Maintenance of the Body
Unit 2: The Cardiovascular System
Unit outline
Blood
Part 1: An Overview of Blood
- Functions of blood
- Composition of blood
- Characteristics of blood
- Blood plasma
Part 2: Production of the Formed Elements
- Sites of hemopoiesis
- Differentiation of formed elements from stem cells
- Hemopoietic growth factors
Part 3: Erythrocytes
- Hemoglobin
Part 4: Leukocytes and Platelets
- Characteristics of leukocytes
- Platelets
- Disorders of platelets
Part 5: Hemostasis
- Vascular Spasm
- Formation of the platelet plug
- Coagulation
- Clotting factors involved in coagulation
- Extrinsic pathway
- Intrinsic pathway
- Common pathway
- Fibrinolysis
- Plasma anticoagulants
- Disorders of clotting
The Heart
Part 1: Heart Anatomy
- Location of the heart
- Chambers and circulation through the heart
- Membranes, surface features, and layers
- Internal structure of the heart
- Heart valve structure and function
- Coronary circulation
Part 2: Cardiac Muscle and Electrical Activity
- Conduction system of the heart
- Electrocardiogram
Part 3: Cardiac Cycle
- Pressure and flow
- Phases of the cardiac cycle
- Heart sounds
- Cardiac output
Part 4: Cardiac Physiology
- Heart rates
- Correlation between heart rates and cardiac output
- Cardiovascular centres
- Other factors influencing heart rate
Blood Vessels and Circulation
Part 1: Structure and function of blood vessels
- Shared structures
- Arteries
- Arterioles
- Capillaries
- Venules
- Veins
Part 2: Blood flow, blood pressure, and resistance
- Components of arterial blood pressure
- Pulse
- Measurement of blood pressure
- Variables affecting blood flow and blood pressure
- Cardiac output
- Compliance
- Blood volume
- Blood viscosity
- Vessel length and diameter
- Venous system
- Skeletal muscle pump
- Respiratory pump
Part 3: Capillary Exchange
Part 4: Hemostatic Regulation of the Vascular System
- Neural regulation
- The cardiovascular centres in the brain
- Baroreceptor reflexes
- Endocrine regulation
- Autoregulation of perfusion
Part 5: Circulatory Pathways
- Pulmonary circulation
- Overview of systemic arteries
- The aorta
- Coronary circulation
- Aortic arch branches
- Thoracic aorta and major branches
- Abdominal aorta and major branches
- Arteries serving the upper and lower limbs
- Overview of systemic veins
- The superior and inferior vena cavae
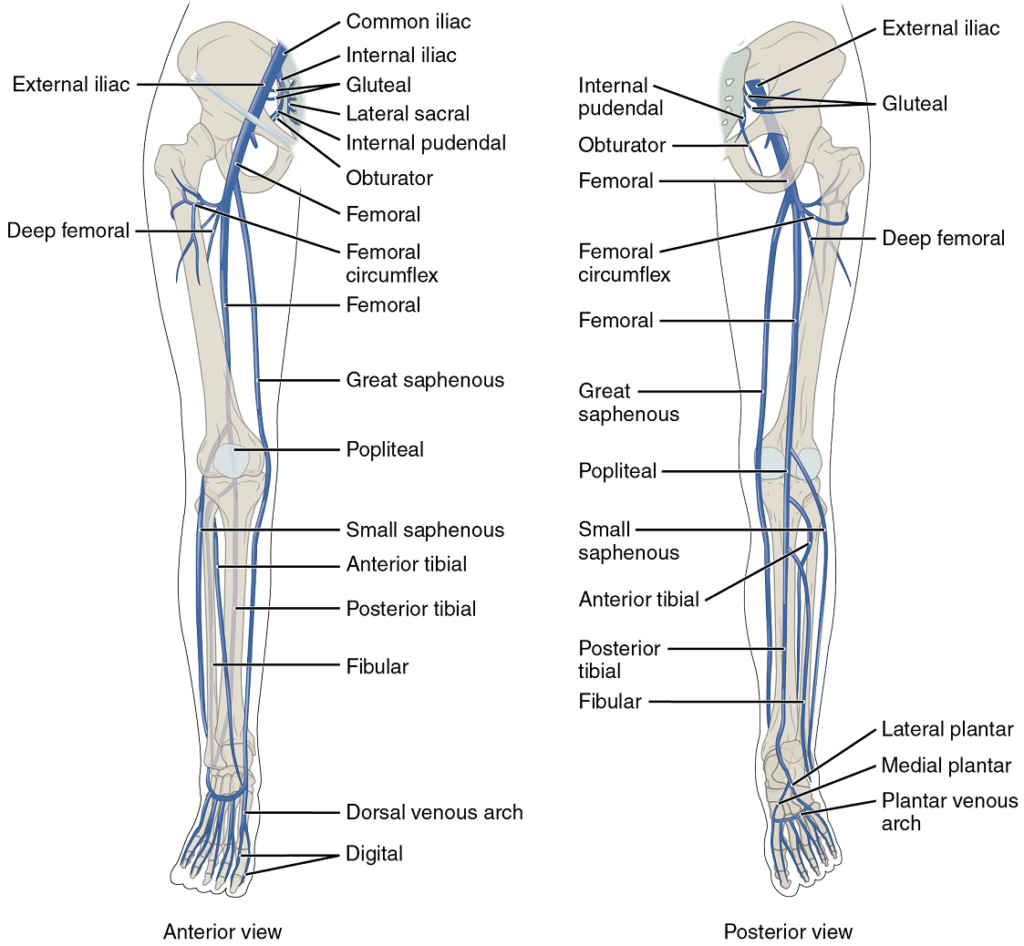
- Veins draining the lower limbs
Learning Objectives
At the end of this unit, you should be able to:
I. Describe the general nature and functions of blood, specify the main components of blood and describe the importance of each.
II. Describe the production of the formed elements of blood.
III. Describe the major factors that stimulate the body to produce more erythrocytes.
IV. Specify the types of leukocytes (white blood cells), their origins and relative quantities in normal blood.
V. Describe the procedure, what information is provided by, and the normal range for the following tests: hemoglobin (Hb), hematocrit (Hct).
VI. Describe the structure and function of platelets.
VII. Specify the two main components of blood that give blood its viscosity, and describe the importance of each to the blood.
VIII. Define hemostasis and describe the mechanisms involved in achieving hemostasis: vascular spasm, platelet plug formation, blood clotting.
IX. Describe the following disorders of hemostasis: thrombus, embolus, hemophilia.
X. Describe how the process of blood clotting is regulated, particularly with respect to prevention of blood clotting when it is not required, rapid initiation and progression of blood clotting when damage occurs, localization of blood clotting to the damaged region, and the dissolution of blood clots (fibrinolysis).
XI. Describe how each of the following affects blood clotting: vitamin K, anticoagulant drugs, thrombolytic agents.
XII. Describe the anatomy of the human heart with respect to the following: location, size, and shape.
XIII. Define and describe the location of the following: pericardium, epicardium, myocardium, endocardium.
XIV. Describe the anatomy and relationship to each other of the four chambers of the heart including the location and general makeup of all valves.
XV. Describe the double circulation and blood flow through the heart and explain the role of the four valves in controlling the direction of blood flow.
XVI. Briefly describe the major components of the coronary circulation and parts of the heart that they feed.
XVII. Specify the components of the conduction system of the heart and describe their functions in the normal conduction of an electrical impulse through the heart and explain the events which constitute and complete the heart beat (i.e. cardiac cycle).
XVIII. Describe the major components of the human electrocardiogram (ECG) and relate these to the electrical and mechanical events of the heart.
XIX. Describe the following major mechanisms that control heart rate: autonomic system, hormones, ionic composition of the blood, and body temperature.
XX. Define the terms systole and diastole in relation to contraction of the chambers of the heart
XXI. Describe relationships between the following components of the cardiovascular system and explain their functions: blood, artery, vein, capillary, atria, and ventricles.
XXII. Compare the structure and function of arteries, veins, and capillaries.
XXIII. Describe what is meant by blood pressure and specify the following: five factors which affect blood pressure, the major mechanisms that control blood pressure, and the average blood pressure of a young adult.
XXIV. Describe what is felt when a pulse is located, and specify four points where an arterial pulse may be felt.
XXV. Describe the following components of the cardiovascular system: the main arteries leaving the heart, and those serving the trunk, appendages, and heart; the main veins entering the heart, and those draining the trunk, appendages, and heart.
Learning Objectives and Guiding Questions
At the end of this unit, you should be able to complete all the following tasks, including answering the guiding questions associated with each task.
I. Describe the general nature and functions of blood, specify the main components of blood and describe the importance of each.
- What are the formed elements of blood?
II. Describe the production of the formed elements of blood.
- Specify the origin and function of each of the formed elements.
- The site(s) of production of formed elements.
III. Describe the major factors that stimulate the body to produce more erythrocytes.
- For the hormone erythropoietin, state:
- Its site of production and release.
- The stimuli for its release.
- Its physiological effects.
- What would the effect on blood pressure be of increasing erythropoietin release or concentration? Explain your reasoning.
IV. Specify the types of leukocytes (white blood cells), their origins and relative quantities in normal blood.
V. Describe the procedure, what information is provided by, and the normal range for the following tests: hemoglobin (Hb), hematocrit (Hct).
- Briefly explain how to determine the hematocrit of a blood sample, then explain:
- What specific information does it give you about an individual’s blood?
- What can that information be used to determine?
- Briefly explain how to determine the hemoglobin content of a blood sample, then explain:
- What specific information does it give you about an individual’s blood?
- What can that information be used to determine?
- Define “anemia”.
VI. Describe the structure and function of platelets.
- What cellular components do platelets possess, and which cellular components do platelets not possess?
VII. Specify the two main components of blood that give blood its viscosity, and describe the importance of each to the blood.
- What factors contribute to the viscosity of blood?
- Describe in general terms how each factor is normally regulated by the body.
VIII. Define hemostasis and describe the mechanisms involved in achieving hemostasis: vascular spasm, platelet plug formation, blood clotting.
- Define “hemostasis” and describe why hemostasis is vital to maintaining homeostasis in the human body.
- What is the specific chemical stimulus that causes the smooth muscle of blood vessels walls to contract when they are damaged, and what is the functional purpose of this contraction?
- Describe in detail the formation of a platelet plug. Include in your description references to the specific stimulus that initially activates platelets, a definition of ‘platelet activation’, and a description of how activated platelets recruit additional platelets to a damaged site.
- Compare and contrast the stimuli, events, and end result of the intrinsic and extrinsic pathways of blood clotting.
- Is it possible to stimulate either the intrinsic or extrinsic pathway of blood clotting, without stimulating the other one? Explain your reasoning.
- Draw a flow chart to describe in detail the intrinsic, extrinsic, and common pathways of blood clotting.
IX. Describe the following disorders of hemostasis: thrombus, embolus, hemophilia.
- What is a ‘thrombus’? How is a thrombus produced, and what is the danger of thrombus production in the human body?
- What is an ‘embolus’? How is an embolus produced, and what is the danger of embolus production in the human body?
- Define ‘hemophilia’. What is the most common cause of hemophilia? Add an annotation to the detailed flow chart you drew of blood clotting to indicate this information.
X. Describe how the process of blood clotting is regulated, particularly with respect to prevention of blood clotting when it is not required, rapid initiation and progression of blood clotting when damage occurs, localization of blood clotting to the damaged region, and the dissolution of blood clots (fibrinolysis).
- Describe the mechanisms in place that both allow rapid production of a blood clot when needed, and prevention of blood clot formation when there is no damage to a blood vessel.
- How are blood clots normally dissolve when they are no longer needed?
XI. Describe how each of the following affects blood clotting: vitamin K, anticoagulant drugs, thrombolytic agents.
- Describe the normal function of vitamin K in the human body. Refer to your detailed flow chart of blood clotting to explain the consequences of a vitamin K deficiency.
- List two examples of anticoagulant drugs and two examples of thrombolytic agents. For each, briefly describe their mechanism of action and contrast their effects on blood clots and/or blood clot formation.
XII. Describe the anatomy of the human heart with respect to the following: location, size, and shape.
- Use correct anatomical terms and complete sentences to describe the position of the human heart in relation to the lungs, diaphragm, vertebral column and thoracic cavity.
XIII. Define and describe the location of the following: pericardium, epicardium, myocardium, endocardium.
- Draw a simple diagram of the heart wall showing all the following structures and on your diagram, wherever possible identify the specific tissue type each layer is composed of:
- Pericardium
- Epicardium
- Myocardium
- Endocardium
XIV. Describe the anatomy and relationship to each other of the four chambers of the heart including the location and general makeup of all valves.
- Distinguish between the upper and lower chambers of the heart
- Describe the partitioning of the heart into left and right chambers.
- Name and describe the structure and location of the valves of the heart
XV. Describe the double circulation and blood flow through the heart and explain the role of the four valves in controlling the direction of blood flow.
- State the names of the two parts of the circulation and their general function in terms of where blood flow is conducted.
- Clearly state the function of each of the four valves found in the human heart. Your description of their function should make reference to the specific location where blood is moving from and to as it passes through each valve.
- Describe the location and function of each of the two main arteries that carry blood out of the heart, and the main veins that carry blood into the human heart.
- List, in order, all the structures through which blood passes as it moves through the heart until it exits, starting from:
- Its entrance into the heart from the venae cavae and coronary sinus
- Its entrance into the heart from the pulmonary veins
- Draw a simplified diagram of the human heart showing the vessels connected directly to its chambers. Show and label all of the following components:
- Pericardium
- Epicardium
- Myocardium
- Endocardium
- Right atrium
- Left atrium
- Right ventricle
- Left ventricle
- Tricuspid valve
- Bicuspid valve
- Coronary sinus
- Aorta
- Superior vena cava
- Inferior vena cava
- Chordae tendinae
- Aortic semilunar valve
- Pulmonary semilunar valve
- Pulmonary trunk
- Right pulmonary arteries
- Left pulmonary arteries
- Right pulmonary veins
- Left pulmonary veins
XVI. Briefly describe the major components of the coronary circulation and parts of the heart that they feed.
- How are the cardiac muscle fibers of the heart supplied with nutrients?
- How is waste removed from the cardiac muscle fibers of the heart?
XVII. Specify the components of the conduction system of the heart and describe their functions in the normal conduction of an electrical impulse through the heart and explain the events which constitute and complete the heart beat (i.e. cardiac cycle).
- Describe, briefly, the general properties of the sinoatrial node (SA Node), atrioventricular node (AV Node), atrioventricular bundle (the bundle of His), right and left bundle branches, and the Purkinje fibers, and the role of each of these in the conduction of a cardiac impulse.
- Describe one heartbeat in detail. Include in your description all the events of the conduction system and the heart muscle, and all the structures the blood passes through (in order!) as it moves through the heart.
- Describe what would happen if the sequence described above is not followed.
XVIII. Describe the major components of the human electrocardiogram (ECG) and relate these to the electrical and mechanical events of the heart.
- Draw and label the major components (P wave, QRS wave, T wave, and the appropriate gaps between them) of a normal ECG tracing. Then describe:
- The electrical events in the heart that underlie each wave.
- The relative times where the ‘lub’ and ‘dub’ sounds of a heartbeat could be heard.
- The physical events in the heart that underlie the ‘lub’ and ‘dub’ sounds of a heartbeat.
XIX. Describe the following major mechanisms that control heart rate: autonomic system, hormones, ionic composition of the blood, and body temperature.
- Describe the influence of proprioceptors, chemoreceptors, and baroreceptors on the cardiovascular centres in the medulla.
- Describe how hormones modify heart rate.
- Describe how the ionic composition of the blood influences heart rate.
- Describe how body temperature affects heart rate.
XX. Define the terms systole and diastole in relation to contraction of the chambers of the heart
XXI. Describe relationships between the following components of the cardiovascular system and explain their functions: blood, artery, vein, capillary, atria, and ventricles.
XXII. Compare the structure and function of arteries, veins, and capillaries.
XXIII. Describe what is meant by blood pressure and specify the following: five factors which affect blood pressure, the major mechanisms that control blood pressure, and the average blood pressure of a young adult.
- Define the term “blood pressure”.
- Describe how blood pressure is measured, and what is considered a “normal” blood pressure.
- Define cardiac output and describe how each of the following physiological factors affect blood pressure:
- Heart rate
- Contractility (strength of contraction) of the heart
- Blood volume
- Peripheral resistance
- Blood viscosity
- Describe how blood pressure is regulated by:
- The nervous system
- The endocrine system
- Autoregulation
XXIV. Describe what is felt when a pulse is located, and specify four points where an arterial pulse may be felt.
- When you manually “take someone’s pulse”, what is causing the pulsing pressure waves you feel?
- List four locations on the human body where a pulse can be taken manually and explain why an arterial pulse can be felt at specific locations rather than just anywhere on the human body.
XXV. Describe the following components of the cardiovascular system: the main arteries leaving the heart, and those serving the trunk, appendages, and heart; the main veins entering the heart, and those draining the trunk, appendages, and heart.
- Draw a flow chart showing the components of the cardiovascular system. Start with the three main components (heart, blood vessels, and blood), and continue by specifying all the constituent parts of each.
- Compare and contrast (clearly!) the anatomical structure and function of arteries, veins, and blood capillaries.
- Draw a simple diagram of the human cardiovascular system that shows both circuits, indicating the vessels blood is moved through as it is passed to and from the head, arms, organs of the abdomen, and lungs. Your diagram should include:
- The main arteries leaving the heart
- The main arteries serving the trunk, appendages and the heart
- The main veins entering the heart
- The main veins draining the trunk, appendages and the heart
Blood
Single-celled organisms do not need blood. They obtain nutrients directly from and excrete wastes directly into their environment. The human organism cannot do that. Our large, complex bodies need blood to deliver nutrients to and remove wastes from our trillions of cells. The heart pumps blood throughout the body in a network of blood vessels. Together, these three components—blood, heart, and vessels—makes up the cardiovascular system.
Part 1: An Overview of Blood
Recall that blood is a connective tissue. Like all connective tissues, it is made up of cellular elements and an extracellular matrix. The cellular elements—referred to as the formed elements—include erythrocytes (red blood cells, or RBCs), leukocytes (white blood cells, or WBCs), and cell fragments called platelets. The extracellular matrix, called plasma, makes blood unique among connective tissues because it is fluid. This fluid, which is mostly water, perpetually suspends the formed elements and enables them to circulate throughout the body within the cardiovascular system.
Functions of Blood: The primary function of blood is to deliver oxygen and nutrients to and remove wastes from body cells, but that is only the beginning of the story. The specific functions of blood also include defense and maintenance of homeostasis.
Transportation: Nutrients from the foods you eat are absorbed in the digestive tract. Most of these travel in the bloodstream directly to the liver, where they are processed and released back into the bloodstream for delivery to body cells. Oxygen from the air you breathe diffuses into the blood, which moves from the lungs to the heart, which then pumps it out to the rest of the body. Moreover, endocrine glands scattered throughout the body release their products, called hormones, into the bloodstream, which carries them to distant target cells. Blood also picks up cellular wastes and by products, and transports them to various organs for removal. For instance, blood moves carbon dioxide to the lungs for exhalation from the body, and various waste products are transported to the kidneys and liver for excretion from the body in the form of urine or bile.
Defense: Many types of leukocytes protect the body from external threats, such as disease-causing bacteria that have entered the bloodstream in a wound. Other leukocytes seek out and destroy internal threats, such as cells with mutated DNA that could multiply to become cancerous, or body cells infected with viruses.
When damage to the vessels results in bleeding, blood platelets and certain proteins dissolved in the plasma, the fluid portion of the blood, interact to block the ruptured areas of the blood vessels involved. This protects the body from further blood loss.
Maintenance of Homeostasis: Recall that body temperature is regulated via a classic negative-feedback loop. If you were exercising on a warm day, your rising core body temperature would trigger several homeostatic mechanisms, including increased transport of blood from your core to your body periphery, which is typically cooler. As blood passes through the vessels of the skin, heat would be dissipated to the environment, and the blood returning to your body core would be cooler. In contrast, on a cold day, blood is diverted away from the skin to maintain a warmer body core. In extreme cases, this may result in frostbite.
Blood also helps to maintain the chemical balance of the body. Proteins and other compounds in blood act as buffers, which thereby help to regulate the pH of body tissues. Blood also helps to regulate the water content of body cells.
Composition of Blood: You have probably had blood drawn from a superficial vein in your arm, which was then sent to a lab for analysis. Some of the most common blood tests—for instance, those measuring lipid or glucose levels in plasma—determine which substances are present within blood and in what quantities. Other blood tests check for the composition of the blood itself, including the quantities and types of formed elements.
One such test, called a hematocrit, measures the percentage of red blood cells, clinically known as erythrocytes, in a blood sample. It is performed by spinning the blood sample in a specialized centrifuge, a process that causes the heavier elements suspended within the blood sample to separate from the lightweight, liquid plasma (Figure 1). Because the heaviest elements in blood are the erythrocytes, these settle at the bottom of the hematocrit tube. Located above the erythrocytes is a pale, thin layer composed of the remaining formed elements of blood.
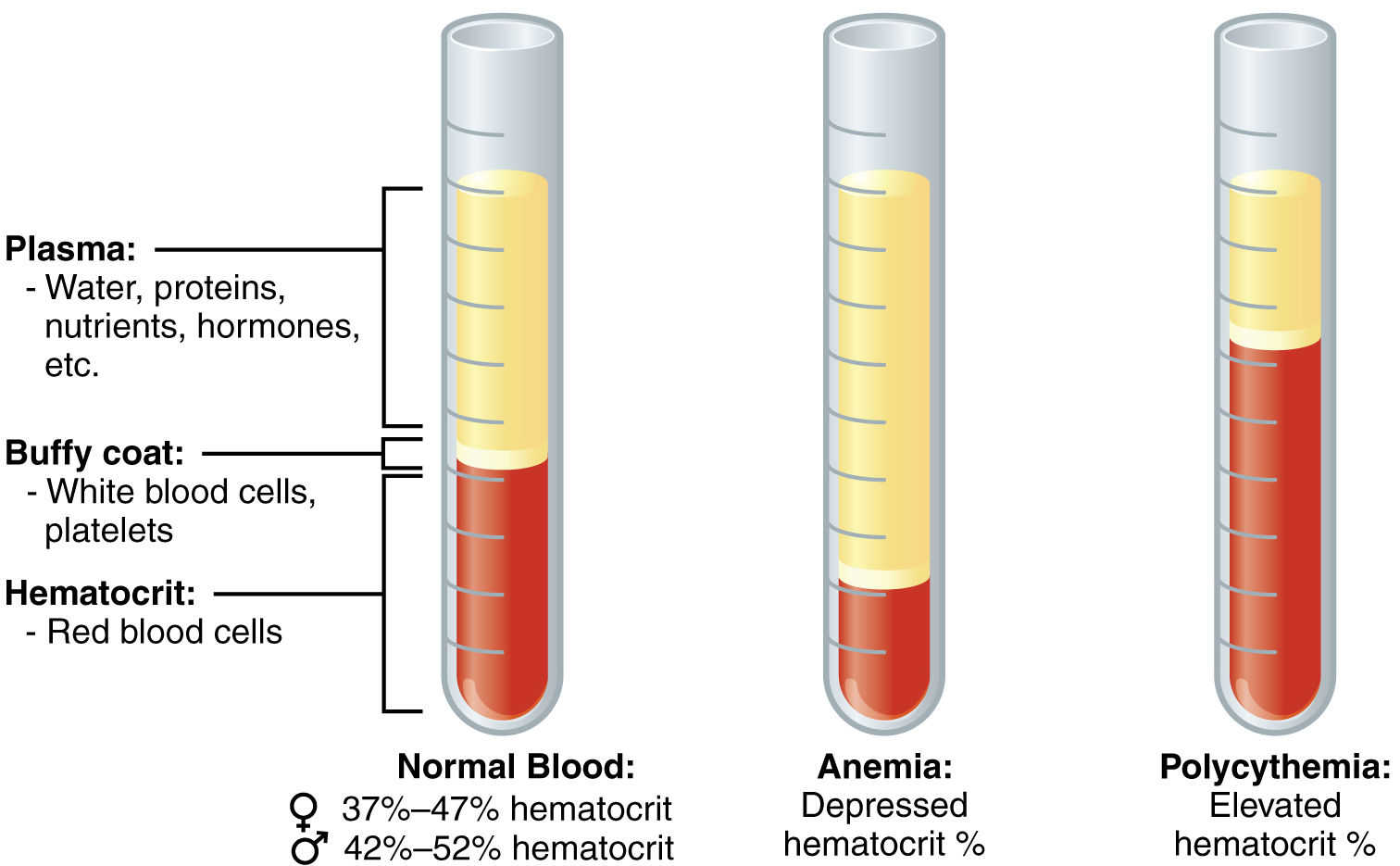
This pale, thin layer of centrifuged blood sample consists of the white blood cells, clinically known as leukocytes, and the platelets, cell fragments also called thrombocytes. This layer is referred to as the buffy coat because of its colour; it normally constitutes less than 1% of a blood sample. Above the buffy coat is the blood plasma, normally a pale, straw-coloured fluid, which constitutes the remainder of the sample.
The volume of erythrocytes after centrifugation is also commonly referred to as packed cell volume (PCV). In normal blood, about 45% of a sample is erythrocytes. The hematocrit of any one sample can vary significantly, and may be 36-50%, depending on sex and other factors. Normal hematocrit values for females range from 37 to 47%, with a mean value of 41%; for males, hematocrit ranges from 42 to 52%, with a mean of 47%. The percentage of other formed elements, the leukocytes and platelets, is extremely small so it is not normally considered with the hematocrit. The mean plasma percentage is the percent of blood that is not erythrocytes: for females, it is approximately 59% (or 100 minus 41), and for males, it is approximately 53% (or 100 minus 47).
Characteristics of Blood: When you think about blood, the first characteristic that probably comes to mind is its colour. Blood that has just taken up oxygen in the lungs is bright red, and blood that has released oxygen in the tissues is a more dusky red. This is because hemoglobin is a pigment that changes colour, depending upon the degree of oxygen saturation.
Blood is viscous and somewhat sticky to the touch. It has a viscosity approximately five times greater than water. Viscosity is a measure of a fluid’s thickness or resistance to flow, and is influenced by plasma proteins and formed elements (usually albumin concentration and the number of erythrocytes) within the blood. The viscosity of blood has a dramatic impact on blood pressure and flow. Consider the difference in flow between water and honey. The more viscous honey would demonstrate a greater resistance to flow than the less viscous water. The same principle applies to blood.
The normal temperature of blood is slightly higher than normal body temperature—about 38 °C (or 100.4 F), compared to 37 °C (or 98.6 F) for an internal body temperature reading, although daily variations of 0.5 °C are normal. Although the surface of blood vessels is relatively smooth, as blood flows through them, it experiences some friction and resistance, especially as vessels age and lose their elasticity, thereby producing heat. This accounts for its slightly higher temperature.
The pH of blood averages about 7.4, but can range from 7.35 to 7.45 in a healthy person. Blood is therefore somewhat more basic (alkaline) on a chemical scale than pure water, which has a pH of 7.0. Blood contains numerous buffers that help to regulate pH.
Blood constitutes approximately 8% of adult body weight. Adult males typically average about 5-6 liters of blood; adult females average 4-5 liters.
Blood Plasma: Like other fluids in the body, plasma is composed primarily of water, and is about 92% water. Dissolved or suspended within this water is a mixture of substances, most of which are proteins. There are literally hundreds of substances dissolved or suspended in the plasma, although many of them are found only in very small quantities.
Plasma Proteins: About 7% of the volume of plasma – nearly all that is not water – is made of proteins. These include several plasma proteins (proteins that are unique to the plasma), plus a much smaller number of regulatory proteins, including enzymes and some hormones (Table 1).
- Albumin is the most abundant of the plasma proteins. Manufactured by the liver, albumin molecules serve as binding proteins—transport vehicles for fatty acids and steroid hormones. Recall that lipids are hydrophobic; however, their binding to albumin enables their transport in the watery plasma. Albumin is also the most significant contributor to the osmotic pressure of blood; that is, its presence holds water inside the blood vessels and draws water from the tissues, across blood vessel walls, and into the bloodstream. This in turn helps to maintain both blood volume and blood pressure. Albumin normally accounts for approximately 54% of the total plasma protein content, in clinical levels of 3.5–5.0 g/dL blood.
- The second most common plasma proteins are the globulins. A heterogeneous group, there are three main subgroups known as alpha, beta, and gamma globulins. The alpha and beta globulins transport iron, lipids, and the fat-soluble vitamins A, D, E, and K to the cells; like albumin, they also contribute to osmotic pressure. The gamma globulins are proteins involved in immunity and are better known as antibodies or immunoglobulins. Although other plasma proteins are produced by the liver, immunoglobulins are produced by specialized leukocytes known as plasma cells. Globulins make up approximately 38% of the total plasma protein volume, in clinical levels of 1.0–1.5 g/dL blood.
- The least abundant plasma protein is fibrinogen. Like albumin and the alpha and beta globulins, fibrinogen is produced by the liver. It is essential for blood clotting, a process described later in this chapter. Fibrinogen accounts for about 7% of the total plasma protein volume, in clinical levels of 0.2–0.45 g/dL blood.
Other Plasma Solutes: In addition to proteins, plasma contains a wide variety of other substances. These include various electrolytes, such as sodium, potassium, and calcium ions; dissolved gases, such as oxygen, carbon dioxide, and nitrogen; various organic nutrients, such as vitamins, lipids, glucose, and amino acids; and metabolic wastes. All of these non-protein solutes combined contribute approximately 1% to the total volume of plasma.

| Component and % of blood | Subcomponent and % of component | Type and % (where appropriate) | Site of production | Major function(s) |
|---|---|---|---|---|
| Plasma 46-63% | Water 92% | Fluid | Absorbed by intestinal tract or produced by metabolism | Transport medium |
| Plasma proteins 7% | Albumin 54-60% | Liver | Maintain osmotic concentration, transport lipid molecules | |
| Globulins 35-38% | Alpha globulins: liver | Transport, maintain osmotic concentration | ||
| Beta globulins: liver | Transport, maintain osmotic concentration | |||
| Gamma globulins (immunoglobulins): plasma cells | Immune responses | |||
| Fibrinogen 4-7% | Liver | Blood clotting in hemostasis | ||
| Regulatory proteins <1% | Hormones and enzymes | Various sources | Regulate various body functions | |
| Other solutes 1% | Nutrients, gases, and wastes | Absorbed by intestinal tract, exchanged in respiratory system, or produced by cells | Numerous and varied | |
| Formed elements 37-54% | Erythrocytes 99% | Erythrocytes | Red bone marrow | Transport gases (primarily O2, some CO2) |
| Leukocytes <1% | Granular leukocytes: neutrophils, eosinophils, basophils | Red bone marrow | Nonspecific immunity | |
| Agranular leukocytes: lymphocytes, monocytes | Lymphocytes: red bone marrow and lymphatic tissue | Lymphocytes: specific immunity | ||
| Monocytes: red bone marrow | Monocytes: nonspecific immunity | |||
| Platelets <1% | Megakaryocytes in red bone marrow | Hemostasis |
Part 2: Production of the Formed Elements
The lifespan of the formed elements is very brief. Although one type of leukocyte called memory cells can survive for years, most erythrocytes, leukocytes, and platelets normally live only a few hours to a few weeks. Thus, the body must form new blood cells and platelets quickly and continuously. When you donate a unit of blood during a blood drive (approximately 475 mL, or about 1 pint), your body typically replaces the donated plasma within 24 hours, but it takes about 4 to 6 weeks to replace the blood cells. This restricts the frequency with which donors can contribute their blood. The process by which this replacement occurs is called hemopoiesis, or hematopoiesis (from the Greek root haima- = “blood”; -poiesis = “production”).
Sites of Hemopoiesis: Prior to birth, hemopoiesis occurs in a number of tissues, beginning with the yolk sac of the developing embryo, and continuing in the foetal liver, spleen, lymphatic tissue, and eventually the red bone marrow. Following birth, most hemopoiesis occurs in the red marrow, a connective tissue within the spaces of spongy (cancellous) bone tissue. In children, hemopoiesis can occur in the medullary cavity of long bones; in adults, the process is largely restricted to the cranial and pelvic bones, the vertebrae, the sternum, and the proximal epiphyses of the femur and humerus.
Differentiation of Formed Elements from Stem Cells: All formed elements arise from stem cells of the red bone marrow. Recall that stem cells undergo mitosis plus cytokinesis (cellular division) to give rise to new daughter cells: One of these remains a stem cell and the other differentiates into one of any number of diverse cell types (Figure 2).
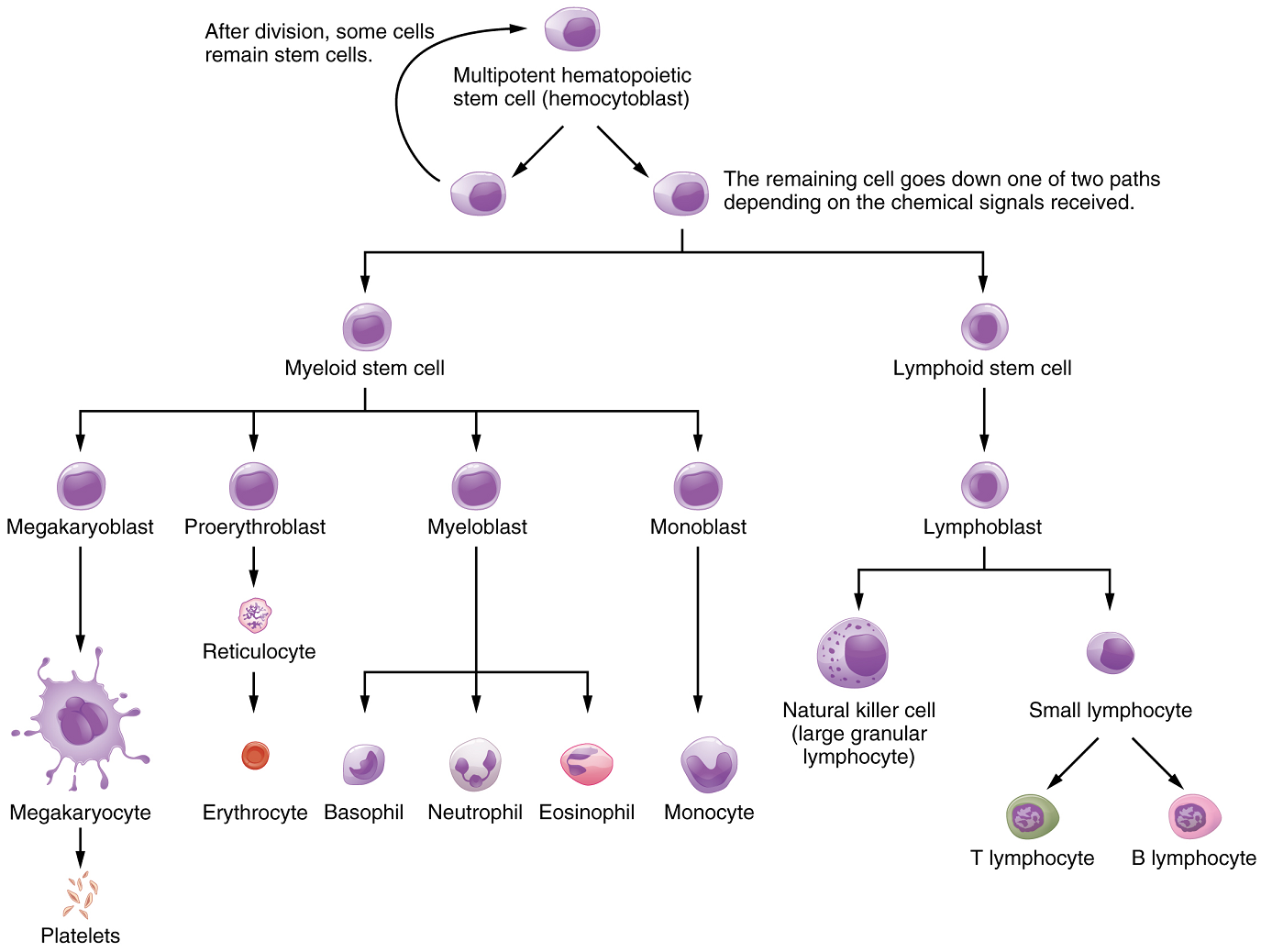
Hemopoietic Growth Factors: Development from stem cells to precursor cells to mature cells is again initiated by hemopoietic growth factors. The growth factor responsible for the production of erythrocytes is erythropoietin (EPO). Erythropoietin is a hormone secreted by the kidneys in response to low oxygen levels. Some athletes use synthetic EPO as a performance-enhancing drug (called blood doping) to increase RBC counts and subsequently increase oxygen delivery to tissues throughout the body. EPO is a banned substance in most organized sports, but it is also used medically in the treatment of certain anemia, specifically those triggered by certain types of cancer, and other disorders in which increased erythrocyte counts and oxygen levels are desirable.
Part 3: Erythrocytes
The erythrocyte, commonly known as a red blood cell (or RBC), is by far the most common formed element: A single drop of blood contains millions of erythrocytes and just thousands of leukocytes. Specifically, males have about 5.4 million erythrocytes per microliter (µL) of blood, and females have approximately 4.8 million per µL. In fact, erythrocytes are estimated to make up about 25% of all cells in the body. As you can imagine, they are quite small cells, with a mean diameter of only about 7–8 micrometers (µm) (Table 2). The primary functions of erythrocytes are to pick up inhaled oxygen from the lungs and transport it to the body’s tissues, and to pick up some (about 24%) of the carbon dioxide waste produced at the tissues and transport it to the lungs for exhalation. Erythrocytes remain within the vascular network. Although leukocytes typically leave the blood vessels to perform their defensive functions, movement of erythrocytes from the blood vessels is abnormal. Their unique structure enables them to change their shape to squeeze through capillaries.
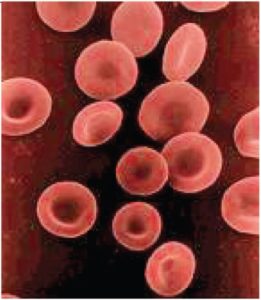
Erythrocytes are biconcave disks; that is, they are plump at their periphery and very thin in the centre (Figure 3). Since they lack most organelles, there is more interior space for the presence of the hemoglobin molecules that transport gases. The biconcave shape also provides a greater surface area across which gas exchange can occur, relative to its volume; a sphere of a similar diameter would have a lower surface area-to-volume ratio. In the capillaries, the oxygen carried by the erythrocytes can diffuse into the plasma and then through the capillary walls to reach the cells, whereas some of the carbon dioxide produced by the cells as a waste product diffuses into the capillaries to be picked up by the erythrocytes. Capillary beds are extremely narrow, slowing the passage of the erythrocytes and providing an extended opportunity for gas exchange to occur. However, the space within capillaries can be so minute that, despite their own small size, erythrocytes may have to fold in on themselves if they are to make their way through.
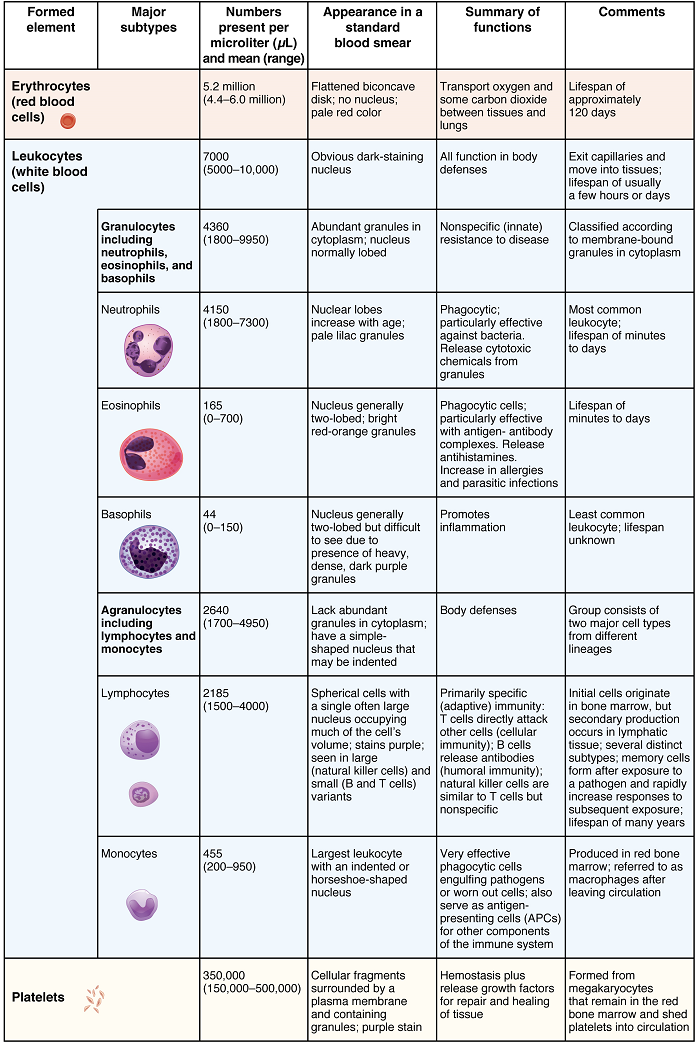
Hemoglobin: Hemoglobin is a large molecule made up of proteins and iron. It consists of four folded chains of a protein called globin, designated alpha 1 and 2, and beta 1 and 2 (Figure 4a). Each of these globin molecules is bound to a red pigment molecule called heme, which contains an ion of iron (Fe2+) (Figure 4b).
Each iron ion in the heme can bind to one oxygen molecule; therefore, each hemoglobin molecule can transport four oxygen molecules. An individual erythrocyte may contain about 300 million hemoglobin molecules, and therefore can bind to and transport up to 1.2 billion oxygen molecules. These oxygen molecules come from the air we breathe; they diffuse across the respiratory membrane in the lungs, then into erythrocytes where they can bind to hemoglobin and be carried back to the heart and then to the rest of the body.
Carbon dioxide enters the bloodstream at the tissue level, and among other transport mechanisms can bind to one end of a subunit of hemoglobin. From the capillaries, the carbon dioxide is carried back to the lungs, where it is released.
Changes in the levels of erythrocytes can have significant effects on the body’s ability to effectively deliver oxygen to the tissues. Ineffective hematopoiesis results in insufficient numbers of erythrocytes and results in one of several forms of anemia. An overproduction of erythrocytes produces a condition called polycythemia. The primary drawback with polycythemia is not a failure to directly deliver enough oxygen to the tissues, but rather the increased viscosity of the blood, which makes it more difficult for the heart to circulate the blood.
In patients with insufficient hemoglobin, the tissues may not receive sufficient oxygen, resulting in another form of anemia.
In contrast to anemia, an elevated erythrocyte count is called polycythemia and is detected in a patient’s elevated hematocrit. It can occur transiently in a person who is dehydrated; when water intake is inadequate or water losses are excessive, the plasma volume falls. As a result, the hematocrit rises. A mild form of polycythemia is chronic but normal in people living at high altitudes; the decreased oxygen availability at high altitudes released in erythropoietin release (discussed earlier in this chapter), resulting in increased erythrocyte production. Some elite athletes train at high elevations specifically to induce this phenomenon. Finally, a type of bone marrow disease called polycythemia vera (from the Greek vera = “true”) causes an excessive production of immature erythrocytes. Polycythemia vera can dangerously elevate the viscosity of blood, raising blood pressure and making it more difficult for the heart to pump blood throughout the body. It is a relatively rare disease that occurs more often in men than women and is more likely to be present in elderly patients those over 60 years of age.
Part 4: Leukocytes and Platelets
The leukocyte, commonly known as a white blood cell (or WBC), is a major component of the body’s defenses against disease. Leukocytes protect the body against invading microorganisms and body cells with mutated DNA, and they clean up debris. Platelets are essential for the repair of blood vessels when damage to them has occurred; they also provide growth factors for healing and repair.
Characteristics of Leukocytes: Although leukocytes and erythrocytes both originate from hematopoietic stem cells in the bone marrow, they differ from each other in many significant ways. The types of leukocytes will be discussed in a succeeding unit (Immunity).
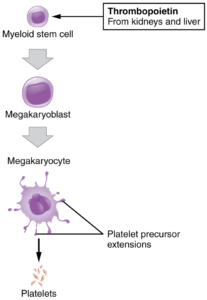
Platelets: You may occasionally see platelets referred to as thrombocytes, but because this name suggests they are a type of cell, it is not accurate. A platelet is not a cell but rather a fragment of the cytoplasm of a cell called a megakaryocyte that is surrounded by a plasma membrane. Megakaryocytes are descended from myeloid stem cells and are large, typically 50–100 µm in diameter, and contain an enlarged, lobed nucleus. Thrombopoietin, a glycoprotein secreted by the kidneys and liver, stimulates the proliferation of megakaryoblasts, which mature into megakaryocytes. These remain within bone marrow tissue (Figure 5) and ultimately form platelet-precursor extensions that extend through the walls of bone marrow capillaries to release into the circulation thousands of cytoplasmic fragments, each enclosed by a bit of plasma membrane. These enclosed fragments are platelets. Each megakarocyte releases 2000–3000 platelets during its lifespan. Following platelet release, megakaryocyte remnants, which are little more than a cell nucleus, are consumed by macrophages (macrophages are discussed further in the Immunity chapter).
Platelets are relatively small, 2–4 µm in diameter, but numerous, with typically 150,000–160,000 per µL of blood. After entering the circulation, approximately one-third migrate to the spleen for storage for later release in response to any rupture in a blood vessel. They then become activated to perform their primary function, which is to limit blood loss. Platelets remain only about 10 days, then are phagocytized by macrophages found in the spleen and liver. Platelets are critical to hemostasis, the stoppage of blood flow following damage to a vessel. They also secrete a variety of growth factors essential for growth and repair of tissue, particularly connective tissue. Infusions of concentrated platelets are now being used in some therapies to stimulate healing.
Disorders of Platelets: Thrombocytosis is a condition in which there are too many platelets. This may trigger formation of unwanted blood clots (thrombosis), a potentially fatal disorder. If there is an insufficient number of platelets, called thrombocytopenia, blood may not clot properly, and excessive bleeding may result.
Part 5: Hemostasis
Platelets are key players in hemostasis, the process by which the body seals a ruptured blood vessel and prevents further loss of blood. Although rupture of larger vessels usually requires medical intervention, hemostasis is quite effective in dealing with small, simple wounds. There are three steps to the process: vascular spasm, the formation of a platelet plug, and coagulation (blood clotting) (Figure 6). Failure of any of these steps will result in hemorrhage – excessive bleeding.
Vascular Spasm: When a vessel is severed or punctured, or when the wall of a vessel is damaged, vascular spasm occurs. In vascular spasm, the smooth muscle in the walls of the vessel contracts dramatically. Small blood vessels have smooth muscle arranged in circular layers; larger vessels also have longitudinal layers of smooth muscle. The circular layers tend to constrict the flow of blood, whereas the longitudinal layers, when present, draw the vessel back into the surrounding tissue, often making it more difficult for a surgeon to locate, clamp, and tie off a severed vessel. The vascular spasm response is believed to be triggered by several chemicals called endothelins that are released by vessel-lining cells and by pain receptors in response to vessel injury. This phenomenon typically lasts for up to 30 minutes, although it can last for hours.
Formation of the Platelet Plug: In the second step, platelets, which normally float free in the plasma, encounter the area of vessel rupture with the exposed underlying connective tissue and collagenous fibers. The platelets begin to clump together, become spiked and sticky, and bind to the exposed collagen and endothelial lining. This process is assisted by a glycoprotein in the blood plasma called von Willebrand factor, which helps stabilize the growing platelet plug. As platelets collect, they simultaneously release chemicals from their granules into the plasma that further contribute to hemostasis. Among the substances released by the platelets are:
- Adenosine diphosphate (ADP), which helps additional platelets to adhere to the injury site, reinforcing and expanding the platelet plug
- Serotonin, which maintains vasoconstriction
- Prostaglandins and phospholipids, which also maintain vasoconstriction and help to activate further clotting chemicals
A platelet plug can temporarily seal a small opening in a blood vessel. Plug formation, in essence, buys the body time while more sophisticated and durable repairs are being made. In a similar manner, even modern naval warships still carry an assortment of wooden plugs to temporarily repair small breaches in their hulls until permanent repairs can be made.
Coagulation: Those more sophisticated and more durable repairs are collectively called coagulation, the formation of a blood clot. The process is sometimes characterized as a cascade, because one event prompts the next as in a multi-level waterfall. The result is the production of a gelatinous but robust clot made up of a mesh of fibrin – an insoluble filamentous protein derived from the blood plasma protein fibrinogen (Table 1) – in which platelets and blood cells are trapped.
Clotting Factors Involved in Coagulation: In the coagulation cascade, chemicals called clotting factors (or coagulation factors) prompt reactions that activate still more coagulation factors (Figure 6b). The process is complex, but is initiated along two basic pathways: the extrinsic pathway which normally is triggered by tissue damage, and the intrinsic pathway which begins in the bloodstream and is triggered by damage to the wall of the vessel.
Both of these merge into a third pathway, referred to as the common pathway (Figure 6b). All three pathways are dependent upon the 12 known clotting factors, including Ca2+ and vitamin K (Table 3). Clotting factors are secreted primarily by the liver and the platelets. The liver requires the fat-soluble vitamin K to produce many of them. Vitamin K (along with biotin and folate) is somewhat unusual among vitamins in that it is not only consumed in the diet but is also synthesized by bacteria residing in the large intestine. The calcium ion, also considered as factor IV, is derived from the diet and from the breakdown of bone. Some recent evidence indicates that activation of various clotting factors occurs on specific receptor sites on the surfaces of platelets.
The 12 clotting factors are numbered I through XIII according to the order of their discovery. Factor VI was once believed to be a distinct clotting factor, but is now thought to be identical to factor V. Rather than renumber the other factors, factor VI was allowed to remain as a placeholder and also a reminder that knowledge changes over time.
Extrinsic Pathway: The quicker responding and more direct extrinsic pathway (also known as the tissue factor pathway) begins when damage occurs to the surrounding tissues, such as in a traumatic injury. Upon contact with blood plasma, the damaged extravascular cells, which are extrinsic to the bloodstream, release factor III (thromboplastin). Ca2+ and factor VII (proconvertin), activated by factor III forms an enzyme complex. This enzyme complex leads to activation of factor X (Stuart–Prower factor), which activates the common pathway discussed below. The events in the extrinsic pathway are completed in a matter of seconds.
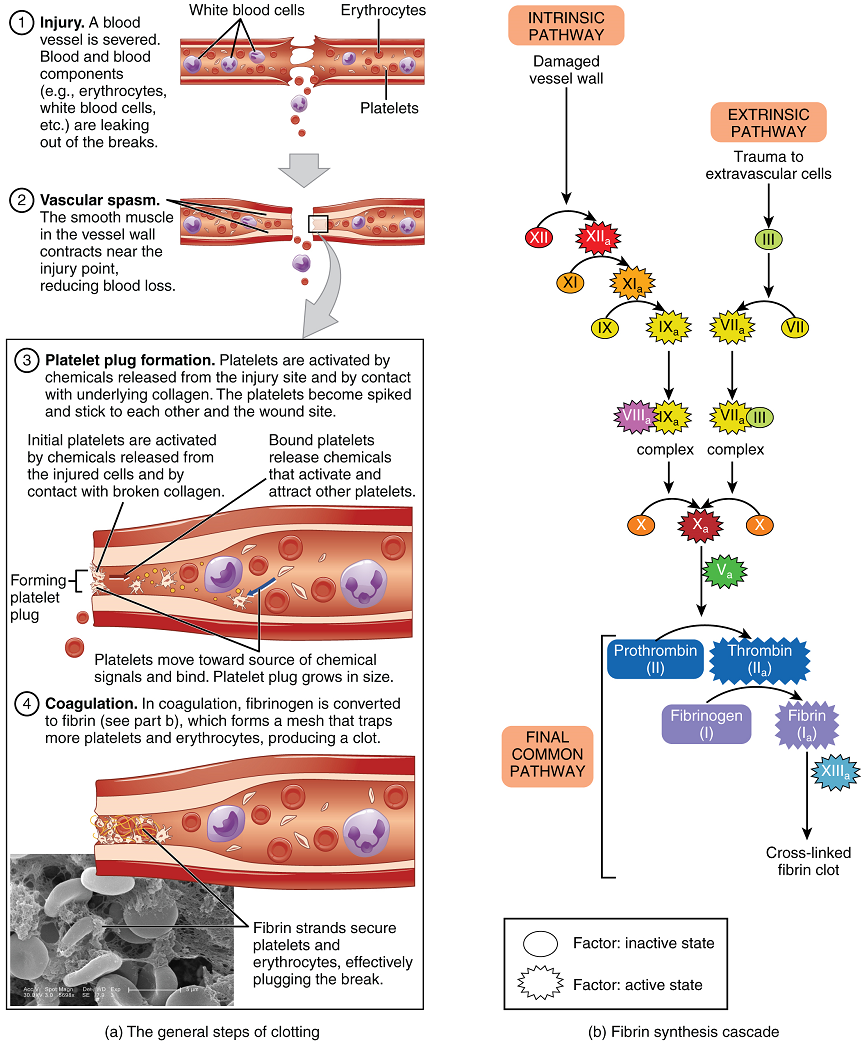
Intrinsic Pathway: The intrinsic pathway (also known as the contact activation pathway) is longer and more complex. In this case, the clotting factors involved are all intrinsic to (present within) the bloodstream. This pathway is prompted by damage to the walls of blood vessels that exposes the initiating clotting factor (clotting factor XII) to collagen. Within the body, factor XII is typically activated when it encounters negatively charged molecules, such as inorganic polymers and phosphate produced earlier in the series of intrinsic pathway reactions. Factor XII sets off a series of reactions to form an enzyme complex that activates factor X (Stuart-Prower factor or thrombokinase), leading to the common pathway. The events in the intrinsic pathway are completed in a few minutes.
| Factor | Name | Type of molecule | Source | Pathway(s) |
|---|---|---|---|---|
| I | Fibrinogen | Plasma protein | Liver | Common; converted into fibrin |
| II | Prothrombin | Plasma protein | Liver* | Common; converted into fibrin |
| III | Tissue thromboplastin or tissue factor (TF) | Lipoprotein mixture | Damaged cells and platelets | Extrinsic |
| IV | Calcium ions (Ca2+) | Inorganic ions in plasma | Diet, platelets, bone matrix | Entire process |
| V | Proaccelerin | Plasma protein | Liver, platelets | Extrinsic and intrinsic |
| VI | Not used (historical use; identical to factor V) | Not used | Not used | Not used |
| VII | Proconvertin | Plasma protein | Liver* | Extrinsic |
| VIII | Antihemolytic factor A | Plasma protein factor | Platelets and endothelial cells | Intrinsic; deficiency results in hemophilia A |
| IX | Antihemolytic factor B (plasma thromboplastin component) | Plasma protein | Liver* | Intrinsic; deficiency results in hemophilia B |
| X | Thrombokinase (Stuart-Prower factor) | Protein | Liver* | Extrinsic and intrinsic |
| XI | Antihemolytic factor C (plasma thromboplastin antecedent) | Plasma protein | Liver | Intrinsic; deficiency results in hemophilia C |
| XII | Hageman factor | Plasma protein | Liver | Intrinsic; initiates clotting in vitro; activates plasmin |
| XIII | Fibrin-stabilizing factor | Plasma protein | Liver, platelets | Stabilizes fibrin; slows fibrinolysis |
Common Pathway: Both the intrinsic and extrinsic pathways lead to the common pathway, in which fibrin is produced to seal off the vessel. Once factor X has been activated by either the intrinsic or extrinsic pathway, the enzyme prothrombinase converts factor II, the inactive enzyme prothrombin, into the active enzyme thrombin. (Note that if the enzyme thrombin were not normally in an inactive form, clots would form spontaneously, a condition not consistent with life.) Then, thrombin converts factor I, the soluble fibrinogen, into insoluble fibrin protein strands. Factor XIII then stabilizes the fibrin clot.
The stabilized clot is acted upon by contractile proteins within the platelets. As these proteins contract, they pull on the fibrin threads, bringing the edges of the clot more tightly together, somewhat as we do when tightening loose shoelaces. This process also wrings out of the clot a small amount of fluid called serum, which is blood plasma without its clotting factors.
Fibrinolysis: To restore normal blood flow as the vessel heals, the clot must eventually be removed. Fibrinolysis is the gradual degradation of the clot. Again, there is a fairly complicated series of reactions that involves factor XII and protein-catabolizing enzymes. During this process, the inactive protein plasminogen is converted into the active plasmin, which gradually breaks down the fibrin of the clot. Additionally, bradykinin, a vasodilator, is released, reversing the effects of the serotonin and prostaglandins from the platelets. This allows the smooth muscle in the walls of the vessels to relax and helps to restore the circulation.
Plasma Anticoagulants: An anticoagulant is any substance that opposes coagulation. Several circulating plasma anticoagulants play a role in limiting the coagulation process to the region of injury and restoring a normal, clot-free condition of blood. For instance, a cluster of proteins collectively referred to as the protein C system inactivates clotting factors involved in the intrinsic pathway. Tissue factor pathway inhibitor (TFPI) inhibits the conversion of the inactive factor VII to the active form in the extrinsic pathway. Antithrombin inactivates factor X and opposes the conversion of prothrombin (factor II) to thrombin in the common pathway. Basophils release heparin, a short-acting anticoagulant that also opposes prothrombin. Heparin is also found on the surfaces of cells lining the blood vessels. A pharmaceutical form of heparin is often administered therapeutically, for example, in surgical patients at risk for blood clots.
Among the many known biochemical activities of aspirin is its role as an anticoagulant. Aspirin (acetylsalicylic acid) is very effective at inhibiting the aggregation of platelets. It is routinely administered during a heart attack or stroke to reduce the adverse effects. Physicians sometimes recommend that patients at risk for cardiovascular disease take a low dose of aspirin on a daily basis as a preventive measure. However, aspirin can also lead to serious side effects, including increasing the risk of ulcers. A patient is well advised to consult a physician before beginning any aspirin regimen.
Disorders of Clotting: Either an insufficient or an excessive production of platelets can lead to severe disease or death. As discussed earlier, an insufficient number of platelets, called thrombocytopenia, typically results in the inability of blood to form clots. This can lead to excessive bleeding, even from minor wounds.
Another reason for failure of the blood to clot is the inadequate production of functional amounts of one or more clotting factors. This is the case in the genetic disorder hemophilia, which is actually a group of related disorders, the most common of which is hemophilia A, accounting for approximately 80% of cases. This disorder results in the inability to synthesize sufficient quantities of factor VIII. Regular infusions of clotting factors isolated from healthy donors can help prevent bleeding in hemophiliac patients. At some point, genetic therapy may become a viable option.
A thrombus (plural = thrombi) is an aggregation of platelets, erythrocytes, and even WBCs typically trapped within a mass of fibrin strands. While the formation of a clot is normal following the hemostatic mechanism just described, thrombi can form within an intact or only slightly damaged blood vessel. A thrombus can seriously impede blood flow to or from a region and will cause a local increase in blood pressure. If flow is to be maintained, the heart will need to generate a greater pressure to overcome the resistance.
When a portion of a thrombus breaks free from the vessel wall and enters the circulation, it is referred to as an embolus. An embolus that is carried through the bloodstream can be large enough to block a vessel critical to a major organ. When it becomes trapped, an embolus is called an embolism. In the heart, brain, or lungs, an embolism may accordingly cause a heart attack, a stroke, or a pulmonary embolism. These are medical emergencies.
A class of drugs collectively known as thrombolytic agents can help speed up the degradation of an abnormal clot. If a thrombolytic agent is administered to a patient within 3 hours following a thrombotic stroke, the patient’s prognosis improves significantly. However, some strokes are not caused by thrombi, but by hemorrhage. Thus, the cause must be determined before treatment begins. Tissue plasminogen activator is an enzyme that catalyzes the conversion of plasminogen to plasmin, the primary enzyme that breaks down clots. It is released naturally by endothelial cells but is also used in clinical medicine as a thrombolytic agent. New research is progressing using compounds isolated from the venom of some species of snakes, particularly vipers and cobras, which may also have therapeutic value as thrombolytic agents.
The Heart
In this section, you will explore the remarkable pump that propels the blood into the vessels. There is no single better word to describe the function of the heart other than “pump,” since its contraction develops the pressure that ejects blood into the major vessels: the aorta and pulmonary trunk. From these vessels, the blood is distributed to the remainder of the body. Although the connotation of the term “pump” suggests a mechanical device made of steel and plastic, the anatomical structure is a living, sophisticated muscle. As you read this chapter, try to keep these twin concepts in mind: pump and muscle.
Although the term “heart” is an English word, cardiac (heart-related) terminology can be traced back to the Latin term, “kardia.” Cardiology is the study of the heart, and cardiologists are the physicians who deal primarily with the heart.
Part 1: Heart Anatomy
The vital importance of the heart is obvious. If one assumes an average rate of contraction of 75 contractions per minute, a human heart would contract approximately 108,000 times in one day, more than 39 million times in one year, and nearly 3 billion times during a 75-year lifespan. Each of the major pumping chambers of the heart ejects approximately 70 mL blood per contraction in a resting adult. This would be equal to 5.25 liters of fluid per minute and approximately 14,000 liters per day. Over one year, that would equal 10,000,000 liters (2.6 million gallons) of blood sent through roughly 96,000 km (60,000 miles) of vessels. In order to understand how that happens, it is necessary to understand the anatomy and physiology of the heart.
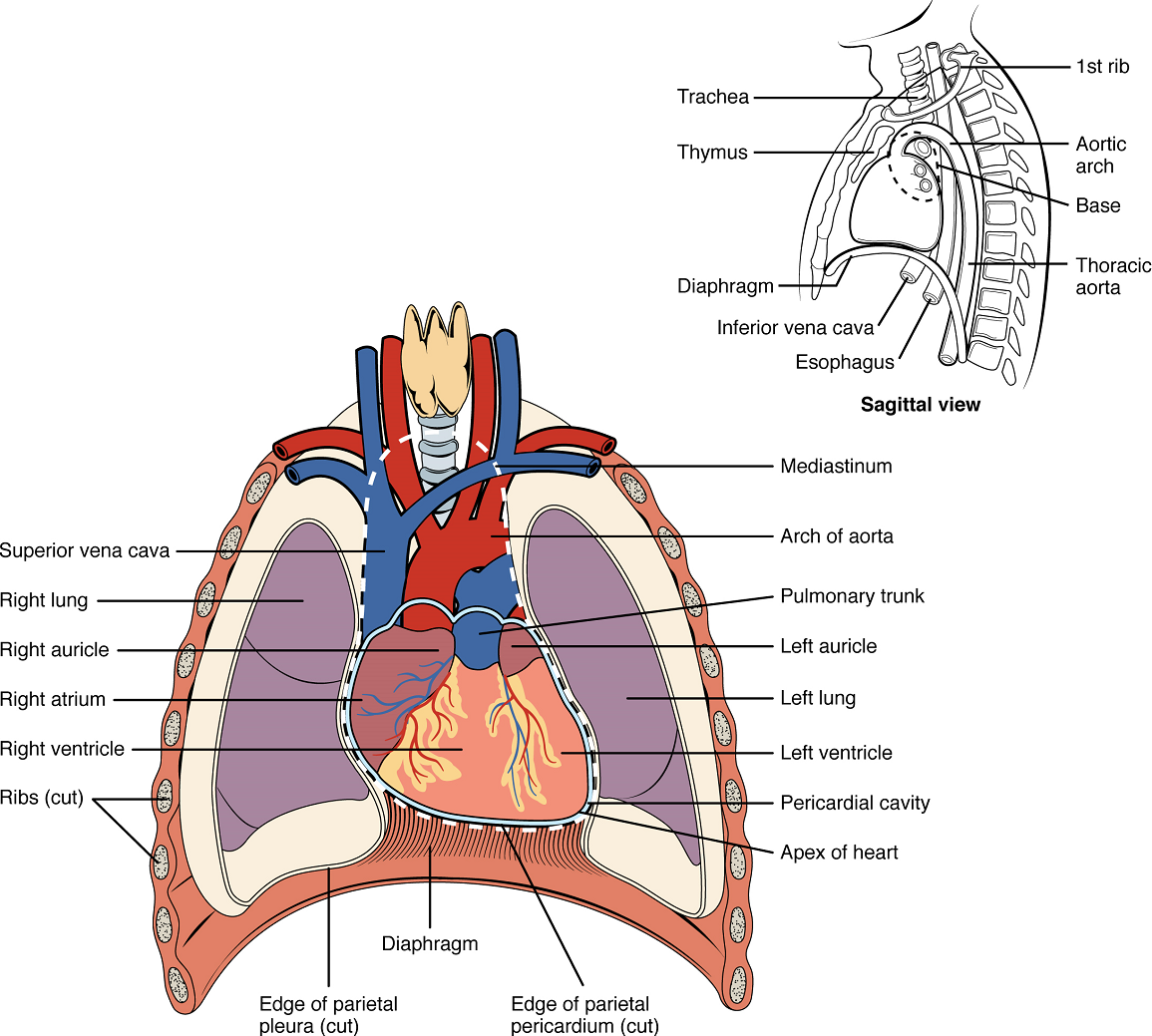
Location of the Heart: The human heart is located within the thoracic cavity, medially between the lungs in the space known as the mediastinum (Figure 7). Within the mediastinum, the heart is separated from the other mediastinal structures by a tough membrane known as the pericardium, or pericardial sac, and sits in its own space called the pericardial cavity. The dorsal surface of the heart lies near the bodies of the vertebrae, and its anterior surface sits deep to the sternum and costal cartilages. The great veins, the superior and inferior venae cavae, and the great arteries, the aorta and pulmonary trunk, are attached to the superior surface of the heart, called the base. The base of the heart is located at the level of the third costal cartilage (Figure 7). The inferior tip of the heart, the apex, lies just to the left of the sternum between the junction of the fourth and fifth ribs near their articulation with the costal cartilages. The right side of the heart is deflected anteriorly, and the left side is deflected posteriorly. It is important to remember the position and orientation of the heart when placing a stethoscope on the chest of a patient and listening for heart sounds, and also when looking at images taken from a midsagittal perspective. The slight deviation of the apex to the left is reflected in a depression in the medial surface of the inferior lobe of the left lung, called the cardiac notch.
Shape and Size of the Heart: The shape of the heart is similar to a pinecone, rather broad at the superior surface and tapering to the apex (Figure 7). A typical heart is approximately the size of your fist: 12 cm (5 in) in length, 8 cm (3.5 in) wide, and 6 cm (2.5 in) in thickness. Given the size difference between most members of the sexes, the weight of a female heart is approximately 250–300 grams (9 to 11 ounces), and the weight of a male heart is approximately 300–350 grams (11 to 12 ounces). The heart of a well-trained athlete, especially one specializing in aerobic sports, can be considerably larger than this. Cardiac muscle responds to exercise in a manner similar to that of skeletal muscle. That is, exercise results in the addition of protein myofilaments that increase the size of the individual cells without increasing their numbers, a concept called hypertrophy. Hearts of athletes can pump blood more effectively at lower rates than those of nonathletes.
Chambers and Circulation through the Heart: The human heart consists of four chambers: The left side and the right side each have one atrium and one ventricle. Each of the upper chambers, the right atrium (plural = atria) and the left atrium, acts as a receiving chamber and contracts to push blood into the lower chambers, the right ventricle and the left ventricle, respectively. The ventricles serve as the primary pumping chambers of the heart, propelling blood to the lungs or to the rest of the body.
There are two distinct but linked circuits in the human circulation called the pulmonary and systemic circuits. Although both circuits transport blood and everything it carries, we can initially view the circuits from the point of view of gases. The pulmonary circuit transports blood to and from the lungs, where it picks up oxygen and delivers carbon dioxide for exhalation. The systemic circuit transports oxygenated blood to virtually all of the tissues of the body and returns relatively deoxygenated blood and carbon dioxide to the heart to be sent back to the pulmonary circulation.
The right ventricle pumps deoxygenated blood into the pulmonary trunk, which leads toward the lungs and bifurcates into the left and right pulmonary arteries. These vessels in turn branch many times before reaching the pulmonary capillaries, where gas exchange occurs: Carbon dioxide exits the blood and oxygen enters. The pulmonary trunk arteries and their branches are the only arteries in the post-natal body that carry relatively deoxygenated blood. Highly oxygenated blood returning from the pulmonary capillaries in the lungs passes through a series of vessels that join together to form the pulmonary veins—the only post-natal veins in the body that carry highly oxygenated blood. The pulmonary veins conduct blood into the left atrium, which pumps the blood into the left ventricle, which in turn pumps oxygenated blood into the aorta and on to the many branches of the systemic circuit. Eventually, these vessels will lead to the systemic capillaries, where exchange with the tissue fluid and cells of the body occurs. In this case, oxygen and nutrients exit the systemic capillaries to be used by the cells in their metabolic processes, and carbon dioxide and waste products will enter the blood.
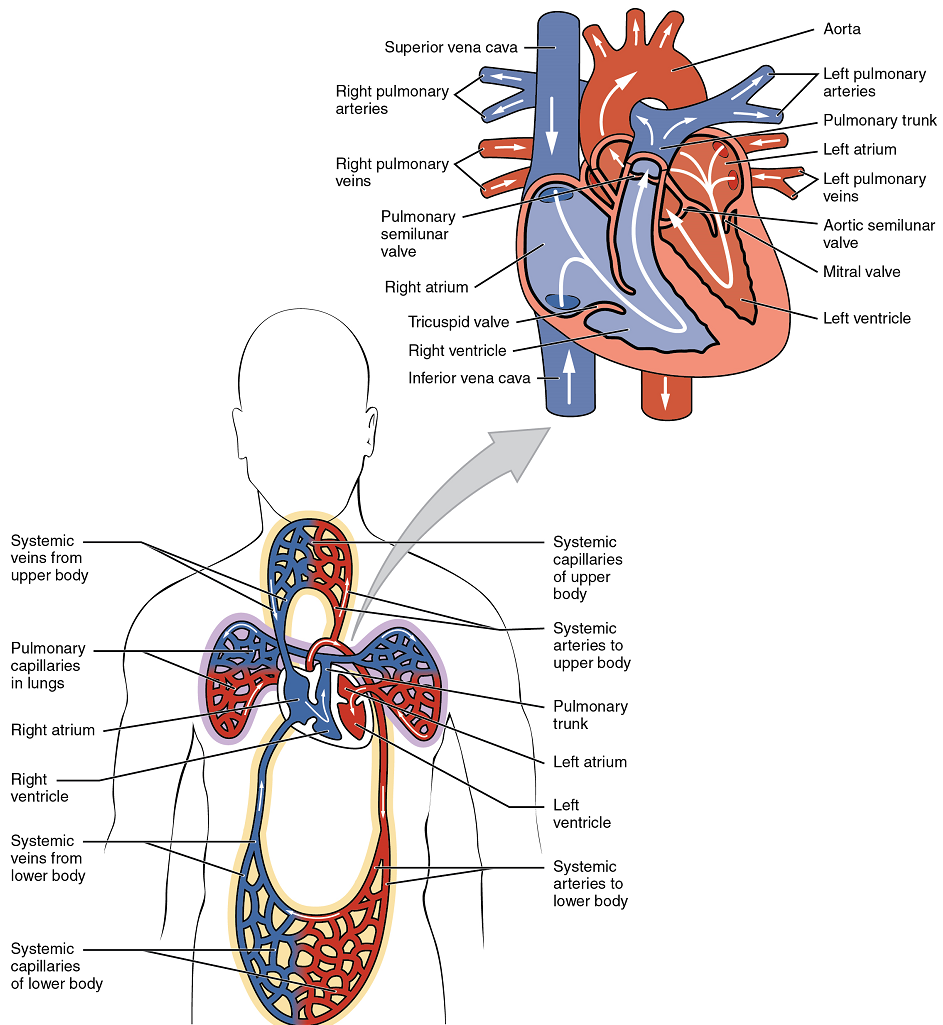
The blood exiting the systemic capillaries is lower in oxygen concentration than when it entered. The capillaries will ultimately unite to form venules, joining to form ever-larger veins, eventually flowing into the two major systemic veins, the superior vena cava and the inferior vena cava, which return blood to the right atrium. The blood in the superior and inferior venae cavae flows into the right atrium, which pumps blood into the right ventricle. This process of blood circulation continues as long as the individual remains alive. Understanding the flow of blood through the pulmonary and systemic circuits is critical to all health professions (Figure 8).
Membranes, Surface Features, and Layers: Our exploration of more in-depth heart structures begins by examining the membrane that surrounds the heart, the prominent surface features of the heart, and the layers that form the wall of the heart. Each of these components plays its own unique role in terms of function.
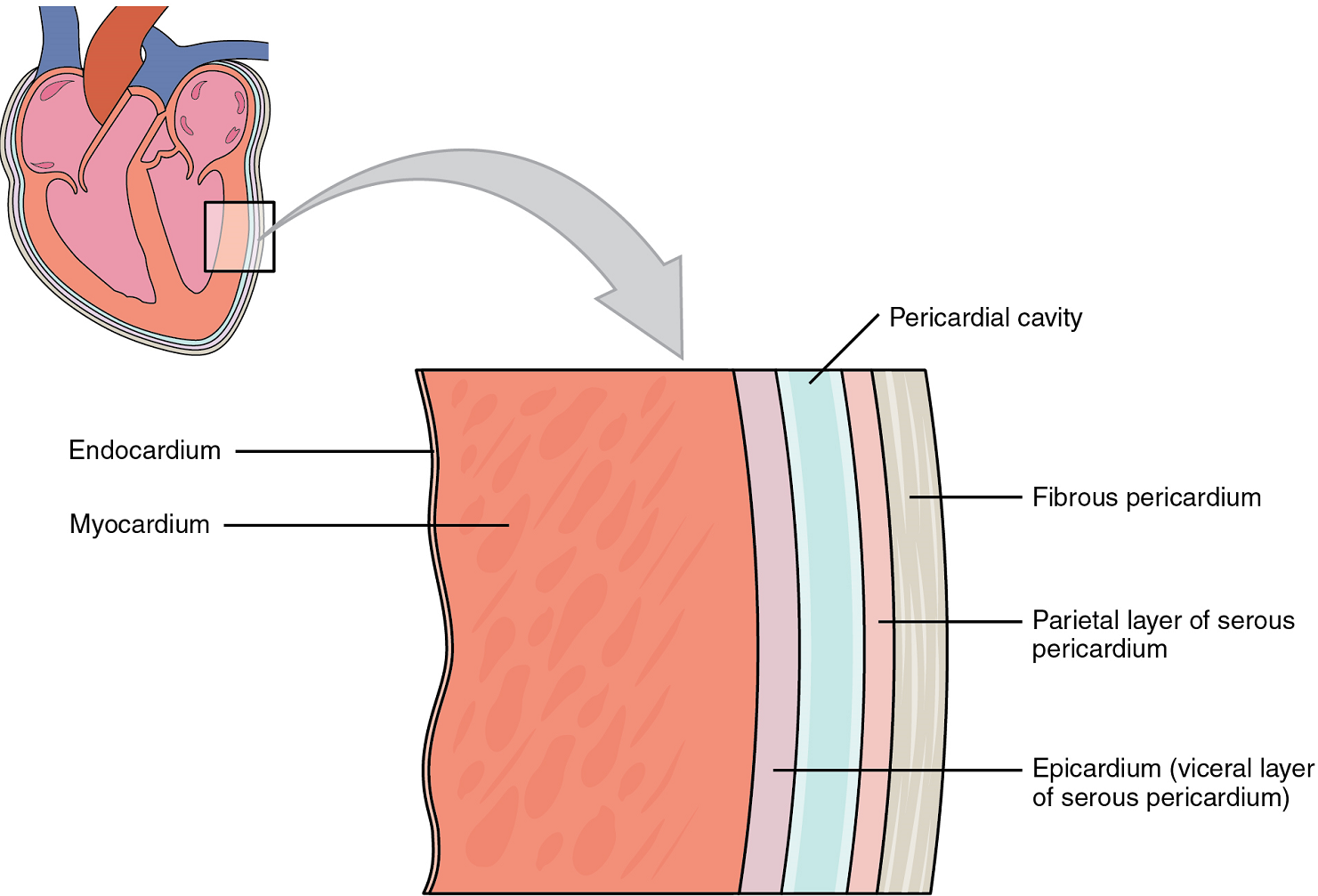
Membranes: The membrane that directly surrounds the heart and defines the pericardial cavity is called the pericardium or pericardial sac. It also surrounds the “roots” of the major vessels, or the areas of closest proximity to the heart. The pericardium, which literally translates as “around the heart,” consists of two distinct sublayers. The sturdy outer layer is the fibrous pericardium, made of tough, dense connective tissue that protects the heart and maintains its position in the thorax. The inner serous pericardium consists of two layers: the outer parietal pericardium, which is fused to the fibrous pericardium, and an inner visceral pericardium, or epicardium, which is fused to the heart and is part of the heart wall. The pericardial cavity, filled with lubricating serous fluid, lies between the epicardium and the pericardium.
In most organs within the body, visceral serous membranes such as the epicardium are microscopic. However, in the case of the heart, it is not a microscopic layer but rather a macroscopic layer, consisting of a simple squamous epithelium called a mesothelium, reinforced with loose, irregular, or areolar connective tissue that attaches to the pericardium (Figure 9). This mesothelium secretes the lubricating serous fluid that fills the pericardial cavity and reduces friction as the heart contracts.
Surface Features of the Heart: Inside the pericardium, the surface features of the heart are visible, including the four chambers (Figure 10). There is a superficial leaf-like extension of the atria near the superior surface of the heart, one on each side, called an auricle—a name that means “ear like”—because its shape resembles the external ear of a human. Auricles are relatively thin-walled structures that can fill with blood and empty into the atria or upper chambers of the heart. You may also hear them referred to as atrial appendages.
Layers: The wall of the heart is composed of three layers of unequal thickness. From superficial to deep, these are the epicardium, the myocardium, and the endocardium (Figure 9). The outermost layer of the wall of the heart is also the innermost layer of the pericardium, the epicardium, or the visceral pericardium discussed earlier.
The middle and thickest layer is the myocardium, made largely of cardiac muscle cells. It is built upon a framework of collagenous fibers, plus the blood vessels that supply the myocardium and the nerve fibers that help regulate the heart. It is the contraction of the myocardium that pumps blood through the heart and into the major arteries. The muscle pattern is elegant and complex, as the muscle cells swirl and spiral around the chambers of the heart (Figure 11). They form a figure 8 pattern around the atria and around the bases of the great vessels. Deeper ventricular muscles also form a figure 8 around the two ventricles and proceed toward the apex. More superficial layers of ventricular muscle wrap around both ventricles. This complex swirling pattern allows the heart to pump blood more effectively than a simple linear pattern would.
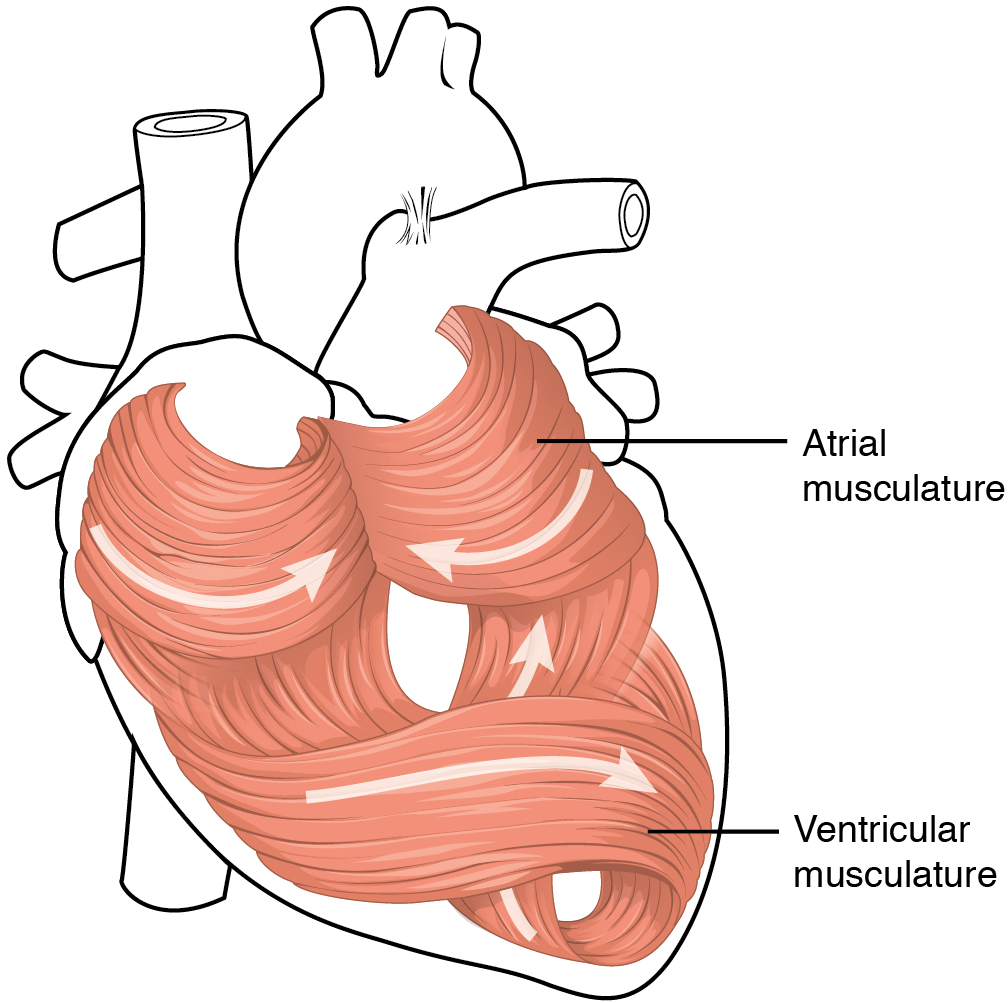
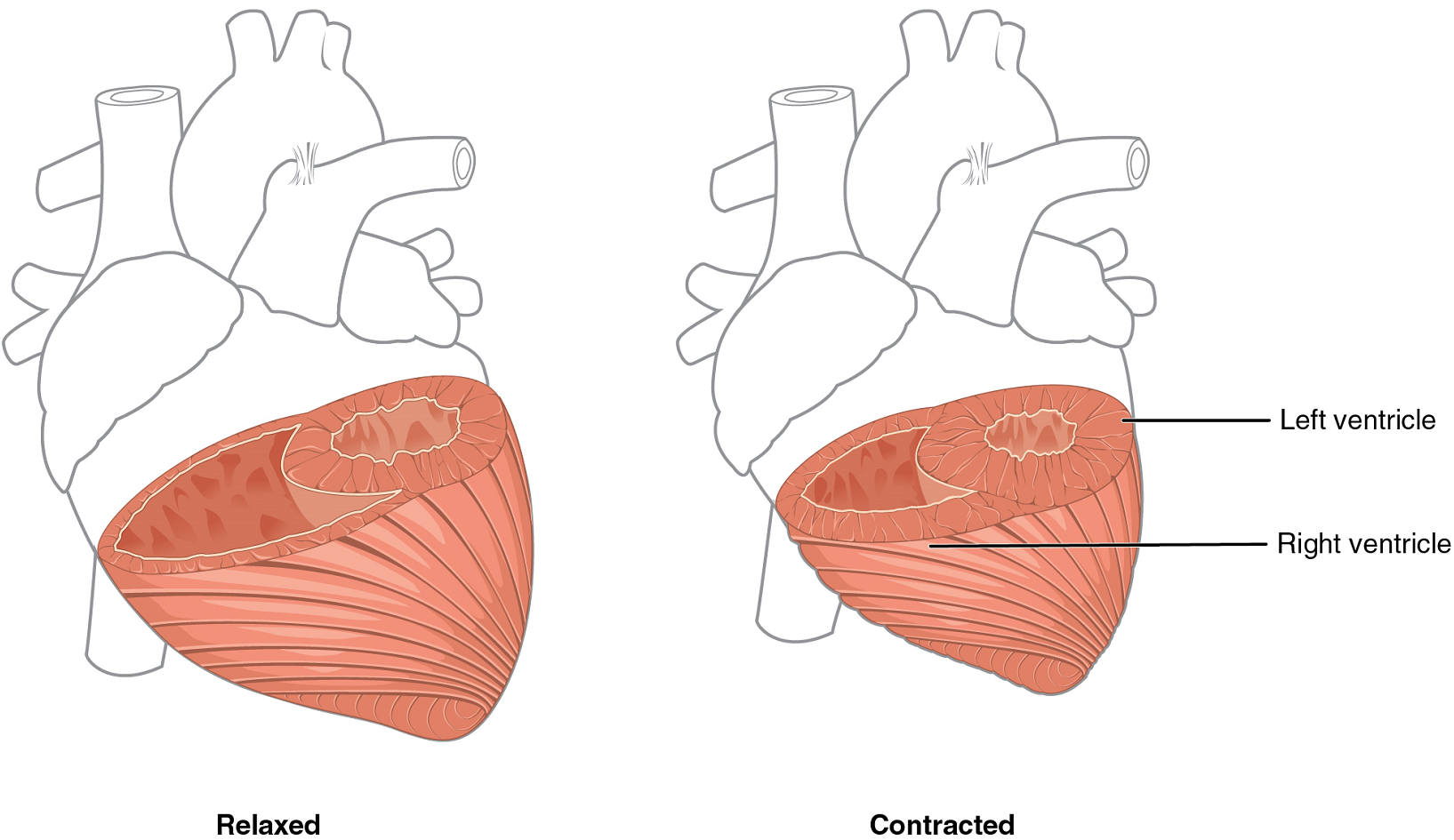
Although the ventricles on the right and left sides pump the same amount of blood per contraction, the muscle of the left ventricle is much thicker and better developed than that of the right ventricle (Figure 12). In order to overcome the high resistance required to pump blood into the long systemic circuit, the left ventricle must generate a great amount of pressure. The right ventricle does not need to generate as much pressure, since the pulmonary circuit is shorter and provides less resistance.
The innermost layer of the heart wall, the endocardium, is joined to the myocardium with a thin layer of connective tissue. The endocardium lines the chambers where the blood circulates and covers the heart valves. It is made of simple squamous epithelium called endothelium, which is continuous with the endothelial lining of the blood vessels (Figure 9).
Once regarded as a simple lining layer, recent evidence indicates that the endothelium of the endocardium and the coronary capillaries may play active roles in regulating the contraction of the muscle within the myocardium.
Internal Structure of the Heart: Recall that the heart’s contraction cycle follows a dual pattern of circulation—the pulmonary and systemic circuits—because of the pairs of chambers that pump blood into the circulation. In order to develop a more precise understanding of cardiac function, it is first necessary to explore the internal anatomical structures in more detail.
Septa of the Heart: The word septum is derived from the Latin for “something that encloses;” in this case, a septum (plural = septa) refers to a wall or partition that divides the heart into chambers. The septa are physical extensions of the myocardium lined with endocardium. Located between the two atria is the interatrial septum. Normally in an adult heart, the interatrial septum bears an oval-shaped depression known as the fossa ovalis, a remnant of an opening in the fetal heart known as the foramen ovale. The foramen ovale allowed blood in the fetal heart to pass directly from the right atrium to the left atrium, allowing some blood to bypass the pulmonary circuit. Within seconds after birth, a flap of tissue known as the septum primum that previously acted as a valve closes the foramen ovale and establishes the typical cardiac circulation pattern.
Between the two ventricles is a second septum known as the interventricular septum (Figure 13). Unlike the interatrial septum, the interventricular septum is normally intact after its formation during fetal development. It is substantially thicker than the interatrial septum, since the ventricles generate far greater pressure when they contract.
The septum between the atria and ventricles is known as the atrioventricular septum. It is marked by the presence of four openings that allow blood to move from the atria into the ventricles and from the ventricles into the pulmonary trunk and aorta. Located in each of these openings between the atria and ventricles is a valve, a specialized structure that ensures one-way flow of blood. The valves between the atria and ventricles are known generically as atrioventricular valves. The valves at the openings that lead to the pulmonary trunk and aorta are known generically as semilunar valves. Since these openings and valves structurally weaken the atrioventricular septum, the remaining tissue is heavily reinforced with dense connective tissue called the cardiac skeleton, or skeleton of the heart. It includes four rings that surround the openings between the atria and ventricles, and the openings to the pulmonary trunk and aorta, and serve as the point of attachment for the valves of the heart. The cardiac skeleton also provides an important boundary in the heart electrical conduction system.
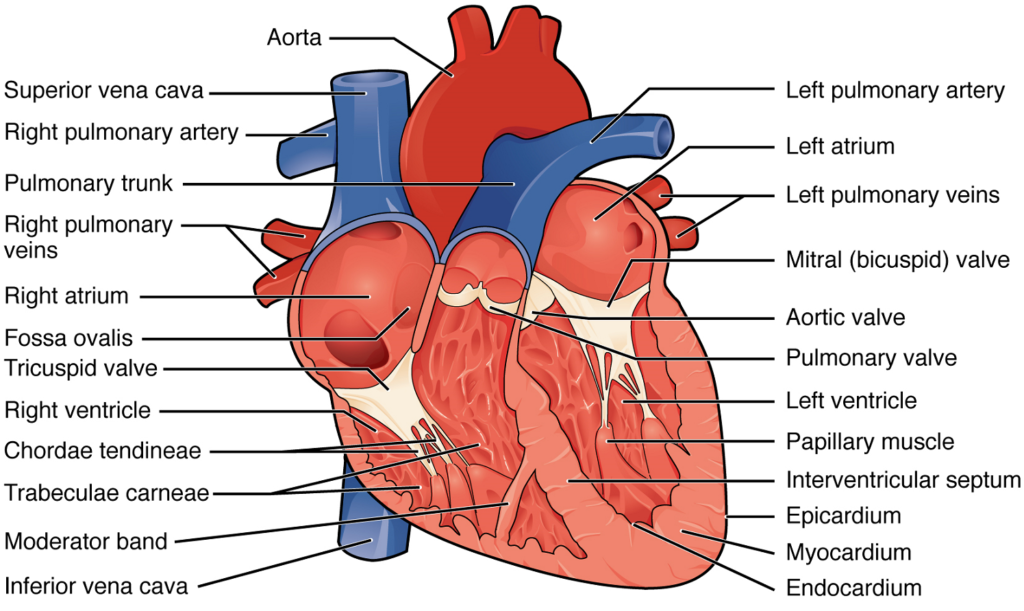
Right Atrium: The right atrium serves as the receiving chamber for blood returning to the heart from the systemic circulation (Figure 13). The two major systemic veins, the superior and inferior venae cavae, and the large coronary vein called the coronary sinus that drains the heart myocardium empty into the right atrium (Figure 18). The superior vena cava drains blood from regions superior to the diaphragm: the head, neck, upper limbs, and the thoracic region. It empties into the superior and posterior portions of the right atrium. The inferior vena cava drains blood from areas inferior to the diaphragm: the lower limbs and abdominopelvic region of the body. It, too, empties into the posterior portion of the atria, but inferior to the opening of the superior vena cava. Immediately superior and slightly medial to the opening of the inferior vena cava on the posterior surface of the atrium is the opening of the coronary sinus. This thin-walled vessel drains most of the coronary veins that return systemic blood from the heart.
The atria receive venous blood on a nearly continuous basis, preventing venous flow from stopping while the ventricles are contracting. While most ventricular filling occurs while the atria are relaxed, they do demonstrate a contractile phase and actively pump blood into the ventricles just prior to ventricular contraction. The opening between the atrium and ventricle is guarded by the tricuspid valve.
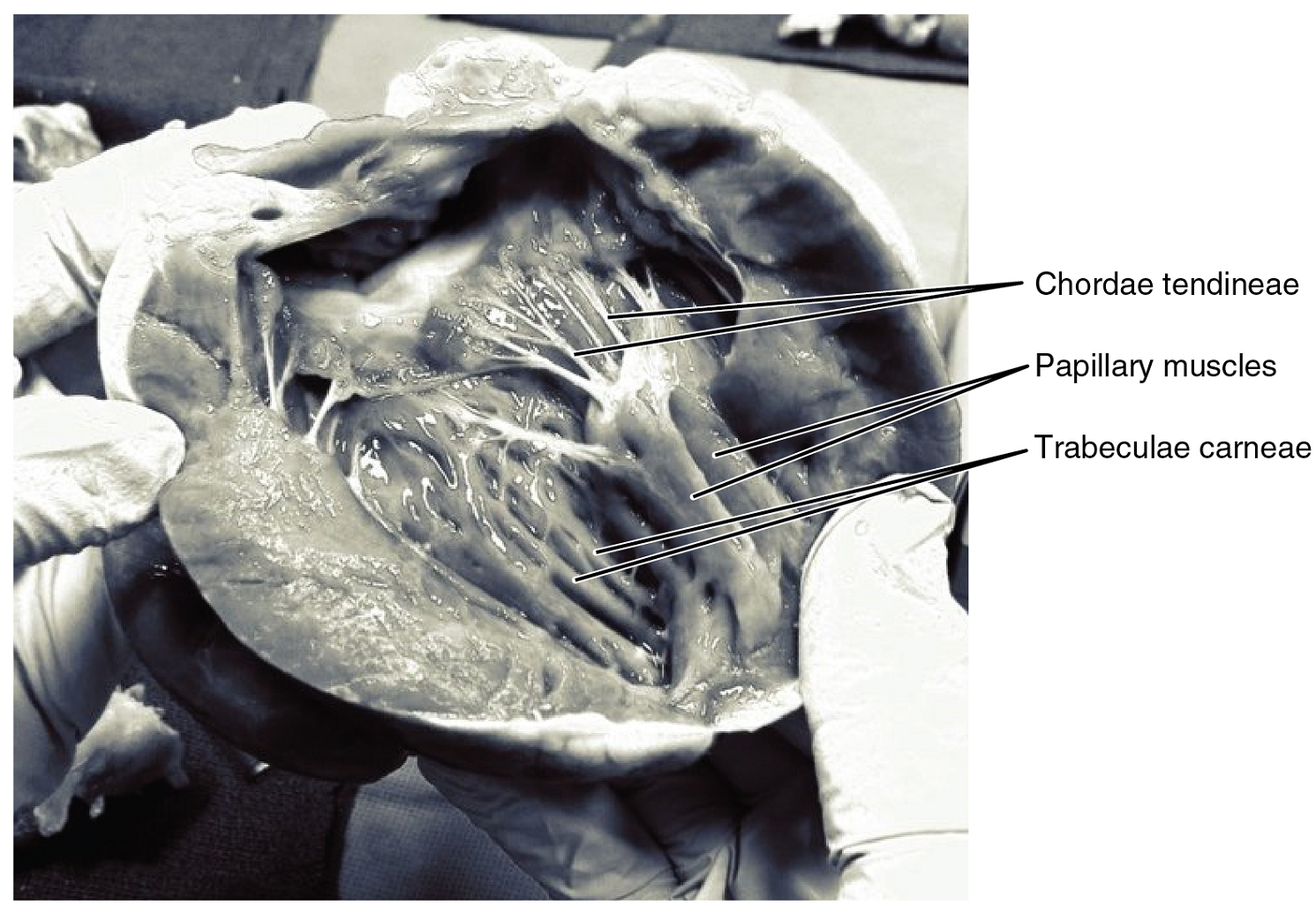
Right Ventricle: The right ventricle receives blood from the right atrium through the tricuspid valve (Figure 13). Each flap of the valve is attached to strong strands of connective tissue, the chordae tendineae, literally “tendinous cords,” or sometimes more poetically referred to as “heart strings” (Figure 14). There are several chordae tendineae associated with each of the flaps. They are composed of approximately 80% collagenous fibers with the remainder consisting of elastic fibers and endothelium. They connect each of the flaps to a papillary muscle that extends from the inferior ventricular surface (Figure 14). There are three papillary muscles in the right ventricle, called the anterior, posterior, and septal muscles, which correspond to the three sections of the valves.
When the myocardium of the ventricle contracts, pressure within the ventricular chamber rises. Blood, like any fluid, flows from higher pressure to lower pressure areas, in this case, toward the pulmonary trunk and the atrium. To prevent any potential backflow, the papillary muscles also contract, generating tension on the chordae tendineae. This prevents the flaps of the valves from being forced into the atria and regurgitation of the blood back into the atria during ventricular contraction.
Left Atrium: After exchange of gases in the pulmonary capillaries, blood returns to the left atrium high in oxygen via one of the four pulmonary veins (Figure 13). Blood flows nearly continuously from the pulmonary veins back into the atrium, which acts as the receiving chamber, and from here through an opening into the left ventricle. Most blood flows passively into the heart while both the atria and ventricles are relaxed, but toward the end of the ventricular relaxation period, the left atrium will contract, pumping blood into the ventricle. This atrial contraction accounts for approximately 20% of ventricular filling. The opening between the left atrium and ventricle is guarded by the mitral valve.
Left Ventricle: Recall that, although both sides of the heart will pump the same amount of blood, the muscular layer is much thicker in the left ventricle compared to the right (Figure 13). Like the right ventricle, the left also has trabeculae carneae. The mitral valve is connected to papillary muscles via chordae tendineae. There are two papillary muscles on the left – the anterior and posterior – as opposed to three on the right.
The left ventricle is the major pumping chamber for the systemic circuit; it ejects blood into the aorta through the aortic semilunar valve.
Heart Valve Structure and Function: A transverse section through the heart slightly above the level of the atrioventricular septum reveals all four heart valves along the same plane (Figure 15). The valves ensure unidirectional blood flow through the heart. Between the right atrium and the right ventricle is the right atrioventricular valve, or tricuspid valve. It typically consists of three flaps, or leaflets, made of endocardium reinforced with additional connective tissue. The flaps are connected by chordae tendineae to the papillary muscles, which control the opening and closing of the valves.
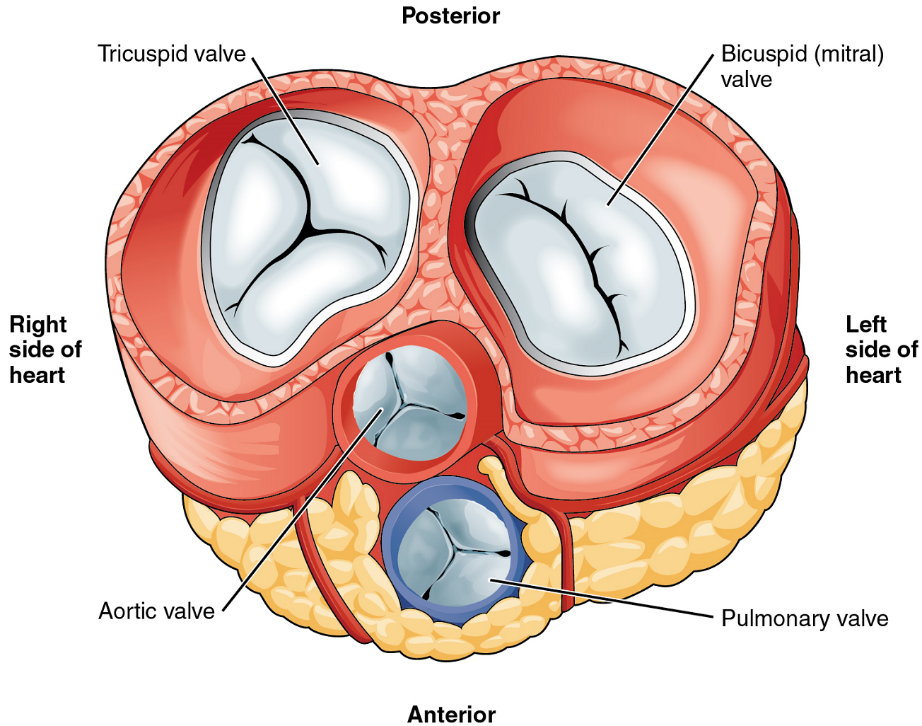
Emerging from the right ventricle at the base of the pulmonary trunk is the pulmonary semilunar valve, or the pulmonary valve; it is also known as the pulmonic valve or the right semilunar valve. The pulmonary valve is comprised of three small flaps of endothelium reinforced with connective tissue. When the ventricle relaxes, the pressure differential causes blood to flow back into the ventricle from the pulmonary trunk. This flow of blood fills the pocket-like flaps of the pulmonary valve, causing the valve to close and producing an audible sound. Unlike the atrioventricular valves, there are no papillary muscles or chordae tendineae associated with the pulmonary valve.
Located at the opening between the left atrium and left ventricle is the mitral valve, also called the bicuspid valve or the left atrioventricular valve. Structurally, this valve consists of two cusps, known as the anterior medial cusp and the posterior medial cusp, compared to the three cusps of the tricuspid valve. In a clinical setting, the valve is referred to as the mitral valve, rather than the bicuspid valve. The two cusps of the mitral valve are attached by chordae tendineae to two papillary muscles that project from the wall of the ventricle.
At the base of the aorta is the aortic semilunar valve, or the aortic valve, which prevents backflow from the aorta. It normally is composed of three flaps. When the ventricle relaxes and blood attempts to flow back into the ventricle from the aorta, blood will fill the cusps of the valve, causing it to close and producing an audible sound.
When both atria and ventricles are relaxed, and when the atria contract to pump blood into the ventricles, the atrioventricular valves are open and the semilunar valves are closed (Figure 16).
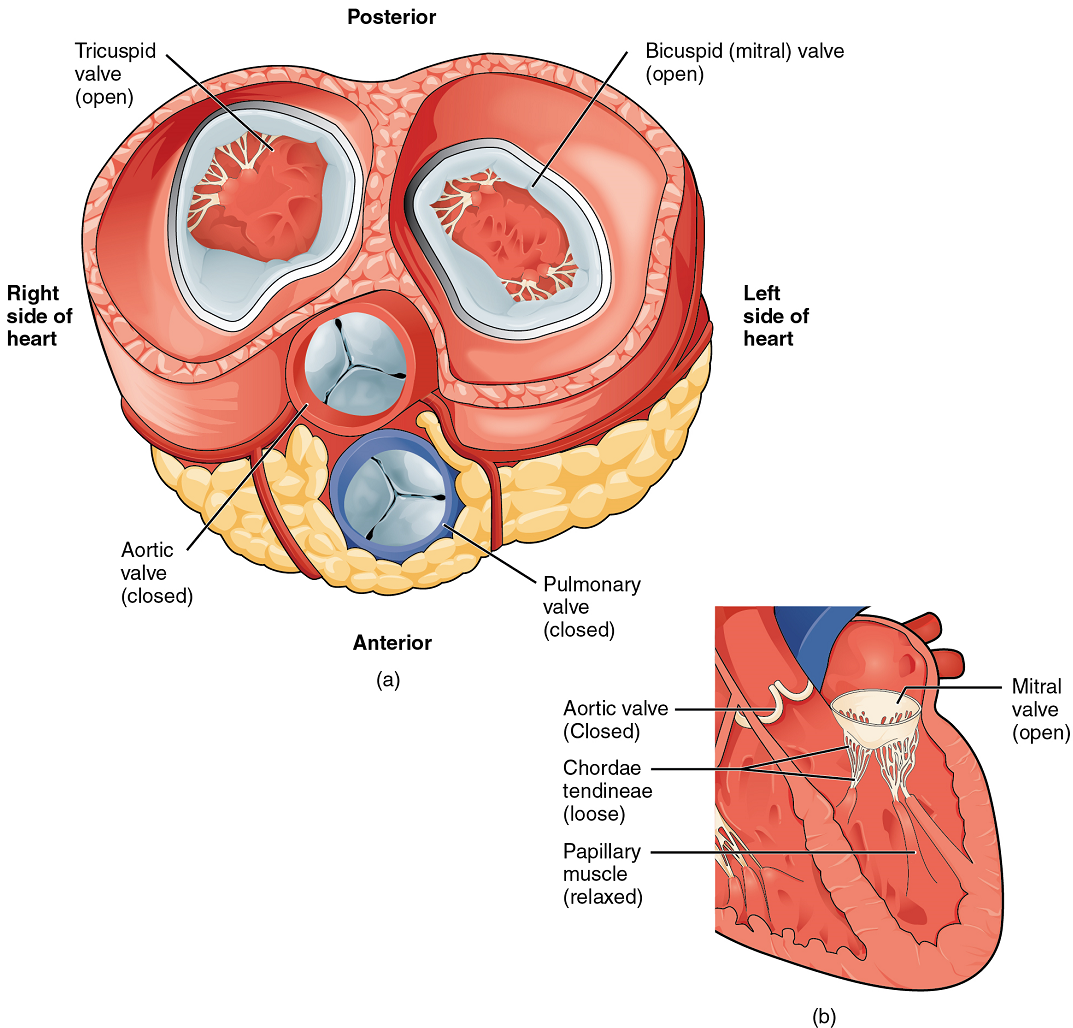
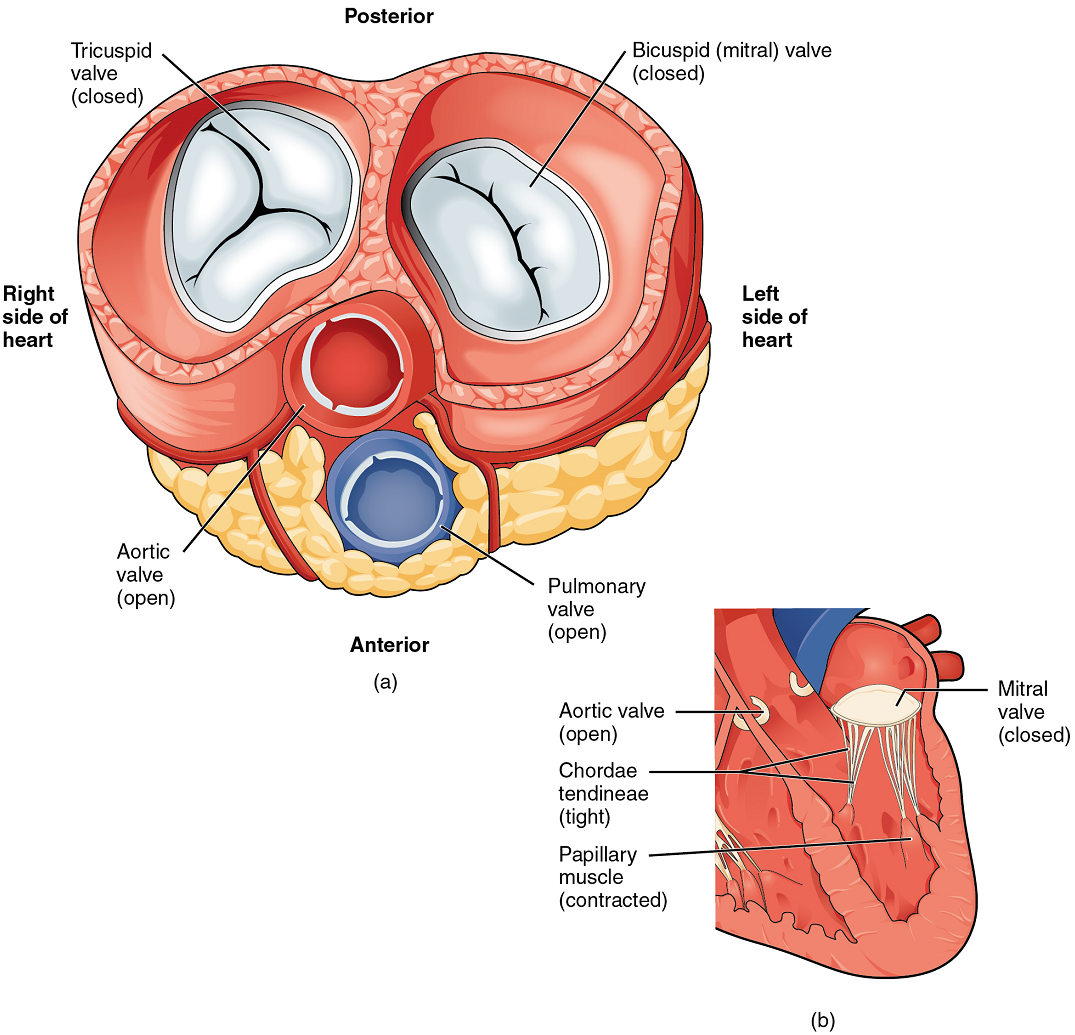
When the ventricles contract to eject blood into the pulmonary trunk and aorta, the atrioventricular valves close and the two semilunar valves open (Figure 17). Closure of the two atrioventricular valves prevents blood from being forced back into the atria.
When the ventricles begin to contract, pressure within the ventricles rises and blood flows toward the area of lowest pressure, which is initially in the atria. This backflow causes the cusps of the tricuspid and mitral (bicuspid) valves to close. These valves are tied down to the papillary muscles by chordae tendineae. During the relaxation phase of the cardiac cycle, the papillary muscles are also relaxed and the tension on the chordae tendineae is slight (Figure 16b). However, as the myocardium of the ventricle contracts, so do the papillary muscles. This creates tension on the chordae tendineae (Figure 17b), helping to hold the cusps of the atrioventricular valves in place and preventing them from being blown back into the atria.
The aortic and pulmonary semilunar valves lack the chordae tendineae and papillary muscles associated with the atrioventricular valves. Instead, they consist of pocket-like folds of endocardium reinforced with additional connective tissue. When the ventricles relax and the change in pressure forces the blood toward the ventricles, the blood presses against these cusps and seals the openings.
Coronary Circulation: You will recall that the heart is a remarkable pump composed largely of cardiac muscle cells that are incredibly active throughout life. Like all other cells, a cardiomyocyte requires a reliable supply of oxygen and nutrients, and a way to remove wastes, so it needs a dedicated, complex, and extensive coronary circulation. And because of the critical and nearly ceaseless activity of the heart throughout life, this need for a blood supply is even greater than for a typical cell. However, coronary circulation is not continuous; rather, it cycles, reaching a peak when the heart muscle is relaxed and nearly ceasing while it is contracting.
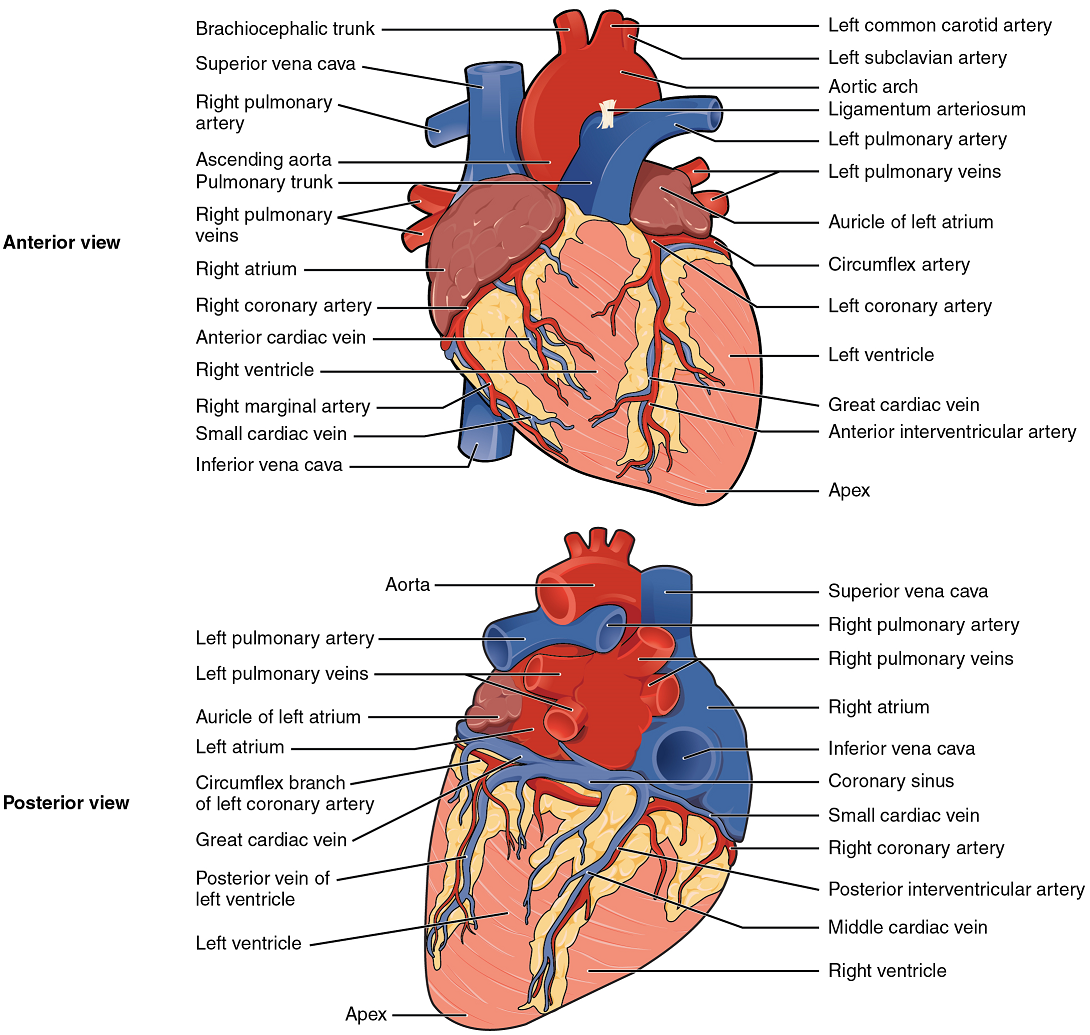
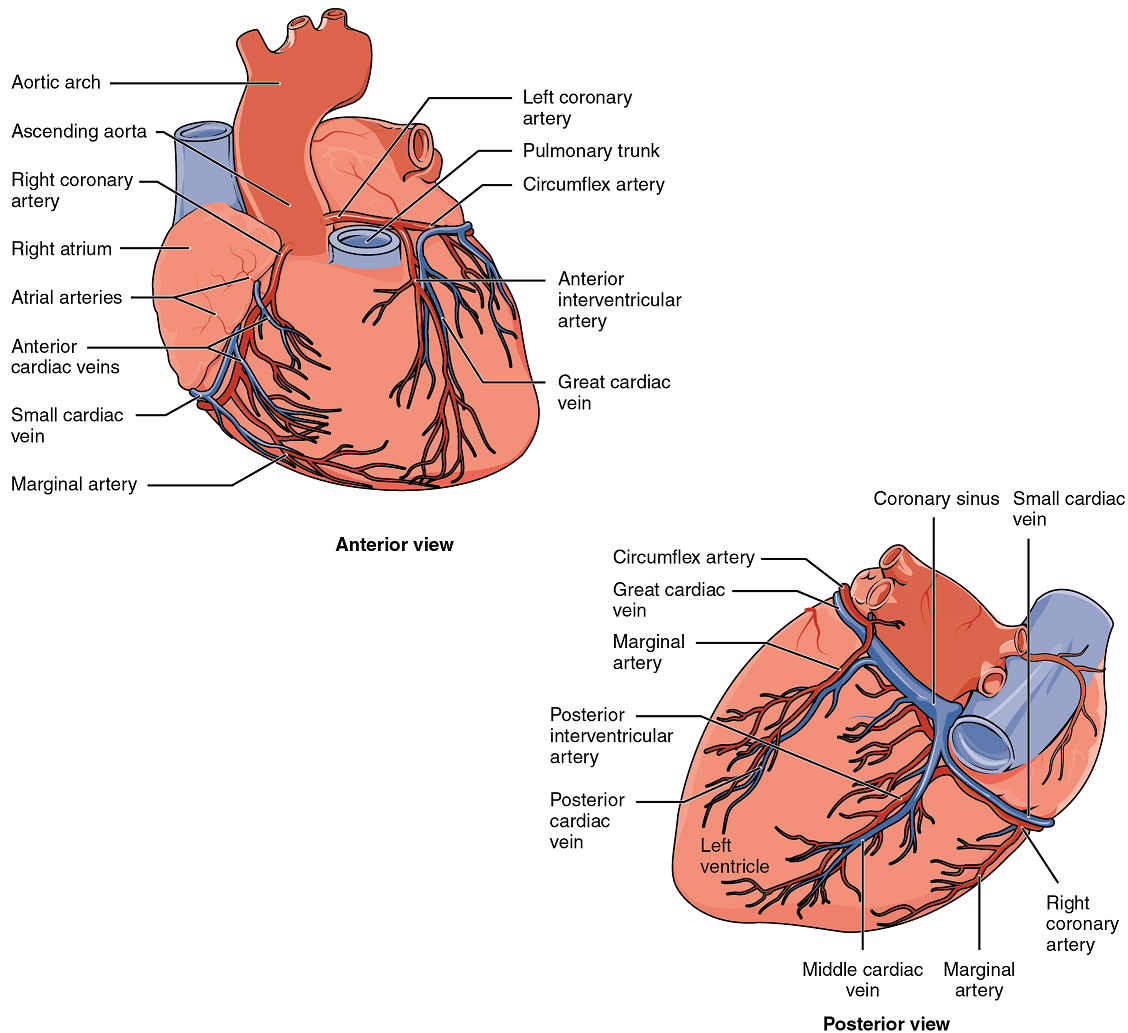
Coronary Arteries: Coronary arteries supply blood to the myocardium and other components of the heart. The first portion of the aorta after it arises from the left ventricle gives rise to the coronary arteries (Figure 18).
The left coronary artery distributes blood to the left side of the heart, the left atrium and ventricle, and the interventricular septum. The circumflex artery arises from the left coronary artery and follows the coronary sulcus to the left anterior descending artery (LAD). A coronary artery blockage often results in death of the cells (myocardial infarction) supplied by the particular vessel.
The right coronary artery proceeds along the coronary sulcus and distributes blood to the right atrium, portions of both ventricles, and the heart conduction system (Figure 19).
Coronary Veins: Coronary veins drain the heart and generally parallel the large surface arteries (Figure 18). Most drain into the coronary sinus. The coronary sinus is a large, thin-walled vein on the posterior surface of the heart lying within the coronary sulcus and emptying directly into the right atrium.

Part 2: Cardiac Muscle and Electrical Activity
Recall that cardiac muscle shares a few characteristics with both skeletal muscle and smooth muscle, but it has some unique properties of its own. Not the least of these exceptional properties is its ability to initiate an electrical potential at a fixed rate that spreads rapidly from cell to cell to trigger the contractile mechanism. This property is known as autorhythmicity. Neither smooth nor skeletal muscle can do this. Even though cardiac muscle has autorhythmicity, heart rate is modulated by the endocrine and nervous systems.
There are two major types of cardiac muscle cells: myocardial contractile cells and myocardial conducting cells. The myocardial contractile cells constitute the bulk (99%) of the cells in the atria and ventricles. Contractile cells conduct impulses and are responsible for contractions that pump blood through the body. The myocardial conducting cells (1% of the cells) form the conduction system of the heart. Except for Purkinje fibers, they are generally much smaller than the contractile cells and have few of the myofibrils or filaments needed for contraction. Their function is similar in many respects to neurons, although they are specialized muscle cells. Myocardial conduction cells initiate and propagate the action potential (the electrical impulse) that travels throughout the heart and triggers the contractions that propel the blood.
Conduction System of the Heart: If embryonic heart cells are separated into a Petri dish and kept alive, each is capable of generating its own electrical impulse followed by contraction. When two independently beating embryonic cardiac muscle cells are placed together, the cell with the higher inherent rate sets the pace, and the impulse spreads from the faster to the slower cell to trigger a contraction. As more cells are joined together, the fastest cell continues to assume control of the rate. A fully developed adult heart maintains the capability of generating its own electrical impulse, triggered by the fastest cells, as part of the cardiac conduction system. The components of the cardiac conduction system include the sinoatrial node (SA node), the atrioventricular node (AV node), the atrioventricular bundle (bundle of His), the atrioventricular bundle branches, and the Purkinje fibers (Figure 19).
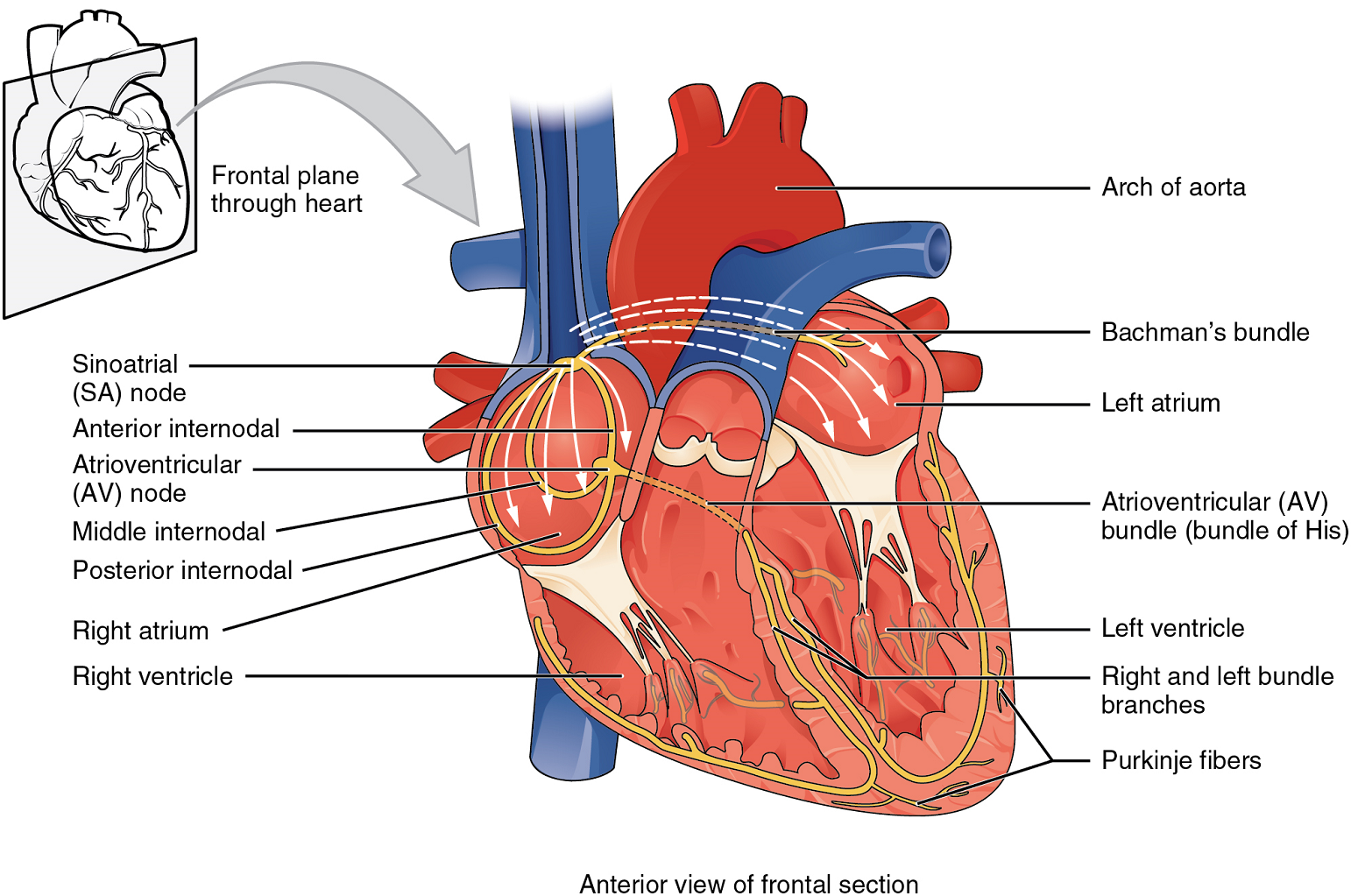
Sinoatrial (SA) Node: Normal cardiac rhythm is established by the sinoatrial (SA) node, a specialized clump of myocardial conducting cells located in the superior and posterior walls of the right atrium in close proximity to the orifice of the superior vena cava. The sinoatrial node has the highest inherent rate of depolarization and is known as the pacemaker of the heart. It initiates the sinus rhythm, or normal electrical pattern followed by contraction of the heart.
This impulse spreads from its initiation in the sinoatrial node throughout the atria to the atrial myocardial contractile cells and the atrioventricular node (Figure 19). The impulse takes approximately 50 ms (milliseconds) to travel between these two nodes. When the impulse reaches the atrioventricular septum, the connective tissue of the cardiac skeleton prevents the impulse from spreading into the myocardial cells in the ventricles except at the atrioventricular node.
The electrical event, the wave of depolarization, is the trigger for muscular contraction. The wave of depolarization begins in the right atrium, and the impulse spreads across the superior portions of both atria and then down through the contractile cells. The contractile cells then begin contraction from the superior to the inferior portions of the atria, efficiently pumping blood into the ventricles.
Atrioventricular (AV) Node: The atrioventricular (AV) node is a second clump of specialized myocardial conductive cells, located in the inferior portion of the right atrium within the atrioventricular septum. The septum prevents the impulse from spreading directly to the ventricles without passing through the atrioventricular node. There is a critical pause before the atrioventricular node depolarizes and transmits the impulse to the atrioventricular bundle (Figure 20).
This delay in transmission is partially attributable to the small diameter of the cells of the node, which slow the impulse. Also, conduction between nodal cells is less efficient than between cardiomyocytes to complete their contraction that pumps blood into the ventricles before the impulse is transmitted to the cells of the ventricle itself. With extreme stimulation by the sinoatrial node, the atrioventricular node can transmit impulses maximally at 220 per minute. This establishes the typical maximum heart rate in a healthy young individual. Damaged hearts or those stimulated by drugs can contract at higher rates, but at these rates, the heart can no longer effectively pump blood. It takes the impulse approximately 100 ms to pass through the atrioventricular node. This pause is critical to heart function, as it allows the atria to empty their blood into the ventricles.
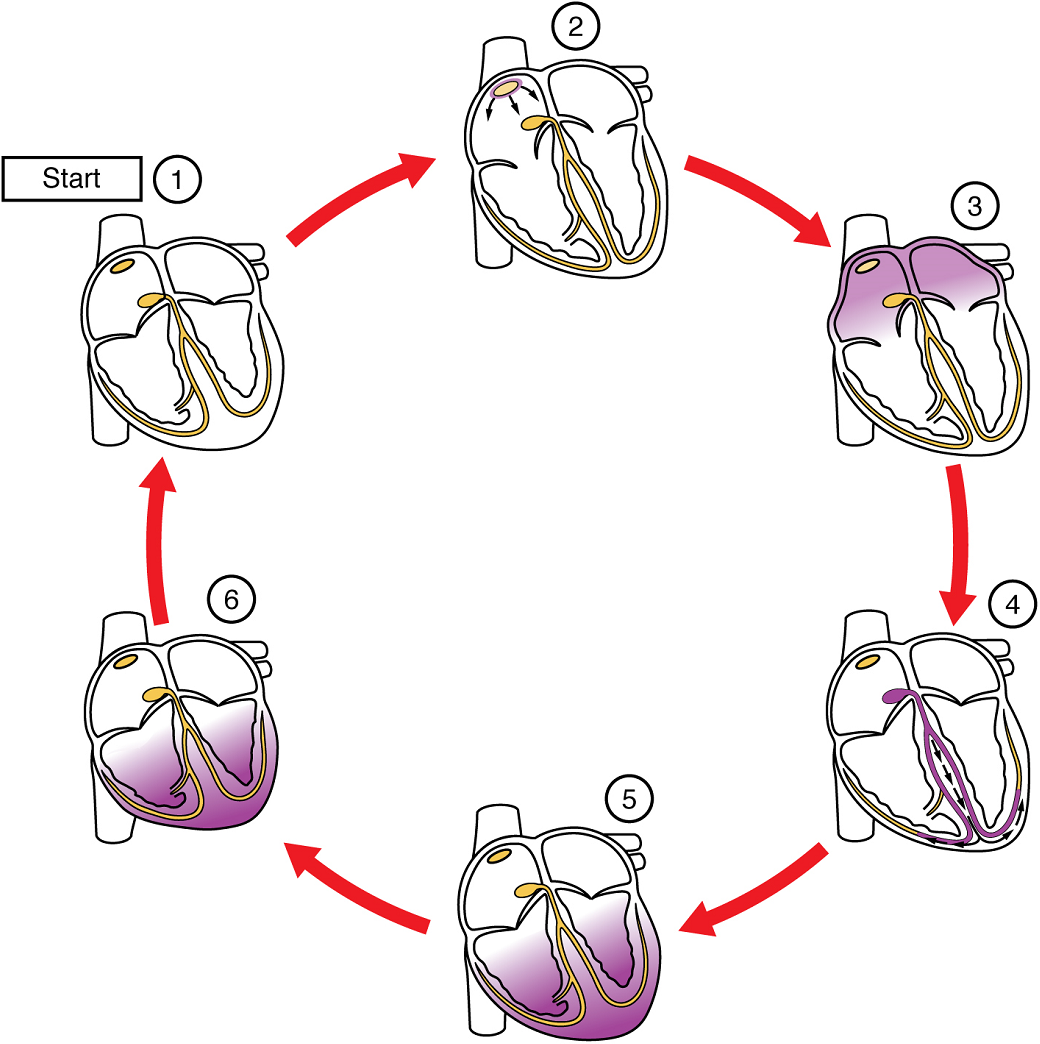
Atrioventricular Bundle (Bundle of His), Bundle Branches, and Purkinje Fibers: Arising from the atrioventricular node, the atrioventricular bundle, or bundle of His, proceeds through the interventricular septum before dividing into two atrioventricular bundle branches, commonly called the left and right bundle branches. The left bundle branch supplies the left ventricle, and the right bundle branch the right ventricle. Both bundle branches descend and reach the apex of the heart where they connect with the Purkinje fibers (Figure 20). This passage takes approximately 25 ms.
The Purkinje fibers are additional myocardial conductive fibers that spread the impulse to the myocardial contractile cells in the ventricles. They extend throughout the myocardium from the apex of the heart toward the atrioventricular septum and the base of the heart. The Purkinje fibers have a fast inherent conduction rate, and the electrical impulse reaches all of the ventricular muscle cells in about 75 ms (Figure 20). Since the electrical stimulus begins at the apex, the contraction also begins at the apex and travels toward the base of the heart, similar to squeezing a tube of toothpaste from the bottom. However, the contraction of the ventricles is asynchronous with the right ventricle contracting slightly ahead of the left ventricle at the apex. This causes a twisting of the ventricles pushing blood towards major vessels leaving the heart. This allows the blood to be pumped out of the ventricles and into the aorta and pulmonary trunk in a more efficient manner. The total time elapsed from the initiation of the impulse in the sinoatrial node until depolarization of the ventricles is approximately 225 ms.
Electrocardiogram
By careful placement of surface electrodes on the body, it is possible to record the complex, compound electrical signal of the heart. This tracing of the electrical signal is the electrocardiogram (ECG), also commonly abbreviated EKG (for “elektrokardiogramm”, the German term for the test). Careful analysis of the ECG reveals a detailed picture of both normal and abnormal heart function, and is an indispensable clinical diagnostic tool. The standard electrocardiograph (the instrument that generates an ECG) uses 3, 5, or 12 leads. The greater the number of leads an electrocardiograph uses, the more information the ECG provides. The term “lead” may be used to refer to the cable from the electrode to the electrical recorder, but it typically describes the voltage difference between two of the electrodes. The 12-lead electrocardiograph uses 10 electrodes placed in standard locations on the patient’s skin (Figure 21).
There are five prominent points on the ECG: the P wave, the QRS complex, and the T wave (Figure 22). The small P wave represents the depolarization of the atria. The atria begin contracting approximately 25 ms after the start of the P wave. The large QRS complex represents the depolarization of the ventricles, which requires a much stronger electrical signal because of the larger size of the ventricular cardiac muscle. The ventricles begin to contract as the QRS reaches the peak of the R wave. Lastly, the T wave represents the repolarization of the ventricles. The repolarization of the atria occurs during the QRS complex, which masks it on an ECG.
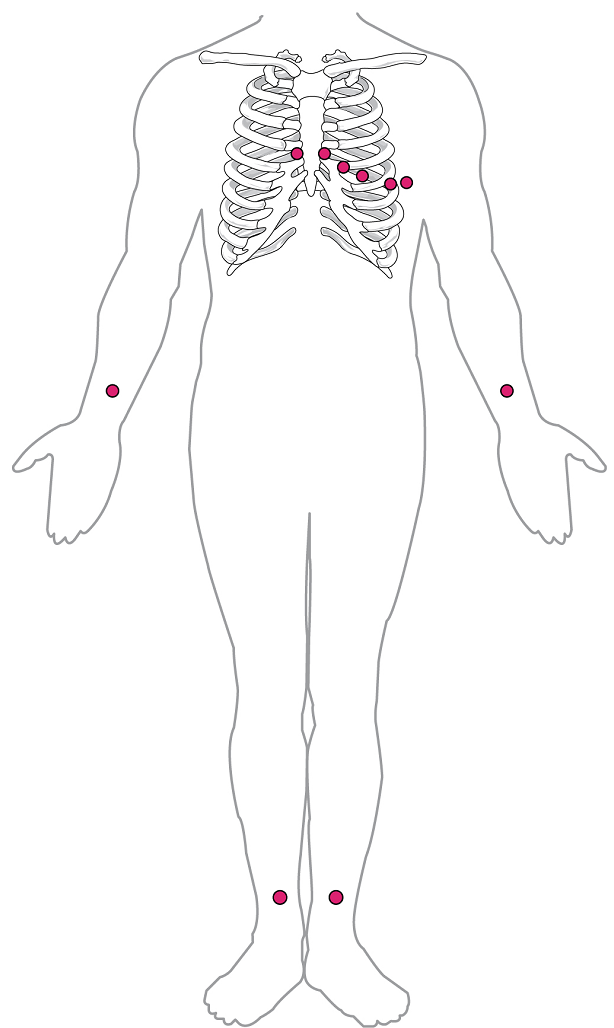
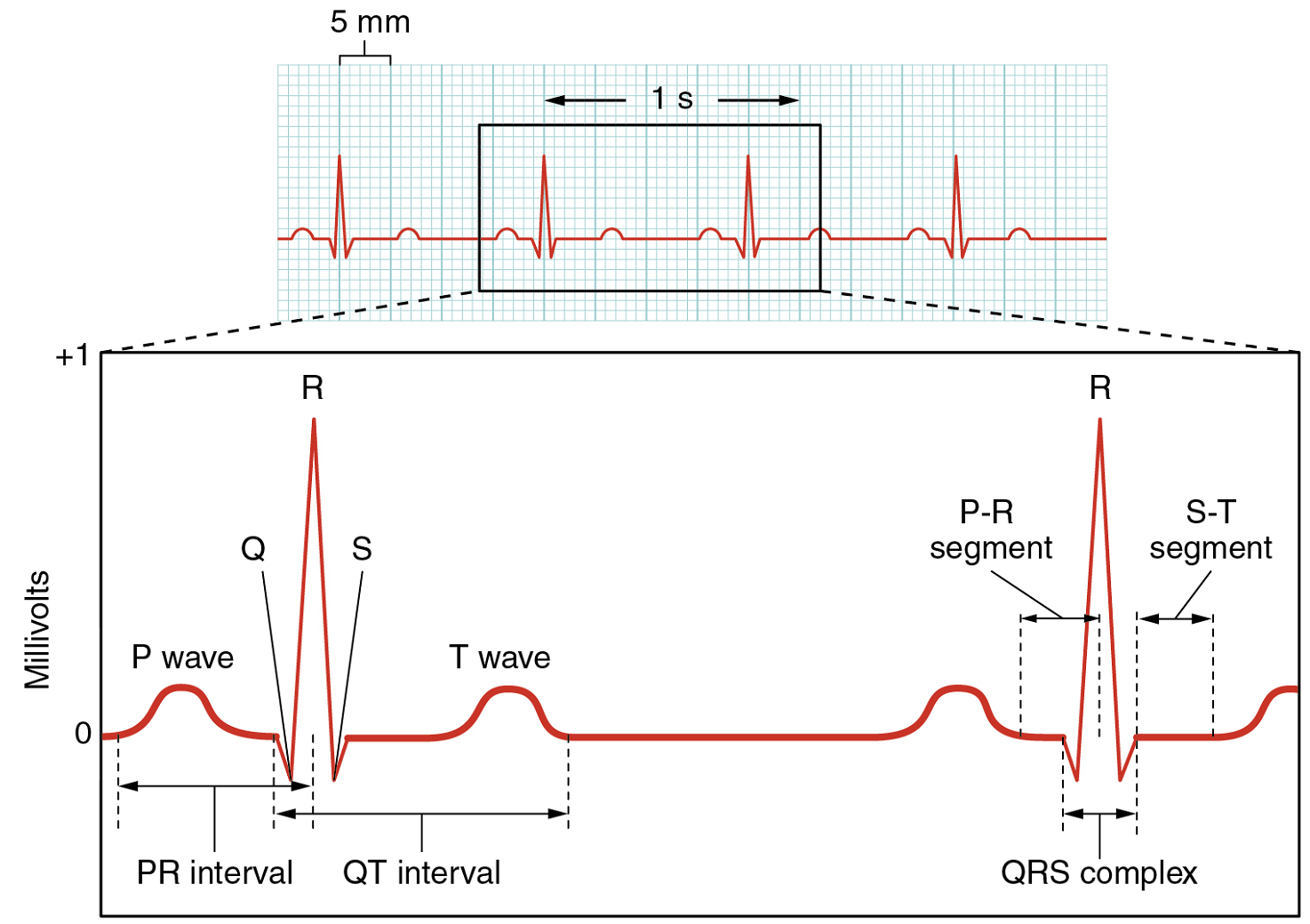
The depolarization events that appear on an ECG tracing should result in contraction of the corresponding chambers (Figure 23). Repolarization events measured then correspond with relaxation of the corresponding chambers.
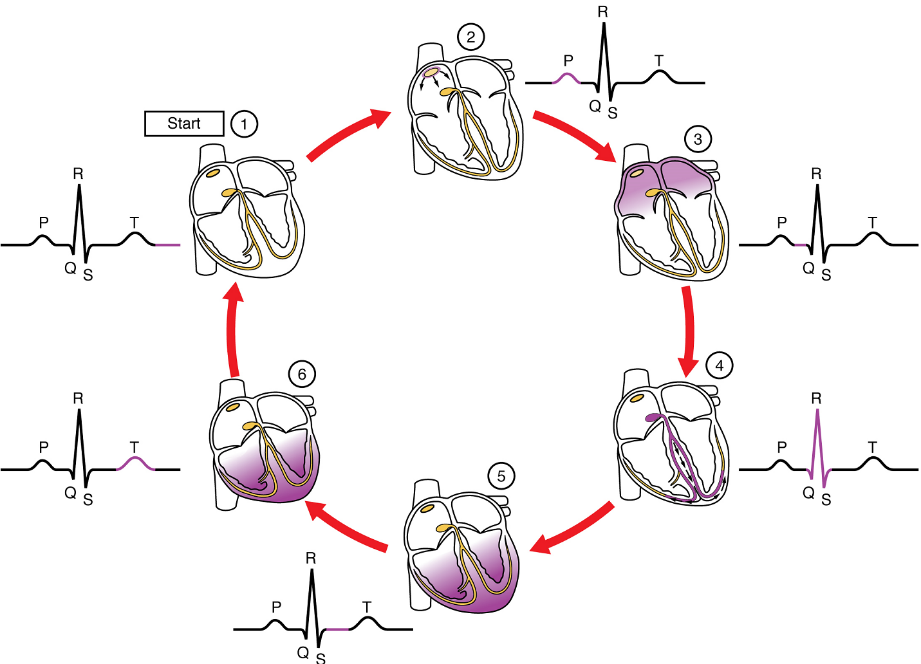
Part 3: Cardiac Cycle
The period of time that begins with contraction of the atria and ends with ventricular relaxation is known as the cardiac cycle (Figure 24). The period of contraction that the heart undergoes while it pumps blood into circulation is called systole. The period of relaxation that occurs as the chambers fill with blood is called diastole. Both the atria and ventricles undergo systole and diastole, and it is essential that these components be carefully regulated and coordinated to ensure blood is pumped efficiently to the body.
Pressures and Flow: Fluids, whether gases or liquids, are materials that flow according to pressure gradients—that is, they move from regions that are higher in pressure to regions that are lower in pressure. Accordingly, when the heart chambers are relaxed (diastole), blood will flow into the atria from the veins, which are higher in pressure. As blood flows into the atria, the pressure will rise, so the blood will initially move passively from the atria into the ventricles. When the action potential triggers the muscles in the atria to contract (atrial systole), the pressure within the atria rises further, pumping blood into the ventricles. During ventricular systole, pressure rises in the ventricles, pumping blood into the pulmonary trunk from the right ventricle and into the aorta from the left ventricle. Again, as you consider this flow and relate it to the conduction pathway, the elegance of the system should become apparent.
Phases of the Cardiac Cycle: At the beginning of the cardiac cycle, both the atria and ventricles are relaxed (diastole). Blood is flowing into the right atrium from the superior and inferior venae cavae and the coronary sinus. Blood flows into the left atrium from the four pulmonary veins. The two atrioventricular valves, the tricuspid and mitral valves, are both open, so blood flows unimpeded from the atria and into the ventricles. Approximately 70–80% of ventricular filling occurs by this method. The two semilunar valves, the pulmonary and aortic valves, are closed, preventing backflow of blood into the right and left ventricles from the pulmonary trunk on the right and the aorta on the left.
Atrial Systole and Diastole: Contraction of the atria follows depolarization, represented by the P wave of the ECG (Figure 25). As the atrial muscles contract from the superior portion of the atria toward the atrioventricular septum, pressure rises within the atria and blood is pumped into the ventricles through the open atrioventricular (tricuspid, and mitral or bicuspid) valves. At the start of atrial systole, the ventricles are normally filled with approximately 70–80% of their capacity due to inflow during diastole. Atrial contraction, also referred to as the “atrial kick,” contributes the remaining 20–30% of filling (Figure 24). Atrial systole lasts approximately 100 ms and ends prior to ventricular systole, as the atrial muscle returns to diastole.
Ventricular Systole: Ventricular systole (Figure 24) follows the depolarization of the ventricles and is represented by the QRS complex in the ECG (Figure 25).
Initially, as the muscles in the ventricle contract, the pressure of the blood within the chamber rises, but it is not yet high enough to open the semilunar (pulmonary and aortic) valves and be ejected from the heart. However, blood pressure quickly rises above that of the atria that are now relaxed and in diastole. This increase in pressure causes blood to flow back toward the atria, closing the tricuspid and mitral valves.
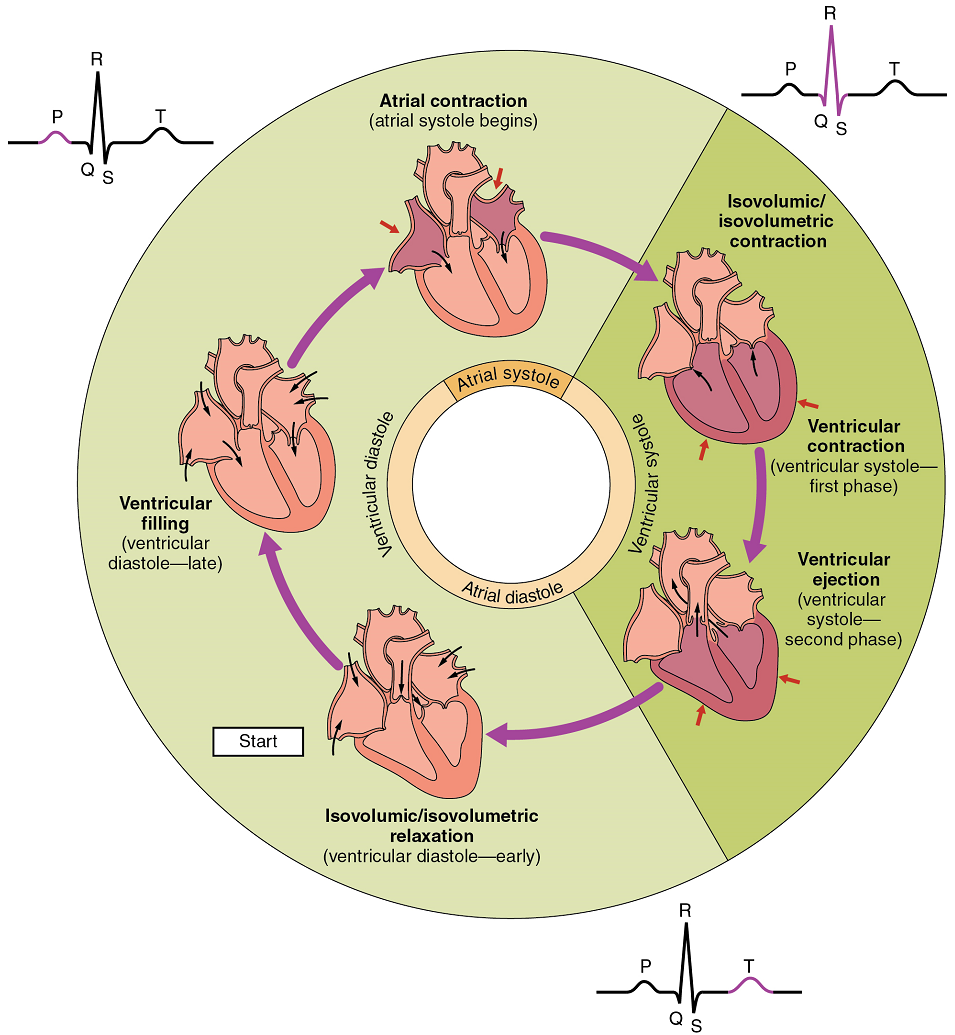
Eventually, the contraction of the ventricular muscle has raised the pressure within the ventricle to the point that it is greater than the pressures in the pulmonary trunk and the aorta. Blood is pumped from the heart, pushing open the pulmonary and aortic semilunar valves. Pressure generated by the left ventricle will be appreciably greater than the pressure generated by the right ventricle, since the existing pressure in the aorta will be so much higher. Nevertheless, both ventricles pump the same amount of blood. This quantity is referred to as stroke volume.
Ventricular Diastole: Ventricular relaxation, or diastole, follows repolarization of the ventricles and is represented by the T wave of the ECG (Figure 25). As the ventricular muscle relaxes, pressure on the remaining blood within the ventricle begins to fall. When pressure within the ventricles drops below pressure in both the pulmonary trunk and aorta, the semilunar valves close to prevent backflow into the heart (Figure 24).
As the ventricular muscle relaxes further, pressure on the blood within the ventricles drops even further. Eventually, it drops below the pressure in the atria. When this occurs, blood flows from the atria into the ventricles, pushing open the tricuspid and mitral valves. As pressure drops within the ventricles, blood flows from the major veins into the relaxed atria and from there into the ventricles. Both chambers are in diastole, the atrioventricular valves are open, and the semilunar valves remain closed (Figure 24). The cardiac cycle is complete.
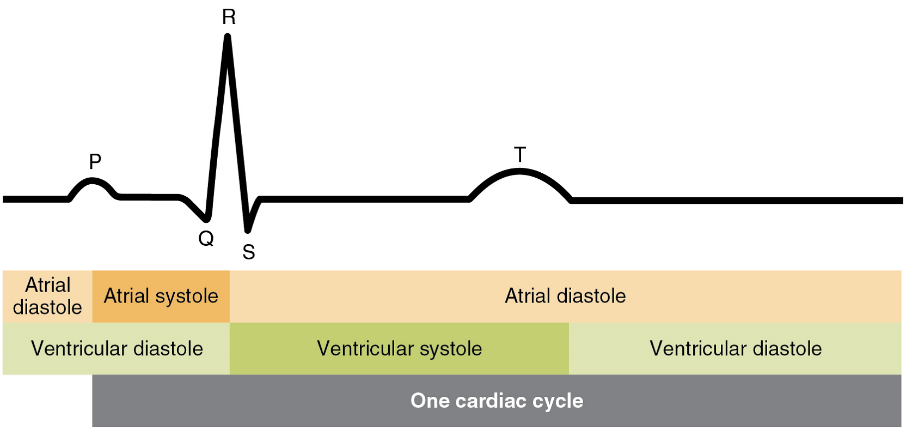
Heart Sounds: One of the simplest, yet effective, diagnostic techniques applied to assess the state of a patient’s heart is auscultation using a stethoscope.
In a normal, healthy heart, there are only two audible heart sounds: S1 and S2. S1 is the sound created by the closing of the atrioventricular valves during ventricular contraction and is normally described as a “lub,” or first heart sound. The second heart sound, S2, is the sound of the closing of the semilunar valves during ventricular diastole and is described as a “dub” (Figure 26). In both cases, as the valves close, the openings within the atrioventricular septum guarded by the valves will become reduced, and blood flow through the opening will become more turbulent until the valves are fully closed.
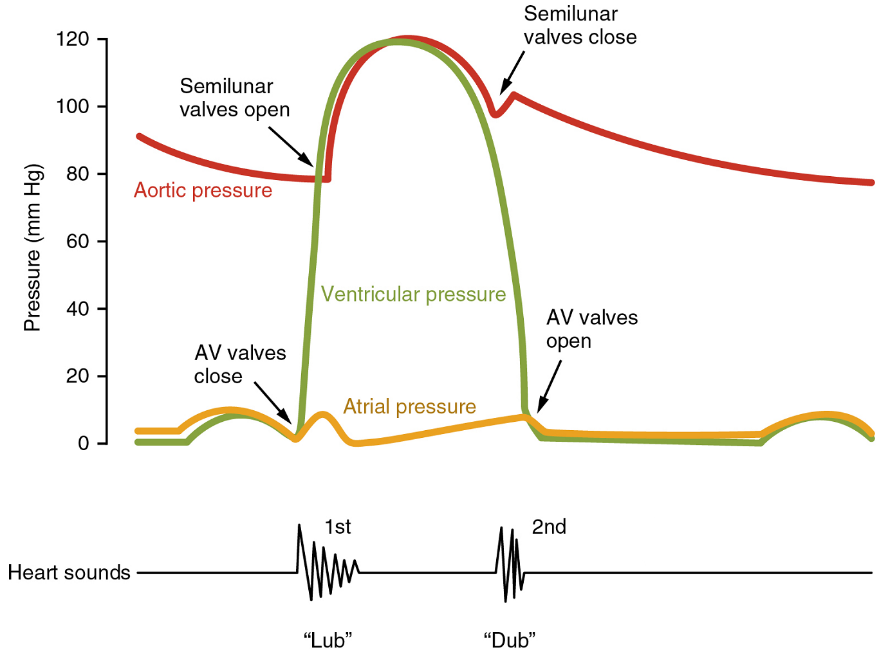
Cardiac output (CO) is a measurement of the amount of blood pumped by each ventricle in one minute. To calculate this value, multiply stroke volume (SV), the amount of blood pumped by each ventricle, by heart rate (HR), in contractions per minute (or beats per minute, bpm). It can be represented mathematically by the following equation:
CO = HR × SV
Stroke volume (SV) is normally measured using an echocardiogram to record end diastolic volume (EDV) and end systolic volume (ESV), and calculating the difference: SV = EDV – ESV. Stroke volume can also be measured using a specialized catheter, but this is an invasive procedure and far more dangerous to the patient. A mean stroke volume for a resting 70 kg (150 lb) individual would be approximately 70 mL. There are several important variables, including size of the heart, physical and mental condition of the individual, sex, contractility, duration of contraction, preload or EDV, and afterload or resistance. Normal range for stroke volume would be 55–100 mL. An average resting heart rate is approximately 75 bpm but can range from 60–100 in some individuals.
Part 4: Cardiac Physiology
The autorhythmicity inherent in cardiac cells keeps the heart beating at a regular pace; however, the heart is regulated by and responds to outside influences as well (Figure 27). Neural and endocrine controls are vital to the regulation of cardiac function. In addition, the heart is sensitive to several environmental factors, including electrolytes.
Heart Rates: Heart rates vary considerably, not only with exercise and fitness levels, but also with age. Newborn resting heart rates may be 120 bpm. Heart rate gradually decreases until young adulthood and then gradually increases again with age.
Maximum heart rates are normally in the range of 200–220 bpm, although there are some extreme cases in which they may reach higher levels. As one ages, the ability to generate maximum rates decreases. This may be estimated by taking the maximal value of 220 bpm and subtracting the individual’s age. So a 40-year-old individual would be expected to hit a maximum rate of approximately 180, and a 60-year-old person would achieve a heart rate of 160.
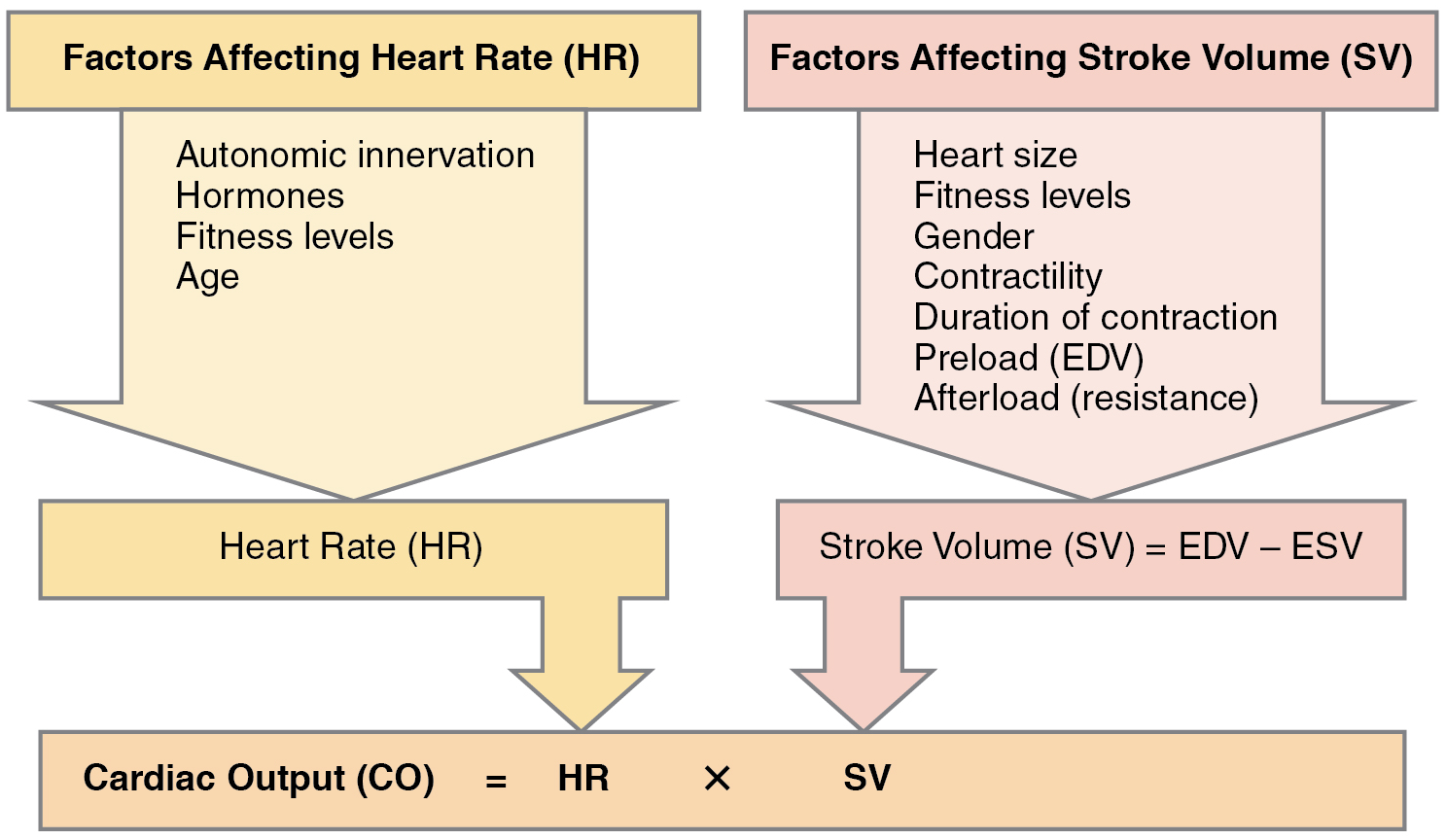
Bradycardia (resting heart rate below 60 bpm) may be caused by either inherent factors or causes external to the heart. While the condition may be inherited, typically it is acquired in older individuals. Inherent causes include abnormalities in either the sinoatrial or atrioventricular node. If the condition is serious, a pacemaker may be required. Other causes include ischemia to the heart muscle or diseases of the heart vessels or valves. External causes include metabolic disorders, pathologies of the endocrine system often involving the thyroid, electrolyte imbalances, neurological disorders including inappropriate autonomic responses, autoimmune pathologies, over-prescription of beta blocker drugs that reduce heart rate, recreational drug use, or even prolonged bed rest. Treatment relies upon establishing the underlying cause of the disorder and may necessitate supplemental oxygen.
Tachycardia (resting heart rate above 100 bpm) is not normal in a resting patient but may be detected in pregnant women or individuals experiencing extreme stress. In the latter case, it would likely be triggered by stimulation from the limbic system or disorders of the autonomic nervous system. In some cases, tachycardia may involve only the atria. Some individuals may remain asymptomatic, but when present, symptoms may include dizziness, shortness of breath, lightheadedness, rapid pulse, heart palpations, chest pain, or fainting (syncope). While tachycardia is defined as a heart rate above 100 bpm, there is considerable variation among people. Further, the normal resting heart rates of children are often above 100 bpm, but this is not considered to be tachycardia Many causes of tachycardia may be benign, but the condition may also be correlated with fever, anaemia, hypoxia, hyperthyroidism, hypersecretion of catecholamines, some cardiomyopathies, some disorders of the valves, and acute exposure to radiation. Elevated rates in an exercising or resting patient are normal and expected. Resting rate should always be taken after recovery from exercise. Treatment depends upon the underlying cause but may include medications, implantable cardioverter defibrillators, ablation, or surgery.
Correlation Between Heart Rates and Cardiac Output: Initially, physiological conditions that cause heart rate to increase also trigger an increase in stroke volume. During exercise, the rate of blood returning to the heart increases. However as the heart rate rises, there is less time spent in diastole and consequently less time for the ventricles to fill with blood. Even though there is less filling time, stroke volume will initially remain high. However, as heart rate continues to increase, stroke volume gradually decreases due to decreased filling time. Cardiac output will initially stabilize as the increasing heart rate compensates for the decreasing stroke volume , but at very high rates, cardiac output will eventually decrease as increasing rates are no longer able to compensate for the decreasing stroke volume. Consider this phenomenon in a healthy young individual. Initially, as heart rate increases from resting to approximately 120 bpm, cardiac output will rise. As heart rate increases from 120 to 160 bpm, cardiac output remains stable, since the increase in rate is offset by decreasing ventricular filling time and, consequently, stroke volume. As heart rate continues to rise above 160 bpm, cardiac output actually decreases as stroke volume falls faster than heart rate increases. So although aerobic exercises are critical to maintain the health of the heart, individuals are cautioned to monitor their heart rate to ensure they stay within the target heart rate range of between 120 and 160 bpm, so cardiac output is maintained. The target heart rate is loosely defined as the range in which both the heart and lungs receive the maximum benefit from the aerobic workout and is dependent upon age.
Cardiovascular Centres: Nervous control over heart rate is centralized within the two paired cardiovascular centres of the medulla oblongata (Figure 28). The cardioacceleratory centre stimulates activity via sympathetic stimulation of the cardioacceleratory nerves, and the cardioinhibitory centre inhibits heart rate via parasympathetic stimulation as one component of the vagus nerve, cranial nerve X.
During rest, both centres provide slight stimulation to the heart, contributing to autonomic tone. This is a similar concept to tone in skeletal muscles. Normally, vagal stimulation predominates; left unregulated, the sinoatrial node would initiate a sinus rhythm of approximately 100 bpm.
The ventricles are more richly innervated by sympathetic fibers than parasympathetic fibers. Sympathetic stimulation causes the release of the neurotransmitter norepinephrine (NE) at the neuromuscular junction of the cardiac nerves. Norepinephrine shortens the repolarization period, thus speeding the rate of depolarization and force of contraction, which results in an increase in heart rate.
Norepinephrine binds mainly to the beta-1 receptors in the heart but there are also beta-2 receptors and norepinephrine’s effect is similar on these. Some cardiac medications (for example, beta blockers) work by blocking these receptors, thereby slowing heart rate and are one possible treatment for hypertension. Overuse of these drugs may lead to bradycardia and even stoppage of the heart with chronic use.
Parasympathetic stimulation originates from the cardioinhibitory region with impulses traveling via the vagus nerve (cranial nerve X). The vagus nerve sends branches to both the sinoatrial and atrioventricular nodes, and to portions of both the atria and ventricles. Parasympathetic stimulation releases the neurotransmitter acetylcholine (ACh) at the neuromuscular junction, and ACh slows heart rate. Without any nervous stimulation, the sinoatrial node would establish a sinus rhythm of approximately 100 bpm. Since resting rates are considerably less than this, it becomes evident that parasympathetic stimulation normally slows heart rate.
Input to the Cardiovascular Centre: The cardiovascular centre receives input from a series of visceral receptors with impulses traveling through visceral sensory fibers within the vagus and sympathetic nerves via the cardiac plexus. Among these receptors are various proprioreceptors, baroreceptors, and chemoreceptors, plus stimuli from the limbic system. Collectively, these inputs normally enable the cardiovascular centres to regulate heart function precisely, a process known as cardiac reflexes. Increased physical activity results in increased rates of firing by various proprioreceptors located in muscles, joint capsules, and tendons. Any such increase in physical activity would logically warrant increased blood flow. The cardiac centres monitor these increased rates of firing, and suppress parasympathetic stimulation and increase sympathetic stimulation as needed in order to increase blood flow.
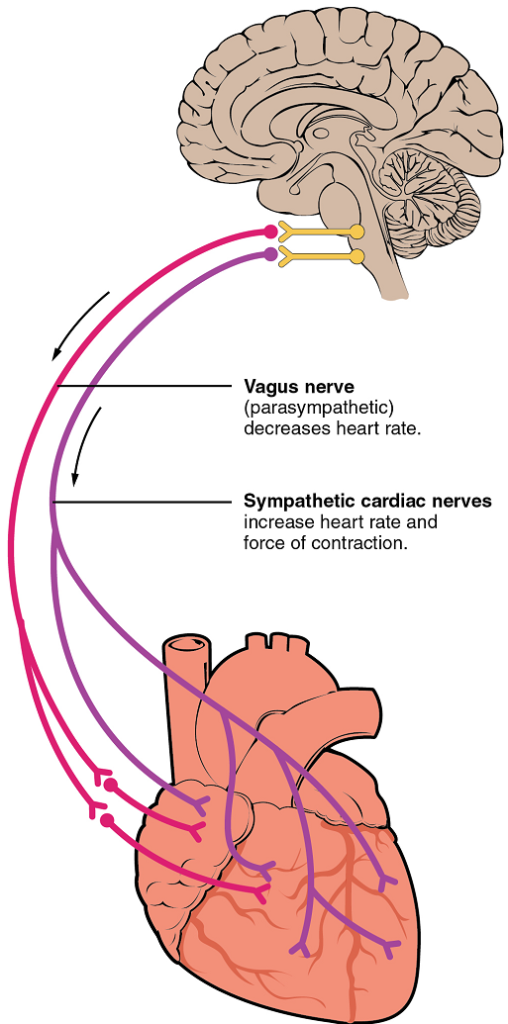
Similarly, baroreceptors are stretch receptors located in the aortic sinus, carotid bodies, the venae cavae, and other locations, including pulmonary vessels and the right side of the heart itself. Rates of firing from the baroreceptors represent blood pressure, level of physical activity, and the relative distribution of blood. The cardiac centres monitor baroreceptor firing to maintain cardiac homeostasis, a mechanism called the baroreceptor reflex. With increased pressure and stretch, the rate of baroreceptor firing increases, and the cardiac centres decrease sympathetic stimulation and increase parasympathetic stimulation. As pressure and stretch decrease, the rate of baroreceptor firing decreases, and the cardiac centres increase sympathetic stimulation and decrease parasympathetic stimulation (Table 4 and Table 5).
| Baroreceptors (aorta, carotid arteries, venae cavae, and atria) | Chemoreceptors (both central nervous system and in proximity to baroreceptors) | |
|---|---|---|
| Sensitive to | Decreasing stretch | Decreasing O2 and increasing CO2, H+, and lactic acid |
| Target | Parasympathetic stimulation suppressed | Sympathetic stimulation increased |
| Response of heart | Increasing heart rate and increasing stroke volume | Increasing heart rate and increasing stroke volume |
| Overall effect | Increasing blood flow and pressure due to increasing cardiac output | Increasing blood flow and pressure due to increasing cardiac output |
| Baroreceptors (aorta, carotid arteries, venae cavae, and atria) | Chemoreceptors (both central nervous system and in proximity to baroreceptors) | |
|---|---|---|
| Sensitive to | Increasing stretch | Increasing O2 and decreasing CO2, H+, and lactic acid |
| Target | Parasympathetic stimulation increased | Sympathetic stimulation suppressed |
| Response of heart | Decreasing heart rate and decreasing stroke volume | Decreasing heart rate and decreasing stroke volume |
| Overall effect | Decreasing blood flow and pressure due to decreasing cardiac output | Decreasing blood flow and pressure due to decreasing cardiac output |
Increased metabolic byproducts associated with increased activity, such as carbon dioxide, hydrogen ions, and lactic acid, plus falling oxygen levels, are detected by a suite of chemoreceptors innervated by the glossopharyngeal and vagus nerves. These chemoreceptors provide feedback to the cardiac centres about the need for increased or decreased blood flow, based on the relative levels of these substances (Table 4 and Table 5).
Other Factors Influencing Heart Rate
Using a combination of autorhythmicity and innervation, the cardiac centres are able to provide relatively precise control over heart rate. However, there are a number of other factors that have an impact on heart rate as well, including epinephrine, norepinephrine, and thyroid hormones; levels of various ions including calcium, potassium, and sodium; body temperature; hypoxia; and pH (Table 6).
Epinephrine and Norepinephrine: The catecholamines (epinephrine and norepinephrine) secreted by the adrenal medulla form one component of the extended fight-or-flight mechanism. The other component is sympathetic stimulation. Epinephrine and norepinephrine have similar effects. There is no parasympathetic stimulation to the adrenal medulla.
Thyroid Hormones: In general, increased levels of thyroid hormone, or thyroxin, increase both the heart rate and the force of contraction (contractility). The impact of thyroid hormone is typically of a much longer duration than that of the catecholamines.
Calcium: Calcium ion levels have great impacts upon both heart rate and contractility; as the levels of calcium ions increase, so do heart rate and contractility. Extremely high levels of calcium may induce cardiac arrest.
| Factor | Effect |
|---|---|
| Increased sympathetic nervous system activity | Increased heart rate and contractility; norepinephrine release |
| Increased parasympathetic nervous system activity | Decreased heart rate |
| Epinephrine/adrenaline and norepinephrine/noradrenaline | Increased heart rate and contractility |
| Thyroxine | Increased heart rate and contractility |
| Blood Ca2+ concentration | High levels increase heart rate and contractility; low levels decrease heart rate and contractility |
| Blood K+ concentration | Low levels increase heart rate and decrease contractility; high levels decrease heart rate and contractility |
| High body temperature | Increased heart rate |
| Low body temperature | Decreased heart rate and contractility |
Blood Vessels and Circulation
In this section, you will learn about the vascular part of the cardiovascular system, that is, the vessels that transport blood throughout the body and provide the physical site where gases, nutrients, and other substances are exchanged with body cells. When vessel functioning is reduced, blood-borne substances do not circulate effectively throughout the body. As a result, tissue injury occurs, metabolism is impaired, and the functions of every bodily system are threatened.
Part 1: Structure and Function of Blood Vessels
Blood is carried through the body via blood vessels. An artery is a blood vessel that carries blood away from the heart, where it branches into ever-smaller vessels. Eventually, the smallest arteries, vessels called arterioles, further branch into tiny capillaries, where nutrients and wastes are exchanged, and then combine with other vessels that exit capillaries to form venules, small blood vessels that carry blood to a vein, a larger blood vessel that returns blood to the heart.
Arteries and veins transport blood in two distinct circuits: the systemic circuit and the pulmonary circuit (Figure 29). Systemic arteries provide blood rich in oxygen to the body’s tissues. The blood returned to the heart through systemic veins has less oxygen, since much of the oxygen carried by the arteries has been delivered to the cells. In contrast, in the pulmonary circuit, arteries carry blood low in oxygen exclusively to the lungs for gas exchange. Pulmonary veins then return freshly oxygenated blood from the lungs to the heart to be pumped back out into systemic circulation. Although arteries and veins differ structurally and functionally, they share certain features.
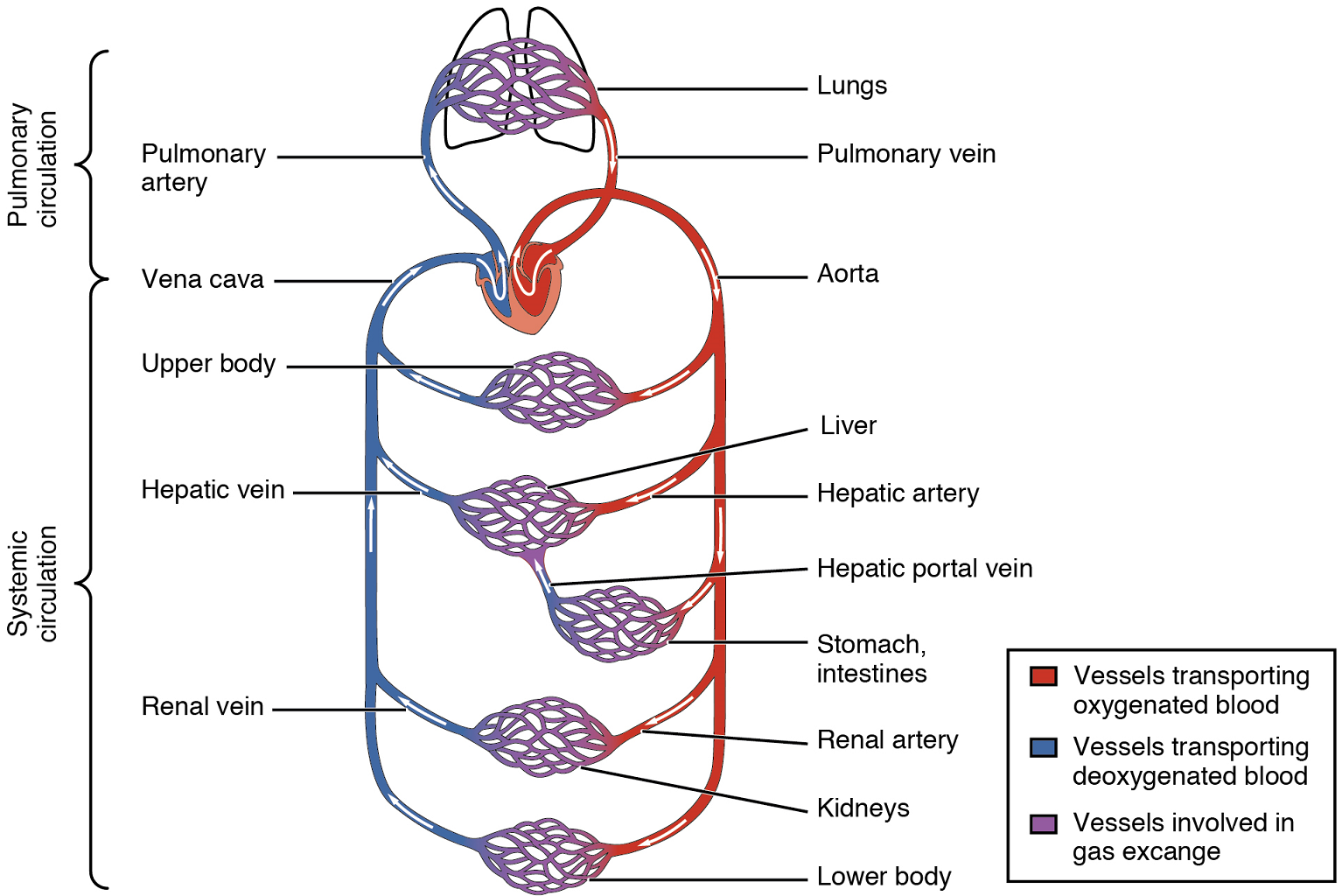
Shared Structures: Different types of blood vessels vary slightly in their structures, but they share the same general features. Arteries and arterioles have thicker walls than veins and venules because they are closer to the heart and receive blood that is surging at a far greater pressure (Figure 30). Each type of vessel has a lumen—a hollow passageway through which blood flows. Arteries have smaller lumens than veins, a characteristic that helps to maintain the pressure of blood moving through the system. Together, their thicker walls and smaller diameters give arterial lumens a more rounded appearance in cross section than the lumens of veins.
By the time blood has passed through capillaries and entered venules, the pressure initially exerted upon it by heart contractions has diminished. In other words, in comparison to arteries, venules and veins withstand a much lower pressure from the blood that flows through them. Their walls are considerably thinner and their lumens are correspondingly larger in diameter, allowing more blood to flow with less vessel resistance. In addition, many veins of the body, particularly those of the limbs, contain valves that assist the unidirectional flow of blood toward the heart. This is critical because blood flow becomes sluggish in the extremities, as a result of the lower pressure and the effects of gravity.
Both arteries and veins have the same three distinct tissue layers, called tunics (from the Latin term tunica), for the garments first worn by ancient Romans; the term tunic is also used for some modern garments. From the most interior layer to the outer, these tunics are the tunica intima, the tunica media, and the tunica externa (Figure 30 and Table 7).
Tunica Intima: The tunica intima (also called the tunica interna) is composed of epithelial and connective tissue layers. Lining the tunica intima is the specialized simple squamous epithelium called the endothelium, which is continuous throughout the entire vascular system, including the lining of the chambers of the heart. Damage to this endothelial lining and exposure of blood to the collagenous fibers beneath is one of the primary causes of clot formation. Until recently, the endothelium was viewed simply as the boundary between the blood in the lumen and the walls of the vessels. Recent studies, however, have shown that it is physiologically critical to such activities as helping to regulate capillary exchange and altering blood flow. The endothelium releases local chemicals called endothelins that can constrict the smooth muscle within the walls of the vessel to increase blood pressure. Uncompensated overproduction of endothelins may contribute to hypertension (high blood pressure) and cardiovascular disease.
Next to the endothelium is the basement membrane, or basal lamina, that effectively binds the endothelium to the connective tissue. The basement membrane provides strength while maintaining flexibility, and it is permeable, allowing materials to pass through it. The thin outer layer of the tunica intima contains a small amount of areolar connective tissue that consists primarily of elastic fibers to provide the vessel with additional flexibility; it also contains some collagenous fibers to provide additional strength.
In larger arteries, there is also a thick, distinct layer of elastic fibers known as the internal elastic membrane (also called the internal elastic lamina) at the boundary with the tunica media. Like the other components of the tunica intima, the internal elastic membrane provides structure while allowing the vessel to stretch. It is permeated with small openings that allow exchange of materials between the tunics. The internal elastic membrane is not apparent in veins. In addition, many veins, particularly in the lower limbs, contain valves formed by sections of thickened endothelium that are reinforced with connective tissue, extending into the lumen.
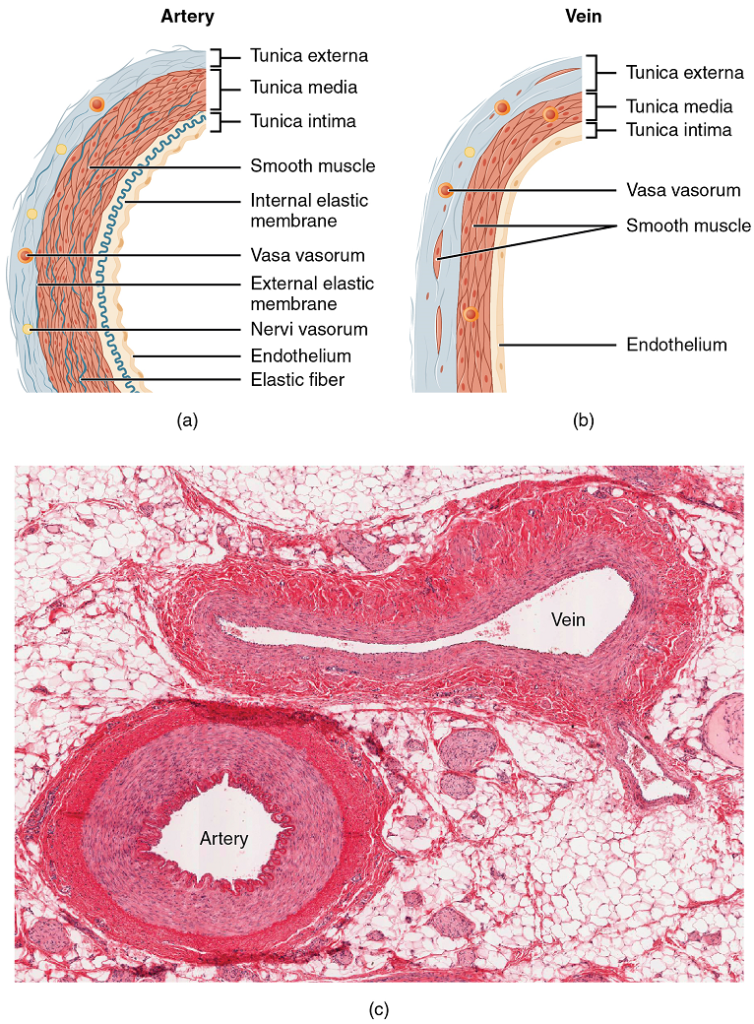
Tunica Media: The tunica media is the substantial middle layer of the vessel wall (Figure 30). It is generally the thickest layer in arteries, and it is much thicker in arteries than it is in veins. The tunica media consists of layers of smooth muscle supported by connective tissue that is primarily made up of elastic fibers, most of which are arranged in circular sheets. Toward the outer portion of the tunic, there are also layers of longitudinal muscle. Contraction and relaxation of the circular muscles decrease and increase the diameter of the vessel lumen, respectively. Specifically, in arteries, vasoconstriction decreases blood flow as the smooth muscle in the walls of the tunica media contracts, making the lumen narrower and increasing blood pressure. Similarly, vasodilation increases blood flow as the smooth muscle relaxes, allowing the lumen to widen and blood pressure to drop.
The smooth muscle layers of the tunica media are supported by a framework of collagenous fibers that also binds the tunica media to the inner and outer tunics. Along with the collagenous fibers are large numbers of elastic fibers that appear as wavy lines in prepared slides.
| Arteries | Veins | Capillaries | |
|---|---|---|---|
| General appearance | Thick walls with small lumens | Thin walls with large lumens | Very (microscopically) thin walls and very small lumens |
| Generally appear rounded | Generally appear flattened | Generally round | |
| Tunica intima | Endothelium usually appears wavy due to constriction of smooth muscle | Endothelium appears smooth | Endothelium appears smooth |
| Internal elastic membrane present in larger vessels | Internal elastic membrane absent | Internal elastic membrane absent | |
| Tunica media | Normally the thickest layer in arteries | Normally thinner than the tunica externa | Tunica media absent |
| Smooth muscle cells and elastic fibers predominate (exact proportions vary with distance from the heart) | Smooth muscle cells and collagenous fibers predominate | ||
| External elastic membrane present in larger vessels | External elastic membrane absent | ||
| Nervi vasorum and vasa vasorum present | |||
| Tunica externa | Normally thinner than tunica media in all but the largest arteries | Normally the thickest layer in veins | Tunica externa absent |
| Collagenous and elastic fibers | Collagenous and smooth fibers predominate | ||
| Nervi vasorum and vasa vasorum present | Nervi vasorum and vasa vasorum present |
Tunica Externa: The outer tunic, the tunica externa (also called the tunica adventitia), is a substantial sheath of connective tissue composed primarily of collagenous fibers. Some bands of elastic fibers are found here as well. The tunica externa in veins also contains groups of smooth muscle fibers. This is normally the thickest tunic in veins and may be thicker than the tunica media in some larger arteries.
Arteries: An artery is a blood vessel that conducts blood away from the heart. All arteries have relatively thick walls that can withstand the high pressure of blood ejected from the heart.
Arterioles: An arteriole is a very small artery that leads to a capillary. Arterioles have the same three tunics as the larger vessels, but the thickness of each is greatly diminished. The critical endothelial lining of the tunica intima is intact. The tunica media is restricted to one or two smooth muscle cell layers in thickness. The tunica externa remains but is very thin (Figure 39).
The importance of the arterioles is that they will be the primary site of both resistance and regulation of blood pressure. The precise diameter of the lumen of an arteriole at any given moment is determined by neural and chemical controls, and vasoconstriction and vasodilation in the arterioles are the primary mechanisms for distribution of blood flow.
Capillaries: A capillary is a microscopic channel that supplies blood to the tissues themselves, a process called perfusion. Exchange of gases and other substances occurs in the capillaries between the blood and the surrounding cells and their tissue fluid (interstitial fluid). The diameter of a capillary lumen is from 5-10 μm; the smallest are just barely wide enough for an erythrocyte to squeeze through. Flow through capillaries is often described as microcirculation.
Unlike the walls of veins and arteries, the wall of a capillary consists of an endothelial layer surrounded by a basement membrane with occasional smooth muscle fibers. There is some variation in wall structure: in a large capillary, several endothelial cells bordering each other may line the lumen; in a small capillary, there may be only a single cell layer that wraps around to contact itself.
Venules: A venule is an extremely small vein, generally 8–100 μm in diameter. Postcapillary venules join multiple capillaries exiting from a capillary bed. Multiple venules join to form veins. The walls of venules consist of endothelium, a thin middle layer with a few muscle cells and elastic fibers, plus an outer layer of connective tissue fibers that constitute a very thin tunica externa. Venules as well as capillaries are the primary sites of emigration or diapedesis, in which the leukocytes adhere to the endothelial lining of the vessels and then squeeze through adjacent cells to enter the tissue fluid.
Veins: A vein is a blood vessel that conducts blood toward the heart. Compared to arteries, veins are thin-walled vessels with large and irregular lumens (Figure 42). Because they are low-pressure vessels, larger veins are commonly equipped with valves that promote the unidirectional flow of blood toward the heart and prevent backflow toward the capillaries caused by the inherent low blood pressure in veins as well as the pull of gravity. Table 8 compares the features of arteries and veins.
| Arteries | Veins | |
|---|---|---|
| Direction of blood flow | Conducts blood away from the heart | Conducts blood toward the heart |
| General appearance | Rounded | Irregular, often collapsed |
| Pressure | High | Low |
| Wall thickness | Thick | Thin |
| Relative oxygen concentration | Higher in systemic arteries; lower in pulmonary arteries | Lower in systemic veins; higher in pulmonary venis |
| Valves | Not present | Present most commonly in limbs and in veins inferior to the heart |
Part 2: Blood Flow, Blood Pressure, and Resistance
Blood flow refers to the movement of blood through a vessel, tissue, or organ, and is usually expressed in terms of volume of blood per unit of time. It is initiated by the contraction of the ventricles of the heart. Ventricular contraction ejects blood into the major arteries, resulting in flow from regions of higher pressure to regions of lower pressure, as blood encounters smaller arteries and arterioles, then capillaries, then the venules and veins of the venous system. This section discusses a number of critical variables that contribute to blood flow throughout the body. It also discusses the factors that impede or slow blood flow, a phenomenon known as resistance.
As noted earlier, hydrostatic pressure is the force exerted by a fluid due to gravitational pull, usually against the wall of the container in which it is located. One form of hydrostatic pressure is blood pressure, the force exerted by blood upon the walls of the blood vessels or the chambers of the heart. Blood pressure may be measured in capillaries and veins, as well as the vessels of the pulmonary circulation; however, the term blood pressure without any specific descriptors typically refers to systemic arterial blood pressure—that is, the pressure of blood flowing in the arteries of the systemic circulation. In clinical practice, this pressure is measured in mm Hg and is usually obtained using the brachial artery of the arm.
Components of Arterial Blood Pressure: Arterial blood pressure in the larger vessels consists of several distinct components (Figure 43): systolic and diastolic pressures, pulse pressure, and mean arterial pressure.
Systolic and Diastolic Pressures: When systemic arterial blood pressure is measured, it is recorded as a ratio of two numbers (e.g., 120/80 is a normal adult blood pressure), expressed as systolic pressure over diastolic pressure. The systolic pressure is the higher value (typically around 120 mm Hg) and reflects the arterial pressure resulting from the ejection of blood during ventricular contraction, or systole. The diastolic pressure is the lower value (usually about 80 mm Hg) and represents the arterial pressure of blood during ventricular relaxation, or diastole.
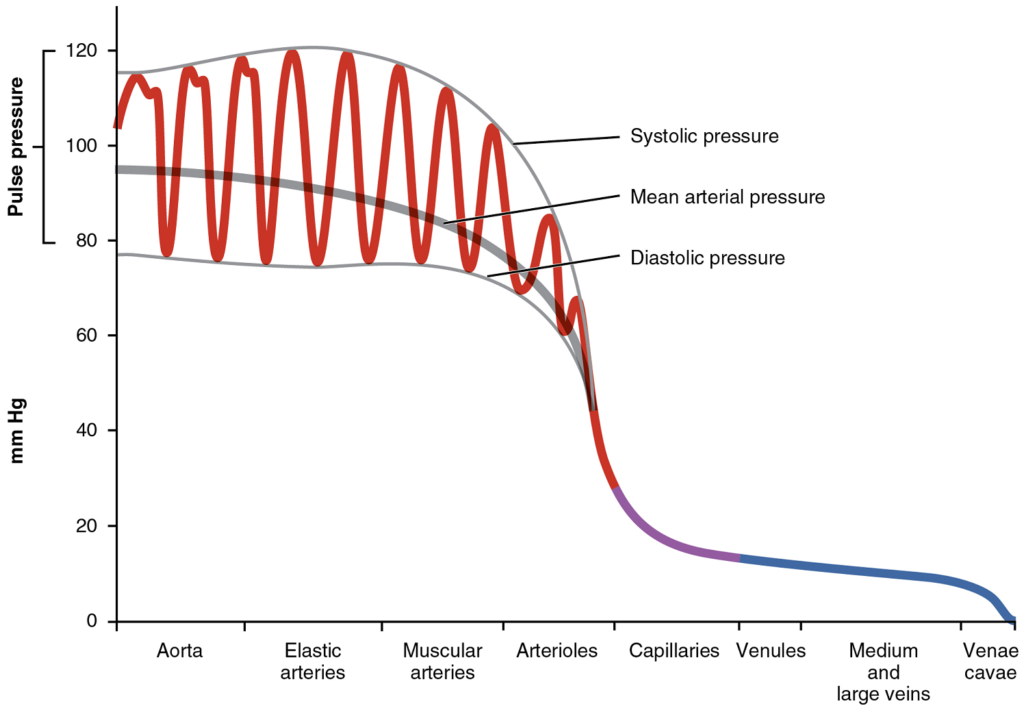
Mean Arterial Pressure: Mean arterial pressure (MAP) represents the “average” pressure of blood in the arteries, that is, the average force driving blood into vessels that serve the tissues. Mean is a statistical concept and is calculated by taking the sum of the values divided by the number of values. Although complicated to measure directly and complicated to calculate, MAP can be approximated by adding the diastolic pressure to one-third of the pulse pressure or systolic pressure minus the diastolic pressure:
MAP = diastolic BP + ((systolic-diastolic BP) / 3)
Normally, the MAP falls within the range of 70–110 mm Hg. If the value falls below 60 mm Hg for an extended time, blood pressure will not be high enough to ensure circulation to and through the tissues, which results in ischemia, or insufficient blood flow. A condition called hypoxia, inadequate oxygenation of tissues, commonly accompanies ischemia. The term hypoxemia refers to low levels of oxygen in systemic arterial blood.
Measurement of Blood Pressure: Blood pressure is one of the critical parameters measured on virtually every patient in every healthcare setting. The technique used today was developed more than 100 years ago by a pioneering Russian physician, Dr. Nikolai Korotkoff. Turbulent blood flow through the vessels can be heard as a soft ticking while measuring blood pressure; these sounds are known as Korotkoff sounds. The technique of measuring blood pressure requires the use of a sphygmomanometer (a blood pressure cuff attached to a measuring device) and a stethoscope. The technique is as follows:
- The clinician wraps an inflatable cuff tightly around the patient’s arm at about the level of the heart.
- The clinician squeezes a rubber pump to inject air into the cuff, raising pressure around the artery and temporarily cutting off blood flow into the patient’s arm.
- The clinician places the stethoscope on the patient’s antecubital region and, while gradually allowing air within the cuff to escape, listens for the Korotkoff sounds.
The first sound heard through the stethoscope—the first Korotkoff sound—indicates systolic pressure. As more air is released from the cuff, blood is able to flow freely through the brachial artery and all sounds disappear. The point at which the last sound is heard is recorded as the patient’s diastolic pressure.
Pulse: After blood is ejected from the heart, elastic fibers in the arteries help maintain a high-pressure gradient as they expand to accommodate the blood, then recoil. This expansion and recoiling effect, known as the pulse, can be palpated manually or measured electronically. Although the effect diminishes over distance from the heart, elements of the systolic and diastolic components of the pulse are still evident down to the level of the arterioles.
Because pulse indicates heart rate, it is measured clinically to provide clues to a patient’s state of health. It is recorded as beats per minute. Both the rate and the strength of the pulse are important clinically. A high or irregular pulse rate can be caused by physical activity or other temporary factors, but it may also indicate a heart condition. The pulse strength indicates the strength of ventricular contraction and cardiac output. If the pulse is strong, then systolic pressure is high. If it is weak, systolic pressure has fallen, and medical intervention may be warranted.
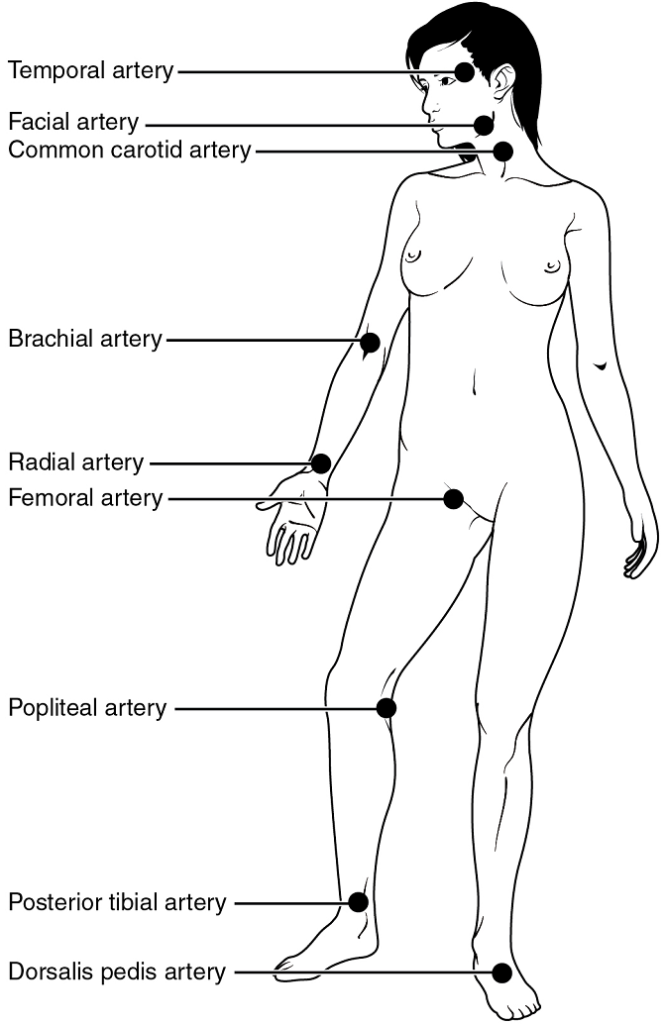
Pulse can be palpated manually by placing the tips of the fingers across an artery that runs close to the body surface and pressing lightly. While this procedure is normally performed using the radial artery in the wrist or the common carotid artery in the neck, any superficial artery that can be palpated may be used (Figure 32). Common sites to find a pulse include temporal and facial arteries in the head, brachial arteries in the upper arm, femoral arteries in the thigh, popliteal arteries behind the knees, posterior tibial arteries near the medial tarsal regions, and dorsalis pedis arteries in the feet. A variety of commercial electronic devices are also available to measure pulse.
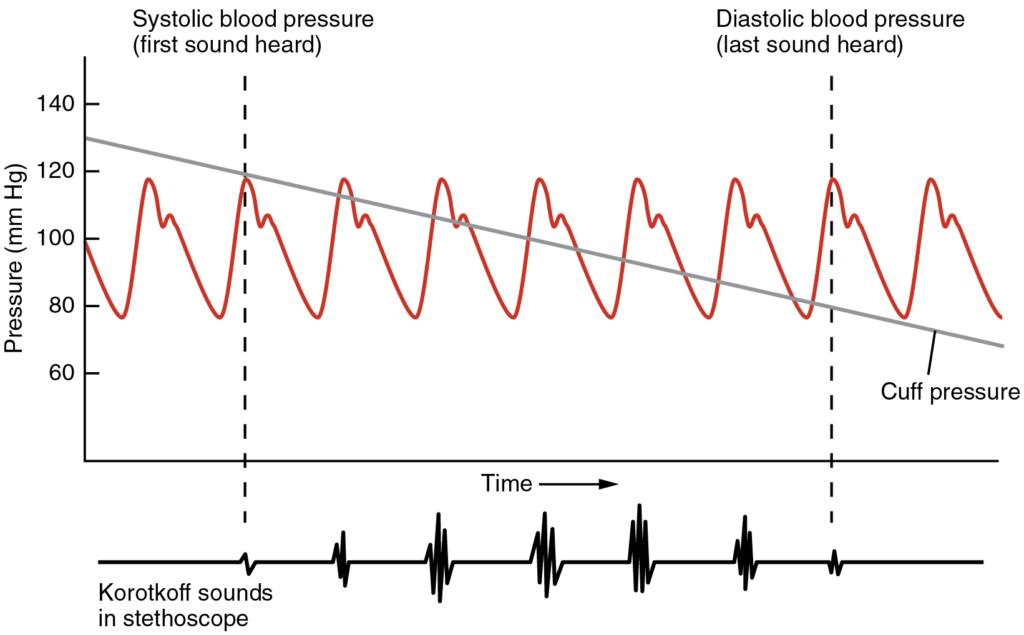
Variables Affecting Blood Flow and Blood Pressure: Five variables influence blood flow and blood pressure:
- Cardiac output
- Compliance
- Volume of the blood
- Viscosity of the blood
- Blood vessel length and diameter
Recall that blood moves from higher pressure to lower pressure. It is pumped from the heart into the arteries at high pressure. If you increase pressure in the arteries (afterload), and cardiac function does not compensate, blood flow will actually decrease. In the venous system, the opposite relationship is true. Increased pressure in the veins does not decrease flow as it does in arteries, but actually increases flow. Since pressure in the veins is normally relatively low, for blood to flow back into the heart, the pressure in the atria during atrial diastole must be even lower. It normally approaches zero, except when the atria contract (Figure 33).
Cardiac Output: Cardiac output is the measurement of blood flow from the heart through the ventricles, and is usually measured in liters per minute. Any factor that causes cardiac output to increase, by elevating heart rate or stroke volume or both, will elevate blood pressure and promote blood flow. These factors include sympathetic stimulation, the catecholamines epinephrine and norepinephrine, thyroid hormones, and increased calcium ion levels. Conversely, any factor that decreases cardiac output, by decreasing heart rate or stroke volume or both, will decrease arterial pressure and blood flow. These factors include parasympathetic stimulation, elevated or decreased potassium ion levels, decreased calcium levels, anoxia, and acidosis.
Compliance: Compliance is the ability of any compartment to expand to accommodate increased content. A metal pipe, for example, is not compliant, whereas a balloon is. The greater the compliance of an artery, the more effectively it is able to expand to accommodate surges in blood flow without increased resistance or blood pressure. Veins are more compliant than arteries and can expand to hold more blood. When vascular disease causes stiffening of arteries, compliance is reduced and resistance to blood flow is increased. The result is more turbulence, higher pressure within the vessel, and reduced blood flow. This increases the work of the heart.
Blood Volume: The relationship between blood volume, blood pressure, and blood flow is intuitively obvious. Water may merely trickle along a creek bed in a dry season, but rush quickly and under great pressure after a heavy rain. Similarly, as blood volume decreases, pressure and flow decrease. As blood volume increases, pressure and flow increase.
Blood Viscosity: Viscosity is the thickness of fluids that affects their ability to flow. Clean water, for example, is less viscous than mud. The viscosity of blood is directly proportional to resistance and inversely proportional to flow; therefore, any condition that causes viscosity to increase will also increase resistance (and therefore blood pressure) and decrease flow. For example, imagine sipping milk, then a milkshake, through the same size straw. You experience more resistance and therefore less flow from the milkshake. Conversely, any condition that causes viscosity to decrease (such as when the milkshake melts) will decrease resistance and increase flow.
Normally the viscosity of blood does not change over short periods of time. The two primary determinants of blood viscosity are the formed elements and plasma proteins. Since the vast majority of formed elements are erythrocytes, any condition affecting erythropoiesis, such as polycythemia or anaemia, can alter viscosity. Viscosity generally increases with increasing numbers of formed elements relative to the amount of plasma. If the concentration of proteins in the plasma is increased, this would also increase viscosity. Since most plasma proteins are produced by the liver, any condition affecting liver function can also change the viscosity and therefore affect blood flow. Liver abnormalities include hepatitis, cirrhosis, alcohol damage, and drug toxicities. While leukocytes and platelets are normally a small component of the formed elements, there are some rare conditions in which there is such a great overproduction of these that viscosity increases.
Vessel Length and Diameter: The length of a vessel is directly proportional to its resistance: the longer the vessel, the greater the resistance and the lower the flow. As with blood volume, this makes intuitive sense, since the increased surface area of the vessel will impede the flow of blood. Likewise, if the vessel is shortened, the resistance will decrease and flow will increase.
In contrast to length, the diameter of blood vessels changes throughout the body, according to the type of vessel, as we discussed earlier. The diameter of any given vessel may also change frequently throughout the day in response to neural and chemical signals that trigger vasodilation and vasoconstriction. The vascular tone of the vessel is the contractile state of the smooth muscle and the primary determinant of diameter, and thus of resistance and flow. The effect of vessel diameter on resistance is inverse: Given the same volume of blood, an increased diameter means there is less blood contacting the vessel wall, thus lower friction and lower resistance, subsequently increasing flow. A decreased diameter means more of the blood contacts the vessel wall, and resistance increases, subsequently decreasing flow.
Vasodilation and vasoconstriction of arterioles play more significant roles in regulating blood pressure than do the vasodilation and vasoconstriction of other vessels.
Venous System: The pumping action of the heart propels the blood into the arteries, from an area of higher pressure toward an area of lower pressure. If blood is to flow from the veins back into the heart, the pressure in the veins must be greater than the pressure in the atria of the heart. Two factors help maintain this pressure gradient between the veins and the heart. First, the pressure in the atria during diastole is very low, often approaching zero when the atria are relaxed (atrial diastole). Second, two physiologic “pumps” increase pressure in the venous system. The use of the term “pump” implies a physical device that speeds flow. These physiological pumps are less obvious.
Skeletal Muscle Pump: In many body regions, the pressure within the veins can be increased by the contraction of the surrounding skeletal muscle. This mechanism, known as the skeletal muscle pump (Figure 34), helps the lower-pressure veins counteract the force of gravity, increasing pressure to move blood back to the heart. As leg muscles contract, for example during walking or running, they exert pressure on nearby veins with their numerous one-way valves. This increased pressure causes blood to flow upward, opening valves superior to the contracting muscles so blood flows through. Simultaneously, valves inferior to the contracting muscles close; thus, blood should not seep back downward toward the feet. Military recruits are trained to flex their legs slightly while standing at attention for prolonged periods. Failure to do so may allow blood to pool in the lower limbs rather than returning to the heart. Consequently, the brain will not receive enough oxygenated blood, and the individual may lose consciousness.
Respiratory Pump: The respiratory pump aids blood flow through the veins of the thorax and abdomen. During inhalation, the volume of the thorax increases, largely through the contraction of the diaphragm, which moves downward and compresses the abdominal cavity. The elevation of the chest caused by the contraction of the external intercostal muscles also contributes to the increased volume of the thorax. The volume increase causes air pressure within the thorax to decrease, allowing us to inhale. Additionally, as air pressure within the thorax drops, blood pressure in the thoracic veins also decreases, falling below the pressure in the abdominal veins. This causes blood to flow along its pressure gradient from veins outside the thorax, where pressure is higher, into the thoracic region, where pressure is now lower. This in turn promotes the return of blood from the thoracic veins to the atria. During exhalation, when air pressure increases within the thoracic cavity, pressure in the thoracic veins increases, speeding blood flow into the heart while valves in the veins prevent blood from flowing backward from the thoracic and abdominal veins. Also notice that, as blood moves from venules to veins, the average blood pressure drops.
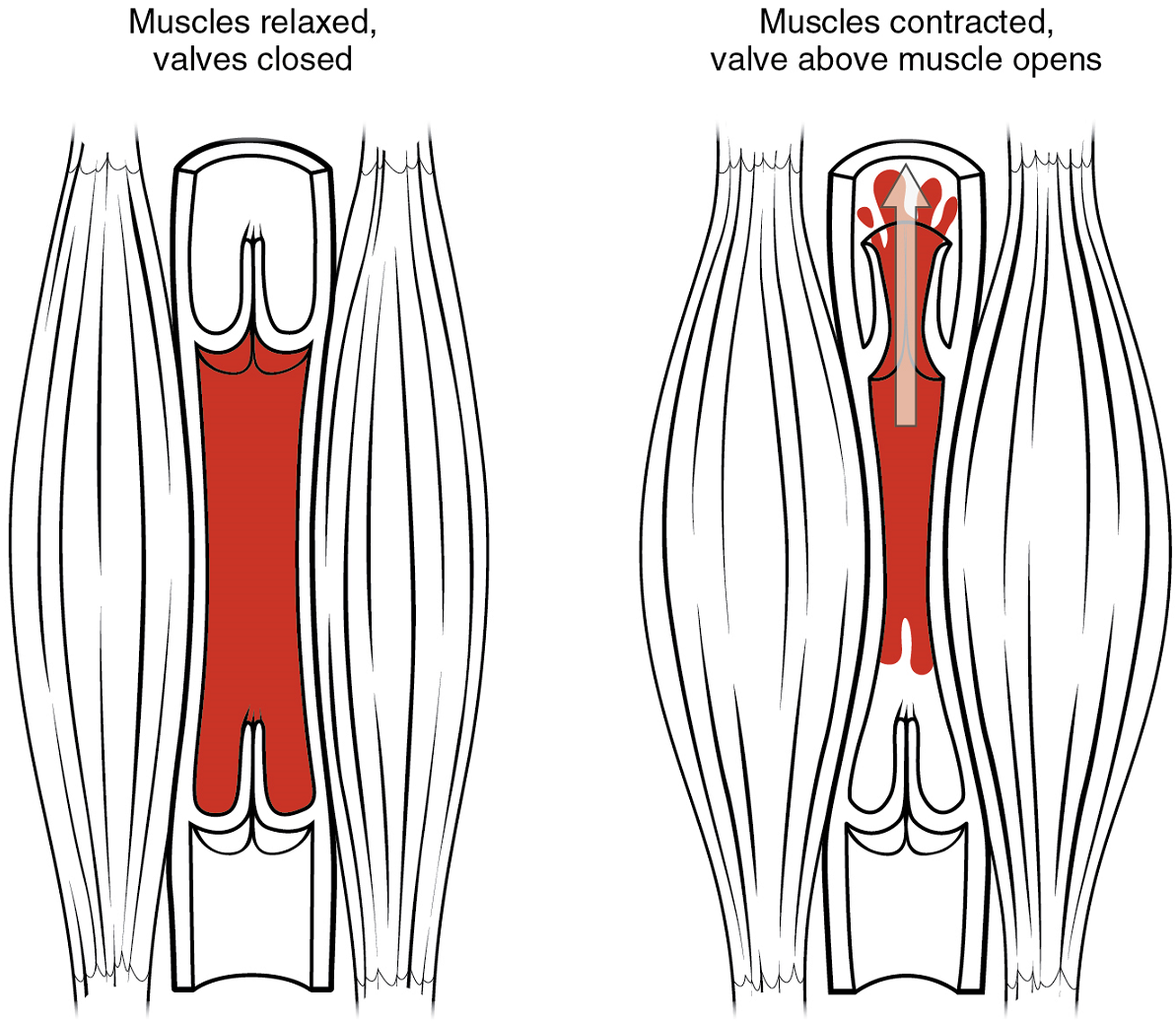
Part 3: Capillary Exchange
The primary purpose of the cardiovascular system is to circulate gases, nutrients, wastes, and other substances to and from the cells of the body. Small molecules, such as gases, lipids, and lipid-soluble molecules, can diffuse directly through the membranes of the endothelial cells of the capillary wall. Glucose, amino acids, and ions—including sodium, potassium, calcium, and chloride—use transporters to move through specific channels in the membrane by facilitated diffusion. Glucose, ions, and larger molecules may also leave the blood through intercellular clefts. Larger molecules can pass through the pores of fenestrated capillaries, and even large plasma proteins can pass through the great gaps in the sinusoids. Some large proteins in blood plasma can move into and out of the endothelial cells packaged within vesicles by endocytosis and exocytosis. Water moves by osmosis.
Part 4: Homeostatic Regulation of the Vascular System
To maintain homeostasis in the cardiovascular system and provide adequate blood to the tissues, blood flow must be redirected continually to the tissues as they become more active. In a very real sense, the cardiovascular system engages in resource allocation, because there is not enough blood flow to distribute blood equally to all tissues simultaneously. For example, when an individual is exercising, more blood will be directed to skeletal muscles, the heart, and the lungs. Following a meal, more blood is directed to the digestive system. Only the brain receives a more or less constant supply of blood whether you are active, resting, thinking, or engaged in any other activity.
Table 9 provides the distribution of systemic blood at rest and during exercise. Although most of the data appears logical, the values for the distribution of blood to the integument may seem surprising. During exercise, the body distributes more blood to the body surface where it can dissipate the excess heat generated by increased activity into the environment. Three homeostatic mechanisms ensure adequate blood flow, blood pressure, distribution, and ultimately perfusion: neural, endocrine, and autoregulatory mechanisms (Figure 35).
| Organ | Resting (mL/min) | Mild exercise (mL/min) | Maximal exercise (mL/min) |
|---|---|---|---|
| Skeletal muscle | 1200 | 4500 | 12,500 |
| Heart | 250 | 350 | 750 |
| Brain | 750 | 750 | 750 |
| Integument | 500 | 1500 | 1900 |
| Kidney | 1100 | 900 | 600 |
| Gastrointestinal | 1400 | 1100 | 600 |
| Others (e.g., liver, spleen) | 600 | 400 | 400 |
| Total | 5800 | 9500 | 17,500 |
Neural Regulation: The nervous system plays a critical role in the regulation of vascular homeostasis. The primary regulatory sites include the cardiovascular centres in the brain that control both cardiac and vascular functions. In addition, more generalized neural responses from the limbic system and the autonomic nervous system are factors.
The Cardiovascular Centres in the Brain: Neurological regulation of blood pressure and flow depends on the cardiovascular centres located in the medulla oblongata. This cluster of neurons responds to changes in blood pressure as well as blood concentrations of oxygen, carbon dioxide, and hydrogen ions. The cardiovascular centre contains three distinct components:
- The cardioacceleratory centre stimulates cardiac function by regulating heart rate and stroke volume via sympathetic stimulation from the cardiac accelerator nerve.
- The cardioinhibitory centre slows cardiac function by decreasing heart rate via parasympathetic stimulation from the vagus nerve.
- The vasomotor centre controls vessel tone or contraction of the smooth muscle in the tunica media. Changes in diameter affect peripheral resistance, pressure, and flow, which affect cardiac output. The majority of these neurons act via the release of the neurotransmitter norepinephrine from sympathetic neurons.
Although each centre functions independently, they are not anatomically distinct.
There is also a small population of neurons that control vasodilation in the vessels of the brain and skeletal muscles by relaxing the smooth muscle fibers in the vessel tunics. Many of these are cholinergic neurons, that is, they release acetylcholine, which in turn stimulates the vessels’ endothelial cells to release nitric oxide (NO), which causes vasodilation. Others release norepinephrine that binds to β2 receptors. A few neurons release NO directly as a neurotransmitter.
Baroreceptor Reflexes: Baroreceptors are specialized stretch receptors located within thin areas of blood vessels and heart chambers that respond to the degree of stretch caused by the presence of blood. They send impulses to the cardiovascular centres to regulate blood pressure. Vascular baroreceptors are found primarily in sinuses (small cavities) within the aorta and carotid arteries: The aortic sinuses are found in the walls of the ascending aorta just superior to the aortic valve, whereas the carotid sinuses are in the base of the internal carotid arteries. There are also low-pressure baroreceptors located in the walls of the venae cavae and right atrium.
When blood pressure increases, the baroreceptors are stretched more tightly and initiate action potentials at a higher rate. At lower blood pressures, the degree of stretch is lower and the rate of firing is slower. When the cardiovascular centres in the medulla oblongata receives this input, they triggers a reflex that maintains homeostasis (Figure 36):
- When blood pressure rises too high, the baroreceptors fire at a higher rate and trigger parasympathetic stimulation of the heart. As a result, cardiac output falls. Sympathetic stimulation of the peripheral arterioles will also decrease, resulting in vasodilation. Combined, these activities cause blood pressure to fall.
- When blood pressure drops too low, the rate of baroreceptor firing decreases. This will trigger an increase in sympathetic stimulation of the heart, causing cardiac output to increase. It will also trigger sympathetic stimulation of the peripheral vessels, resulting in vasoconstriction. Combined, these activities cause blood pressure to rise.
The baroreceptors in the venae cavae and right atrium monitor blood pressure as the blood returns to the heart from the systemic circulation. Normally, blood flow into the aorta is the same as blood flow back into the right atrium. If blood is returning to the right atrium more rapidly than it is being ejected from the left ventricle, the atrial receptors will stimulate the cardiovascular centres to increase sympathetic firing and increase cardiac output until homeostasis is achieved. The opposite is also true. This mechanism is referred to as the atrial reflex.
Chemoreceptor Reflexes: In addition to the baroreceptors are chemoreceptors that monitor levels of oxygen, carbon dioxide, and hydrogen ions (pH), and thereby contribute to vascular homeostasis. Chemoreceptors monitoring the blood are located in close proximity to the baroreceptors in the aortic and carotid sinuses. They signal the cardiovascular centres as well as the respiratory centres in the medulla oblongata.
Since tissues consume oxygen and produce carbon dioxide and acids as waste products, when the body is more active, oxygen levels fall and carbon dioxide levels rise as cells undergo cellular respiration to meet the energy needs of activities. This causes more hydrogen ions to be produced, causing the blood pH to drop. When the body is resting, oxygen levels are higher, carbon dioxide levels are lower, more hydrogen is bound, and pH rises.
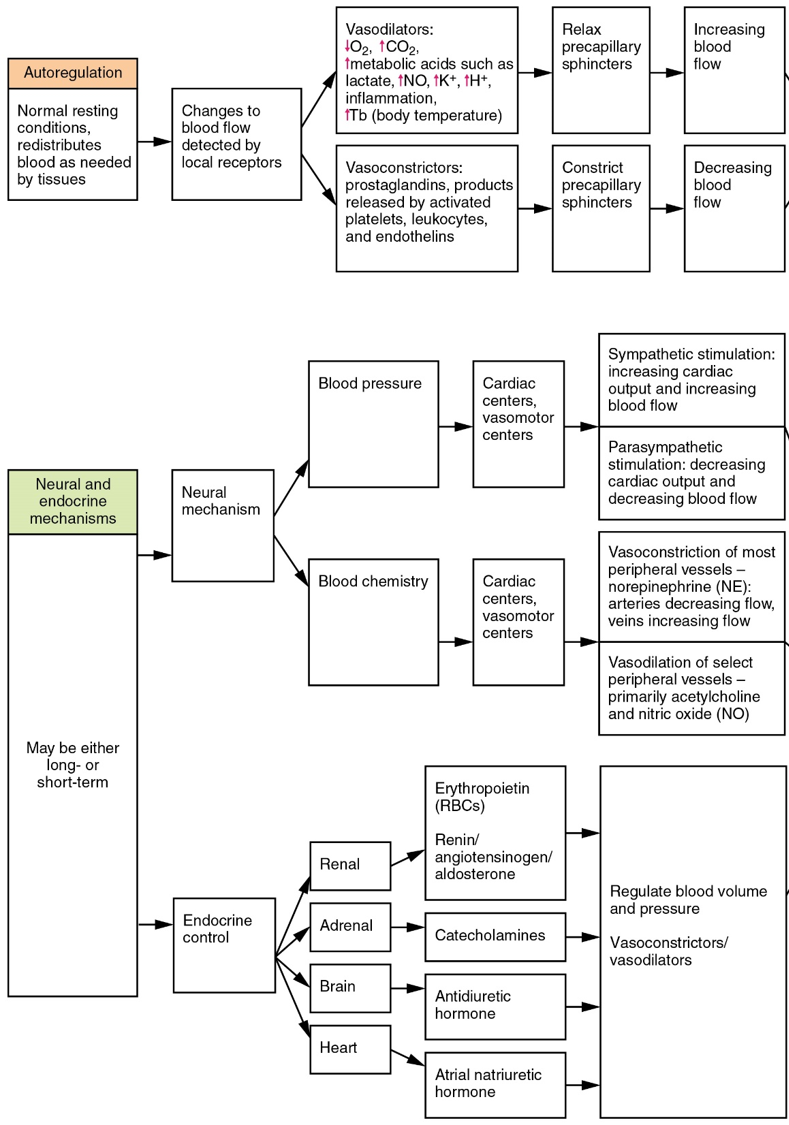
The chemoreceptors respond to increasing carbon dioxide and hydrogen ion levels (falling pH) by stimulating the cardioacceleratory and vasomotor centres, increasing cardiac output and constricting peripheral vessels. The cardioinhibitory centre is suppressed. With falling carbon dioxide and hydrogen ion levels (increasing pH), the cardioinhibitory centre is stimulated, and the cardioacceleratory and vasomotor centres are suppressed, decreasing cardiac output and causing peripheral vasodilation. In order to maintain adequate supplies of oxygen to the cells and remove waste products such as carbon dioxide, it is essential that the respiratory system respond to changing metabolic demands. In turn, the cardiovascular system will transport these gases to the lungs for exchange, again in accordance with metabolic demands. This interrelationship of cardiovascular and respiratory control cannot be overemphasized.
Other neural mechanisms can also have affect cardiovascular function. These include the limbic system that links physiological responses to psychological stimuli, as well as generalized sympathetic and parasympathetic stimulation.
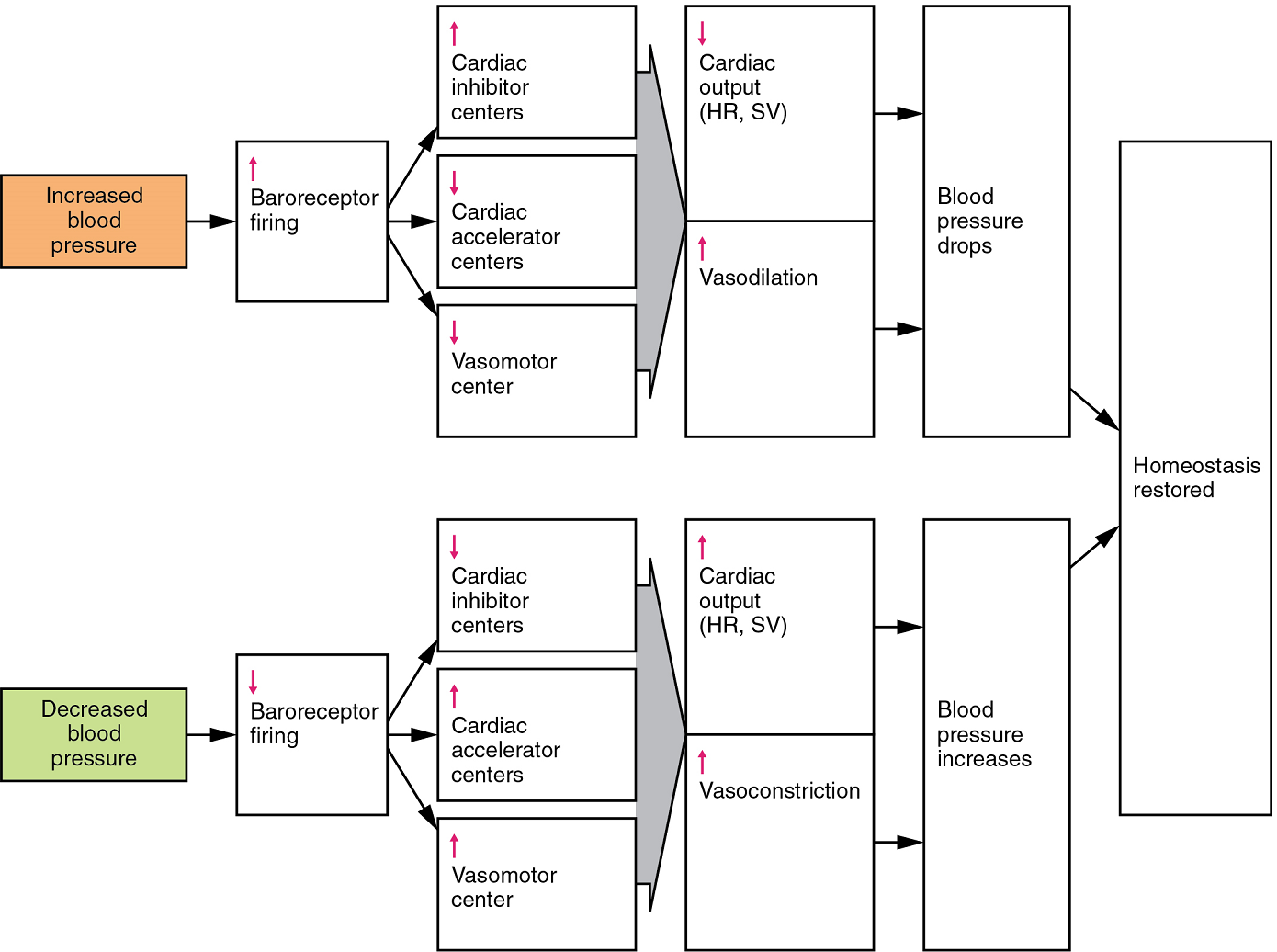
Endocrine Regulation: Endocrine control over the cardiovascular system involves the catecholamines, epinephrine and norepinephrine, as well as several hormones that interact with the kidneys in the regulation of blood volume.
Epinephrine and Norepinephrine: The catecholamines epinephrine and norepinephrine are released by the adrenal medulla, and enhance and extend the body’s sympathetic or “fight-or-flight” response (Figure 37). They increase heart rate and force of contraction, while temporarily constricting blood vessels to organs not essential for flight-or-fight responses and redirecting blood flow to the liver, muscles, and heart.
Antidiuretic Hormone: Antidiuretic hormone (ADH), also known as vasopressin, is secreted by the cells in the hypothalamus and transported via the hypothalamic-hypophyseal tracts to the posterior pituitary where it is stored until released upon nervous stimulation. The primary trigger prompting the hypothalamus to release antiduiretic hormone is increasing osmolarity of tissue fluid, usually in response to significant loss of blood volume (Figure 38). ADH signals its target cells in the kidneys to reabsorb more water, thus preventing the loss of additional fluid in the urine. This will increase overall fluid levels and help restore blood volume and pressure. In addition, antiduiretic hormone constricts peripheral vessels.
Renin-Angiotensin-Aldosterone Mechanism: The renin-angiotensin-aldosterone mechanism has a major effect upon the cardiovascular system (Figure 37). Renin is an enzyme, although because of its importance in the renin-angiotensin-aldosterone pathway, some sources identify it as a hormone. Specialized cells in the kidneys found in the juxtaglomerular apparatus respond to decreased blood flow by secreting renin into the blood. Renin converts the plasma protein angiotensinogen, which is produced by the liver, into its active form—angiotensin I. Angiotensin I circulates in the blood and is then converted into angiotensin II in the lungs. This reaction is catalyzed by the enzyme angiotensin-converting enzyme (ACE).
Angiotensin II is a powerful vasoconstrictor, greatly increasing blood pressure. It also stimulates the release of antiduiretic hormone and aldosterone, a hormone produced by the adrenal cortex. Aldosterone increases the reabsorption of sodium into the blood by the kidneys. Since water follows sodium, this increases the reabsorption of water. This in turn increases blood volume, raising blood pressure. Angiotensin II also stimulates the thirst centre in the hypothalamus, so an individual will likely consume more fluids, again increasing blood volume and pressure.
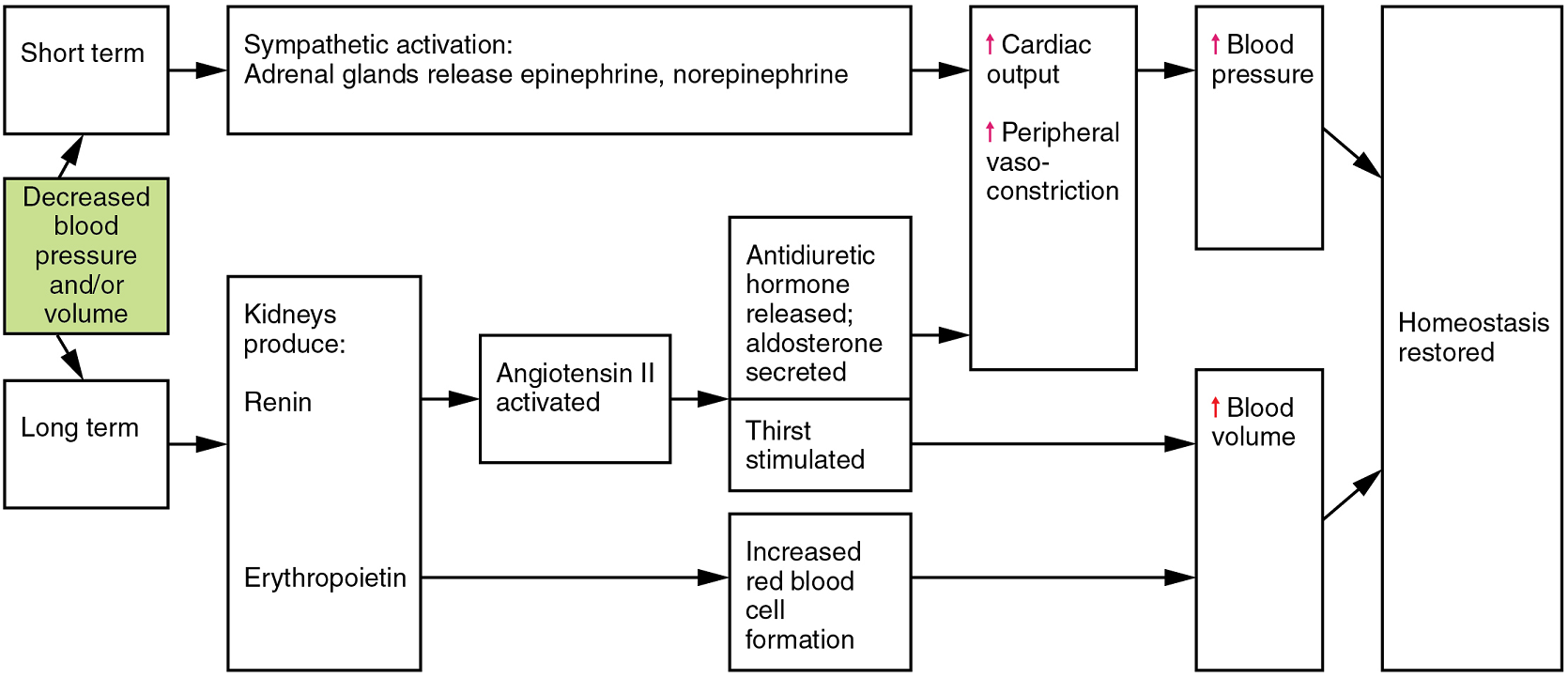
Erythropoietin: Erythropoietin (EPO) is released by the kidneys when blood flow and/or oxygen levels decrease. Erythropoietin stimulates the production of erythrocytes within the bone marrow. Erythrocytes are the major formed element of the blood and may contribute 40% or more to blood volume, a significant factor of viscosity, resistance, pressure, and flow. In addition, erythropoietin is a vasoconstrictor. Overproduction of erythropoietin or excessive intake of synthetic erythropoietin, often to enhance athletic performance, will increase viscosity, resistance, and pressure, and decrease flow in addition to its contribution as a vasoconstrictor.
Autoregulation of Perfusion: Autoregulation mechanisms require neither specialized nervous stimulation nor endocrine control. Rather, these are local, self-regulatory mechanisms that allow each region of tissue to adjust its blood flow, and thus its perfusion. These local mechanisms include chemical signals and myogenic controls.
Chemical Signals Involved in Autoregulation: Chemical signals work at the level of the precapillary sphincters to trigger either constriction or relaxation. Opening a precapillary sphincter allows blood to flow into that particular capillary, whereas constricting a precapillary sphincter temporarily shuts off blood flow to that region. The factors involved in regulating the precapillary sphincters include the following:
- Opening of the sphincter is triggered in response to decreased oxygen concentrations; increased carbon dioxide concentrations; increasing levels of lactic acid or other byproducts of cellular metabolism; increasing concentrations of potassium ions or hydrogen ions (falling pH); inflammatory chemicals such as histamines; and increased body temperature. These conditions in turn stimulate the release of NO, a powerful vasodilator, from endothelial cells.
- Contraction of the precapillary sphincter is triggered by the opposite levels of the regulators, which prompt the release of endothelins, powerful vasoconstricting peptides secreted by endothelial cells. Platelet secretions and certain prostaglandins may also trigger constriction.
Again, these factors alter tissue perfusion via their effects on the precapillary sphincter mechanism, which regulates blood flow to capillaries. Since the amount of blood is limited, not all capillaries can fill at once, so blood flow is allocated based upon the needs and metabolic state of the tissues as reflected in these parameters. Bear in mind, however, that dilation and constriction of the arterioles feeding the capillary beds is the primary control mechanism.
The Myogenic Response: The myogenic response is a reaction to the stretching of the smooth muscle in the walls of arterioles as changes in blood flow occur through the vessel. This may be viewed as a largely protective function against dramatic fluctuations in blood pressure and blood flow to maintain homeostasis. If perfusion of an organ is too low (ischemia), the tissue will experience low levels of oxygen (hypoxia). In contrast, excessive perfusion could damage the organ’s smaller and more fragile vessels. The myogenic response is a localized process that serves to stabilize blood flow in the capillary network that follows that arteriole. When blood flow is low, the vessel’s smooth muscle will be only minimally stretched. In response, it relaxes, allowing the vessel to dilate and thereby increase the movement of blood into the tissue. When blood flow is too high, the smooth muscle will contract in response to the increased stretch, prompting vasoconstriction that reduces blood flow.
Part 5: Circulatory Pathways
Virtually every cell, tissue, organ, and system in the body is impacted by the circulatory system. This includes the generalized and more specialized functions of transport of materials, capillary exchange, maintaining health by transporting leukocytes and various immunoglobulins (antibodies), hemostasis, regulation of body temperature, and helping to maintain acid-base balance. In addition to these shared functions, many systems enjoy a unique relationship with the circulatory system (Figure 39).
As you learn about the vessels of the systemic and pulmonary circuits, notice that many arteries and veins share the same names, parallel one another throughout the body, and are very similar on the right and left sides of the body. For example, you will find a pair of femoral arteries and a pair of femoral veins, with one vessel on each side of the body. In contrast, some vessels closer to the midline of the body, such as the aorta, are unique. Another phenomenon that can make the study of vessels challenging is that names of vessels can change with location. Like a street that changes name as it passes through an intersection, an artery or vein can change names as it passes an anatomical landmark. For example, the left subclavian artery becomes the axillary artery as it passes through the body wall and into the axillary region, and then becomes the brachial artery as it flows from the axillary region into the upper arm (or brachium).
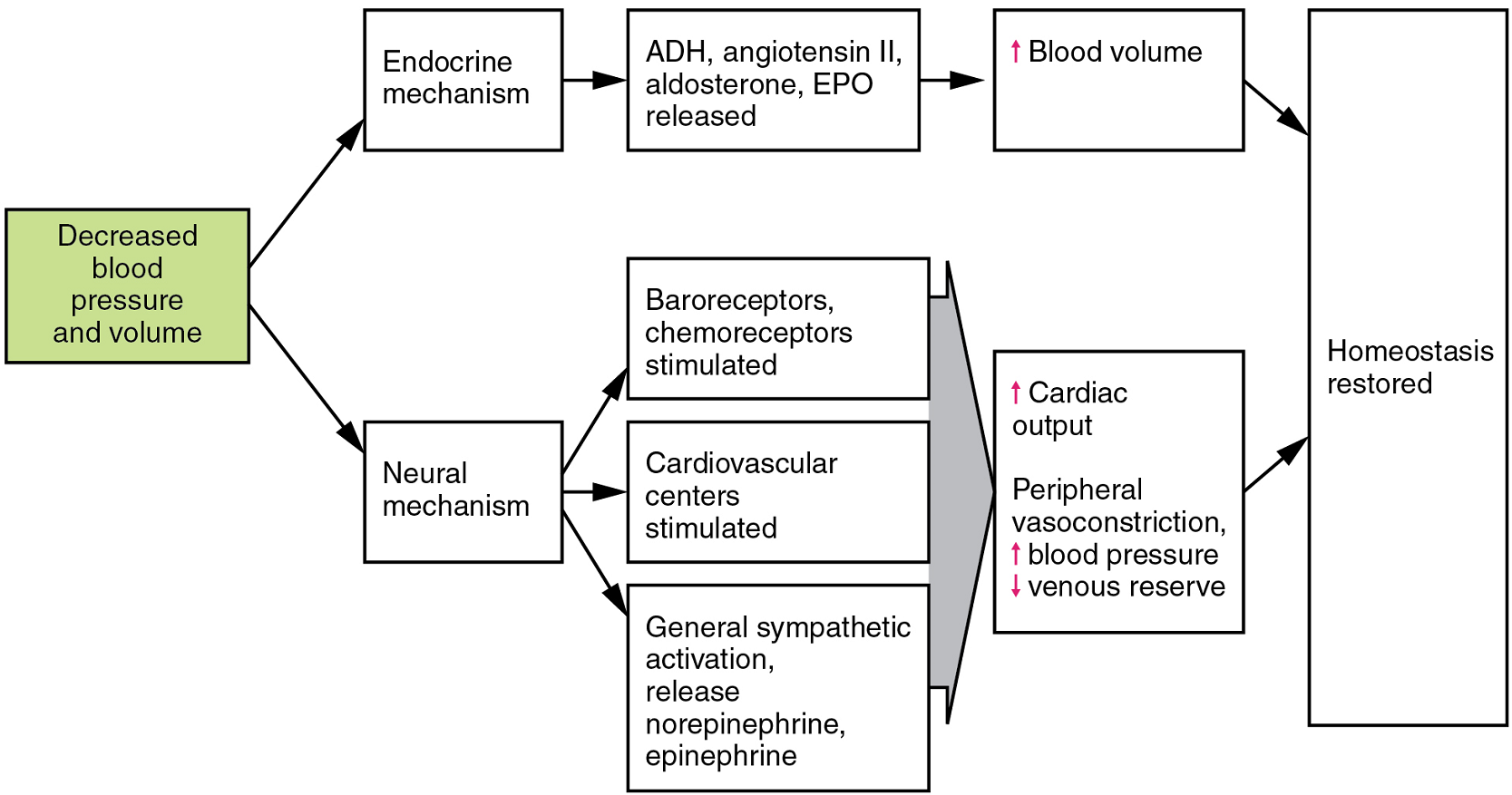
Pulmonary Circulation: Recall that blood returning from the systemic circuit enters the right atrium (Figure 40) via the superior and inferior venae cavae and the coronary sinus, which drains the blood supply of the heart muscle. These vessels will be described more fully later in this section. This blood is relatively low in oxygen and relatively high in carbon dioxide, since much of the oxygen has been extracted for use by the tissues and the waste gas carbon dioxide was picked up to be transported to the lungs for elimination. From the right atrium, blood moves into the right ventricle, which pumps it to the lungs for gas exchange. This system of vessels is referred to as the pulmonary circuit.
The single vessel exiting the right ventricle is the pulmonary trunk. At the base of the pulmonary trunk is the pulmonary semilunar valve, which prevents backflow of blood into the right ventricle during ventricular diastole. As the pulmonary trunk reaches the superior surface of the heart, it curves posteriorly and rapidly bifurcates (divides) into two branches, a left and a right pulmonary artery. To prevent confusion between these vessels, it is important to refer to the vessel exiting the heart as the pulmonary trunk, rather than also calling it a pulmonary artery.
The pulmonary arteries in turn branch many times within the lung, forming a series of smaller arteries and arterioles that eventually lead to the pulmonary capillaries. The pulmonary capillaries surround lung structures known as alveoli that are the sites of oxygen and carbon dioxide exchange.
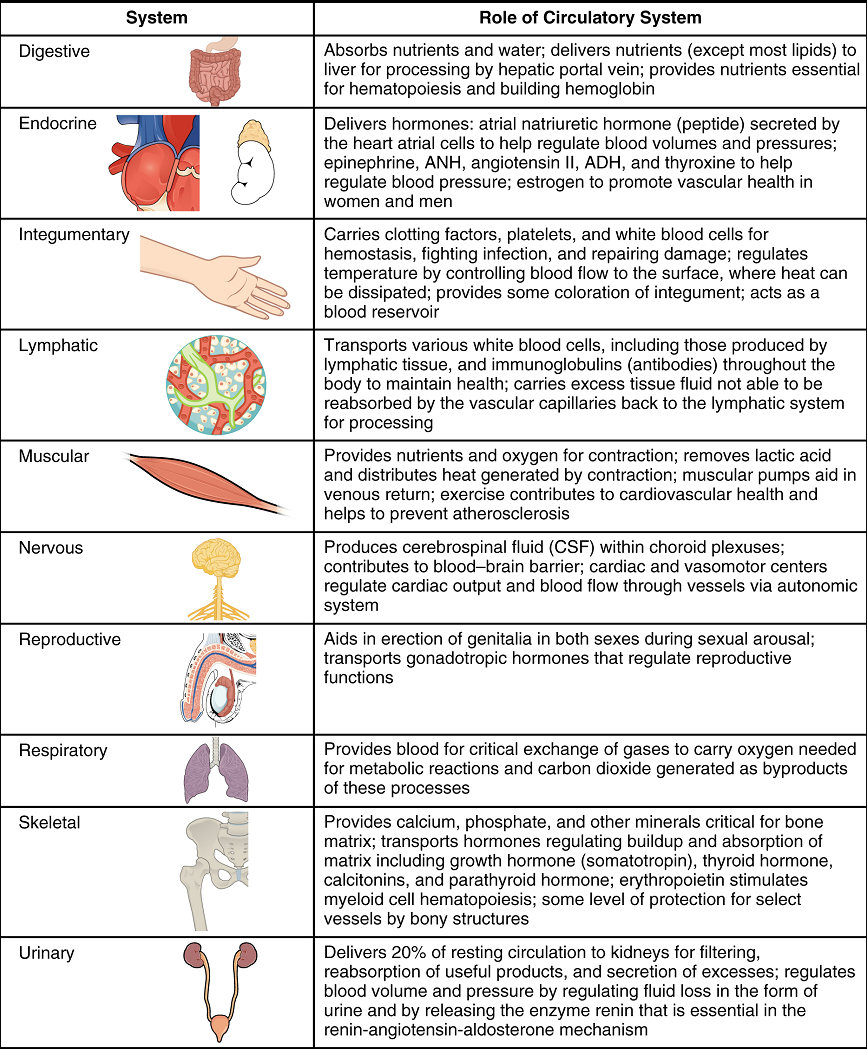
Once gas exchange is completed, oxygenated blood flows from the pulmonary capillaries into a series of pulmonary venules that eventually lead to a series of larger pulmonary veins. Four pulmonary veins, two on the left and two on the right, return blood to the left atrium. At this point, the pulmonary circuit is complete. Table 10 defines the major arteries and veins of the pulmonary circuit discussed in the text.
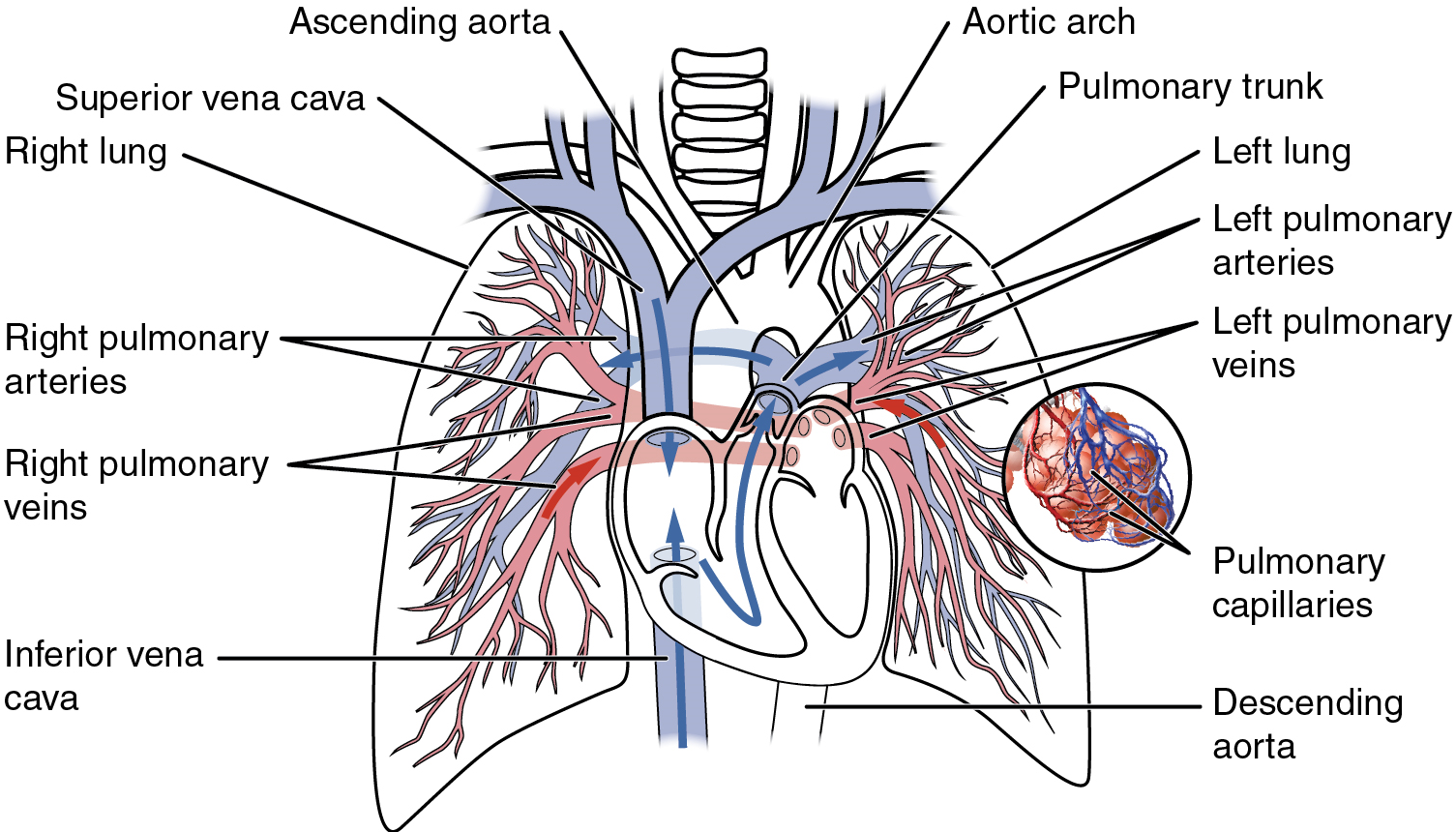

Overview of Systemic Arteries: Blood relatively high in oxygen concentration is returned from the pulmonary circuit to the left atrium via the four pulmonary veins. From the left atrium, blood moves into the left ventricle, which pumps blood into the aorta. The aorta and its branches—the systemic arteries—send blood to virtually every organ of the body (Figure 41).
| Vessel | Description |
|---|---|
| Pulmonary trunk | Single large vessel exiting the right ventricle (divides to form the right and left pulmonary arteries) |
| Pulmonary arteries (left pulmonary artery, right pulmonary artery) | Two vessels that form from the pulmonary trunk and lead to smaller arterioles and eventually to the pulmonary capillaries |
| Pulmonary veins (left superior pulmonary vein, left inferior pulmonary vein, right superior pulmonary vein, right inferior pulmonary vein) | Two sets of paired vessels (one pair from each side) that are formed from venules, leading blood away from the pulmonary capillaries to flow into the left atrium |
The Aorta: The aorta is the largest artery in the body (Figure 42). It arises from the left ventricle and eventually descends to the abdominal region, where it bifurcates at the level of the fourth lumbar vertebra into the two common iliac arteries. The aorta consists of the ascending aorta, the aortic arch, and the descending aorta (Table 11) which passes through the diaphragm, a landmark that divides into the superior thoracic and inferior abdominal components. Arteries originating from the aorta ultimately distribute blood to virtually all tissues of the body. At the base of the aorta is the aortic semilunar valve that prevents backflow of blood into the left ventricle while the heart is relaxing.
After exiting the heart, the ascending aorta moves in a superior direction for approximately 5 cm and ends at the sternal angle. Following this ascent, it reverses direction, forming a graceful arc to the left, called the aortic arch. The aortic arch descends toward the inferior portions of the body and ends at the level of the intervertebral disk between the fourth and fifth thoracic vertebrae. Beyond this point, the descending aorta continues close to the bodies of the vertebrae and passes through an opening in the diaphragm. Superior to the diaphragm, the aorta is called the thoracic aorta, and inferior to the diaphragm, it is called the abdominal aorta. The abdominal aorta terminates when it bifurcates into the two common iliac arteries at the level of the fourth lumbar vertebra. See Figure 55 for an illustration of the ascending aorta, the aortic arch, and the initial segment of the descending aorta plus major branches.
Coronary Circulation: The first vessels that branch from the ascending aorta are the paired coronary arteries (see Figure 42), which arise from two of the three sinuses in the ascending aorta just superior to the aortic semilunar valve. These sinuses contain the aortic baroreceptors and chemoreceptors critical to maintain cardiac function. The left coronary artery arises from the left posterior aortic sinus. The right coronary artery arises from the anterior aortic sinus. Normally, the right posterior aortic sinus does not give rise to a vessel.
The coronary arteries encircle the heart, forming a ring-like structure that divides into the next level of branches that supplies blood to the heart tissues.
Aortic Arch Branches: There are three major branches of the aortic arch: the brachiocephalic artery, the left common carotid artery, and the left subclavian (literally “under the clavicle”) artery. As you would expect based upon proximity to the heart, each of these vessels is classified as an elastic artery.
The brachiocephalic artery is located only on the right side of the body; there is no corresponding artery on the left. The brachiocephalic artery branches into the right subclavian artery and the right common carotid artery. The left subclavian and left common carotid arteries arise independently from the aortic arch but otherwise follow a similar pattern and distribution to the corresponding arteries on the right side (see Figure 42).
Each subclavian artery supplies blood to the arms, chest, shoulders, back, and central nervous system.
The common carotid artery divides into internal and external carotid arteries. The right common carotid artery arises from the brachiocephalic artery and the left common carotid artery arises directly from the aortic arch. The branches of the carotid arteries supply blood to numerous structures within the head and neck. Each internal carotid artery initially forms an expansion known as the carotid sinus, containing the carotid baroreceptors and chemoreceptors. Like their counterparts in the aortic sinuses, the information provided by these receptors is critical to maintaining cardiovascular homeostasis (see Figure 41).
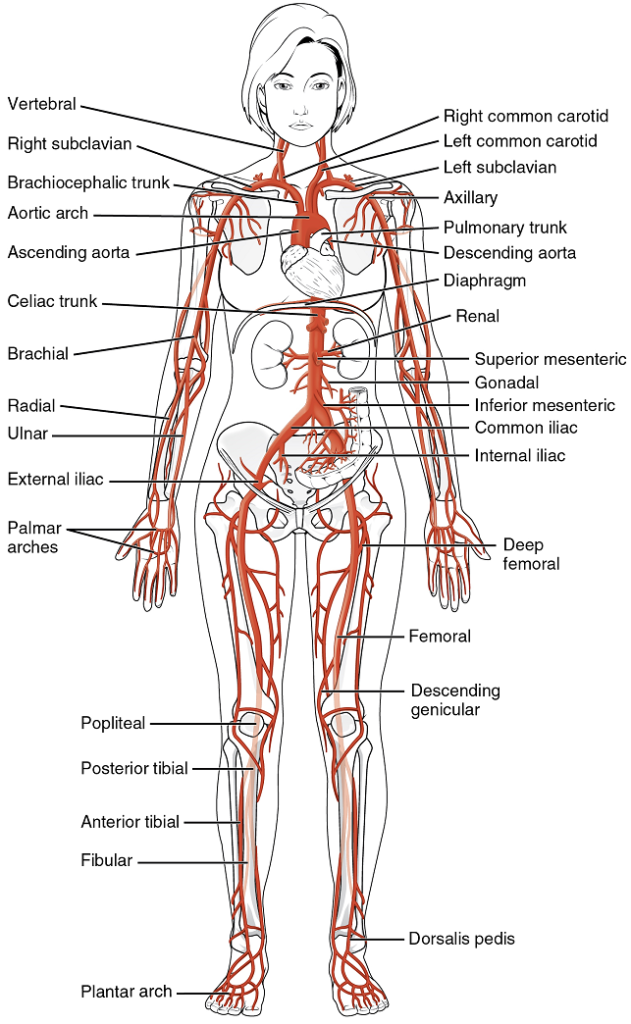
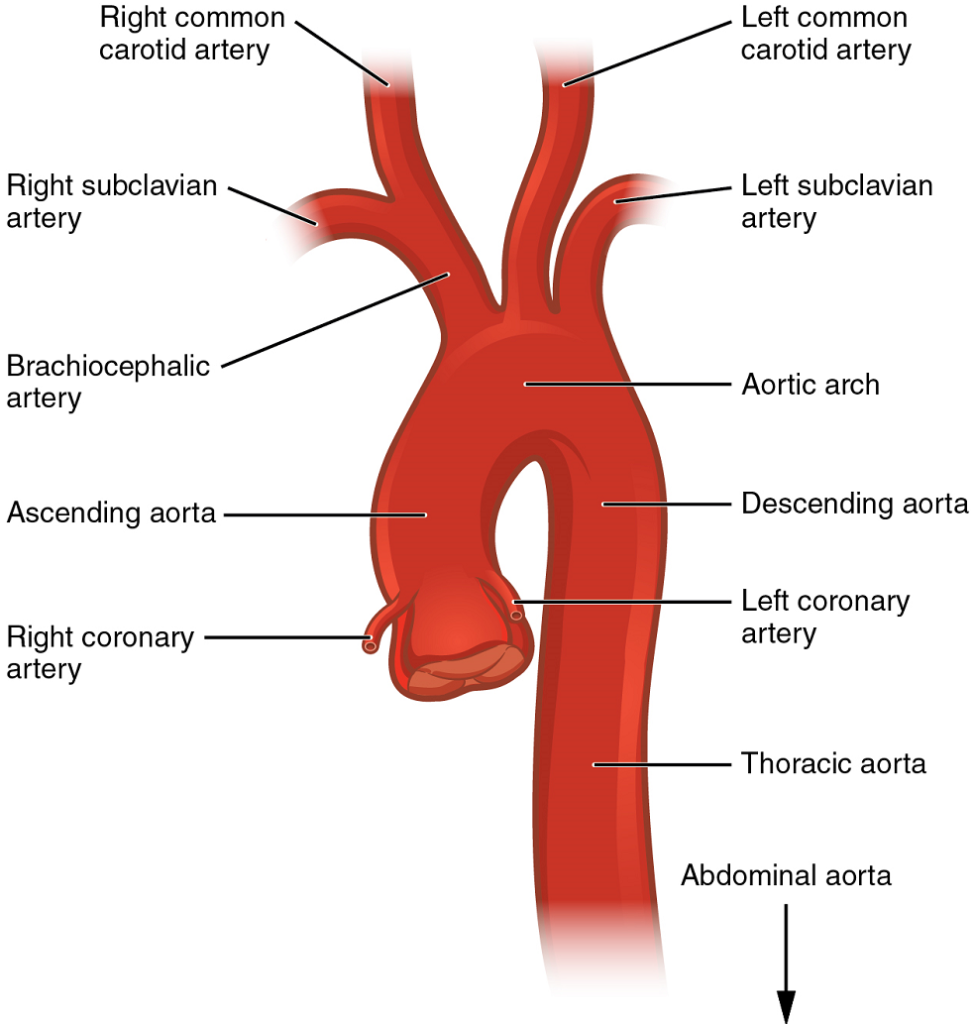
| Vessel | Description |
|---|---|
| Aorta | Largest artery in the body; originates from the left ventricle and descends to the abdominal region then bifurcates into the left and right common iliac arteries at the level of the fourth lumbar vertebra |
| Ascending aorta | Initial portion of the aorta; rises superiorly from the left ventricle for a distance of approximately 5 cm |
| Aortic arch | Graceful arc to the left that connects the ascending aorta to the descending aorta; ends at the intervertebral disk between the fourth and fifth thoracic vertebrae |
| Descending aorta | Continues inferiorly from the end of the aortic arch; subdivided into the thoracic aorta and the abdominal aorta |
| Thoracic aorta | Portion of the descending aorta superior to the aortic hiatus |
| Abdominal aorta | Portion of the aorta inferior to the aortic hiatus; ends at its bifurcation into the left common iliac artery and the right common iliac artery |
Thoracic Aorta and Major Branches: The thoracic aorta begins at the level of vertebra T5 and continues through to the diaphragm at the level of T12, initially traveling within the mediastinum to the left of the vertebral column. As it passes through the thoracic region, the thoracic aorta gives rise to several branches (Figure 43).
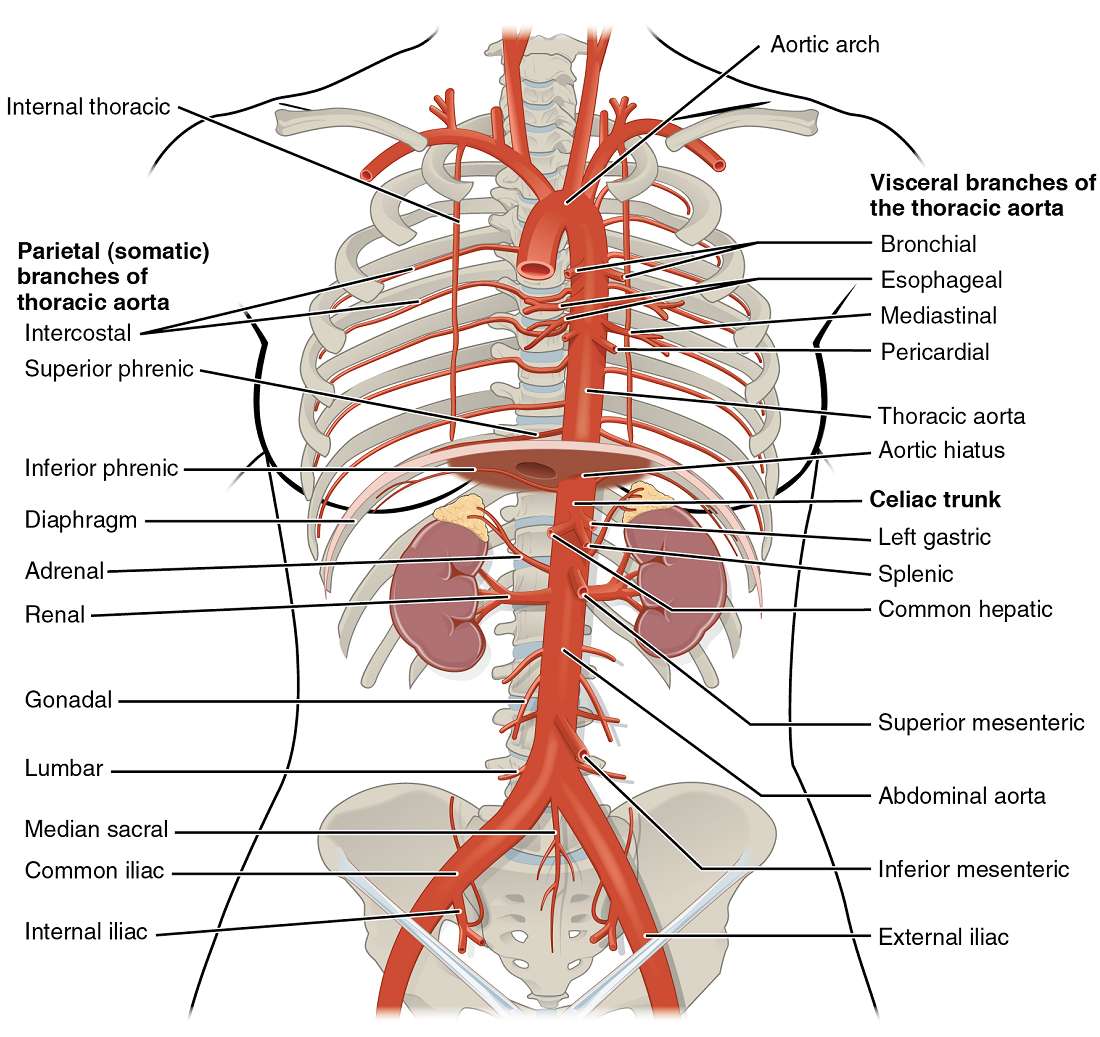
Abdominal Aorta and Major Branches: After crossing through the diaphragm, the thoracic aorta is called the abdominal aorta. This vessel remains to the left of the vertebral column and is embedded in adipose tissue behind the peritoneal cavity. It formally ends at approximately the level of vertebra L4, where it bifurcates to form the two (left and right) common iliac arteries. Before this division, the abdominal aorta gives rise to several important branches. The common iliac arteries provide blood to the pelvic region and ultimately to the lower limbs.
Arteries Serving the Upper Limbs: As each subclavian artery exits the thorax into the axillary region, it is renamed the axillary artery. Although each axillary artery does branch and supply blood to the region near the head of the humerus (via the humeral circumflex arteries), the majority of the vessel continues into the upper arm, or brachium, and becomes the brachial artery.
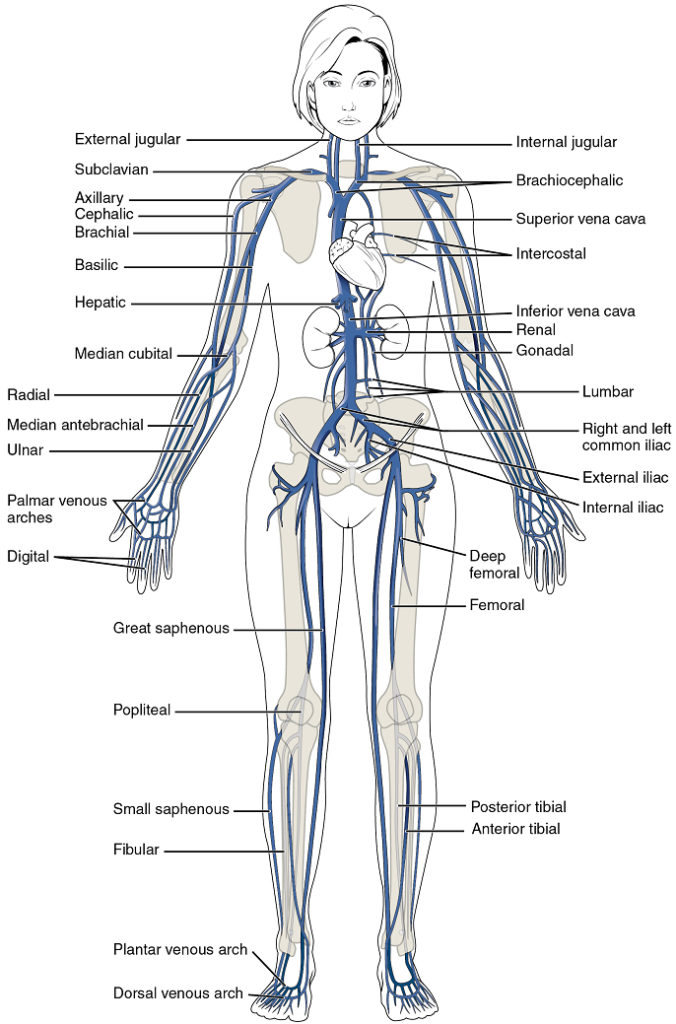
Arteries Serving the Lower Limbs: Each external iliac artery exits the body cavity and enters the femoral region of the lower leg. As it passes through the body wall, it is renamed the femoral artery. Each femoral artery gives rise to the genicular artery, which provides blood to the region of the knee. As each femoral artery passes posterior to the knee near the popliteal fossa, it is called the popliteal artery. Each popliteal artery branches into anterior and posterior tibial arteries.
Overview of Systemic Veins: Systemic veins return blood to the right atrium. Since the blood has already passed through the systemic capillaries, it will be relatively low in oxygen concentration (Figure 44).
The right atrium receives all of the systemic venous return. Most of the blood flows into either the superior vena cava or inferior vena cava. If you draw an imaginary line at the level of the diaphragm, systemic venous circulation from above that line will generally flow into the superior vena cava; this includes blood from the head, neck, chest, shoulders, and upper limbs. The exception to this is that most venous blood flow from the coronary veins flows directly into the coronary sinus and from there directly into the right atrium. Beneath the diaphragm, systemic venous flow enters the inferior vena cava, that is, blood from the abdominal and pelvic regions and the lower limbs.
The Superior Vena Cava: The superior vena cava drains most of the body superior to the diaphragm (Figure 45). On both the left and right sides, the subclavian vein forms when the axillary vein passes through the body wall from the axillary region. Each subclavian vein joins with the external and internal jugular veins from the head and neck to form the brachiocephalic vein.
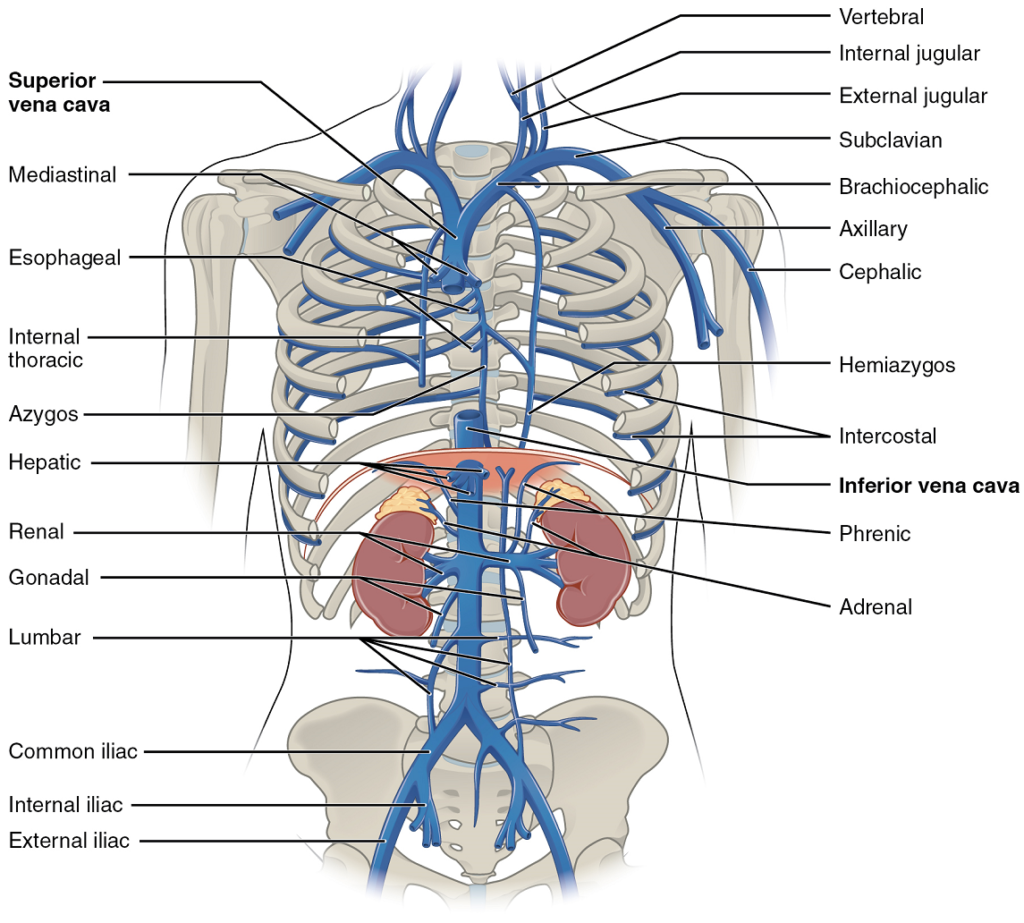
The Inferior Vena Cava: Most of the blood inferior to the diaphragm drains into the inferior vena cava before it is returned to the heart (see Figure 45). Lying just beneath the parietal peritoneum in the abdominal cavity, the inferior vena cava parallels the abdominal aorta, where it can receive blood from abdominal veins.
Veins Draining the Lower Limbs: As each femoral vein penetrates the body wall from the femoral portion of the upper limb, it becomes the external iliac vein, a large vein that drains blood from the leg to the common iliac vein (Figure 46). The pelvic organs and integument drain into the internal iliac vein on either side of the body, which forms from several smaller veins in the region, including the umbilical veins that run on either side of the bladder. The external and internal iliac veins combine near the inferior portion of the sacroiliac joint on either side to form the common iliac vein. In addition to blood supply from the external and internal iliac veins, the middle sacral vein drains the sacral region into the common iliac vein. Similar to the common iliac arteries, the two common iliac veins come together at the level of L5 to form the inferior vena cava.