet at 4 to 18 perforations in the nipple, called nipple pores.
The Process of Lactation: The pituitary hormone prolactin is instrumental in the establishment and maintenance of breast milk supply. It also is important for the mobilization of maternal micronutrients for breast milk.
Near the fifth week of pregnancy, the level of circulating prolactin begins to increase, eventually rising to approximately 10–20 times the pre-pregnancy concentration. During pregnancy, prolactin and other hormones prepare the breasts anatomically for the secretion of milk. The level of prolactin plateaus in late pregnancy, at a level high enough to initiate milk production. However, estrogen, progesterone, and other placental hormones inhibit prolactin-mediated milk synthesis during pregnancy. It is not until the placenta is expelled that this inhibition is lifted and milk production commences.
After childbirth, the baseline prolactin level drops sharply, but it is restored for a 1-hour spike during each feeding to stimulate the production of milk for the next feeding. With each prolactin spike, estrogen and progesterone also increase slightly.
When the infant suckles, sensory nerve fibers in the areola trigger a neuroendocrine reflex that results in milk secretion from lactocytes into the alveoli. The posterior pituitary releases oxytocin, which stimulates myoepithelial cells to squeeze milk from the alveoli so it can drain into the lactiferous ducts, collect in the lactiferous sinuses, and discharge through the nipple pores. It takes less than 1 minute from the time when an infant begins suckling (the latent period) until milk is secreted via the let-down reflex (Figure 24).
Changes in the Composition of Breast Milk: In the final weeks of pregnancy, the alveoli swell with colostrum, a thick, yellowish substance that is high in protein but contains less fat and glucose than mature breast milk. Colostrum is secreted during the first 48–72 hours postpartum. Only a small volume of colostrum is produced—approximately 3 ounces in a 24-hour period—but it is sufficient for the newborn in the first few days of life. Colostrum is rich with immunoglobulins, which confer gastrointestinal, and also likely systemic, immunity as the newborn adjusts to a nonsterile environment.