Maintenance of the Body
Unit 8: The Urinary System
Unit outline
Part 1: The Urinary Tract
- Gross Anatomy of the Kidney
- External Anatomy
- Internal Anatomy
- Renal Hilum
- Nephrons and Vessels
- Ureters
- Bladder
- Urethra
Part 2: Microscopic Anatomy of the Kidney
- Cortex
- Nephrons
- Renal Corpuscle
- Proximal Convoluted Tubule (PCT)
- Loop of Henle
- Distal Convoluted Tubule (DCT)
- Collecting Ducts
Part 3: Physiology of Urine Formation
- Filtration, Tubular Reabsorption and Secretion
- Reabsorption and Secretion in the Proximal Convoluted Tubule
- Descending Loop
- Ascending Loop
- Reabsorption and Secretion in the Distal Convoluted Tubule
- Collecting Ducts and Recovery of Water
Part 4: Micturition Reflex
- Micturition
Part 5: Physical Characteristics of Urine
- Urinalysis
Learning Objectives
At the end of this unit, you should be able to:
I. Specify the location and describe the function(s) of each of the organs of the renal system.
II. Describe the gross anatomy of the kidney.
III. Describe the formation and composition of urine.
IV. Describe the hormonal control of urine production.
V. Describe the nervous control of micturition.
VI. Describe the physical properties and chemical composition of urine.
Learning Objectives and Guiding Questions
At the end of this unit, you should be able to complete all the following tasks, including answering the guiding questions associated with each task.
I. Specify the location and describe the function(s) of each of the organs of the renal system.
- Sketch a diagram with annotations specifying the location (using correct anatomical terms) and function (one-sentence summary for each structure) of the following structures:
- Kidneys
- Ureters
- Urinary bladder
- Urethra
II. Describe the gross anatomy of the kidney.
- Sketch a diagram of the kidneys showing its gross anatomy and blood supply. Your diagram should include all the following structures:
- Renal cortex
- Renal medulla
- Renal pelvis
- Renal artery
- Renal vein
- Describe in layman’s terms the appearance (size, shape, colour) of a human kidney.
- Describe the structure of a nephron and explain how the nephrons are arranged in the kidney.
- Use correct anatomical terms to describe the location and function (one-sentence summary for each structure) of all the following structures of/at a nephron:
- Afferent arteriole
- Proximal convoluted tubule
- Glomerulus
- Descending Loop of Henle
- Efferent arteriole
- Ascending Loop of Henle
- Peritubular capillaries
- Distal convoluted tubule
- Glomerular capsule
- Collecting duct
III. Describe the formation and composition of urine.
- Describe each of the three main processes that occur in a nephron that result in urine production from blood:
- Glomerular filtration
- Tubular reabsorption
- Tubular secretion
- Compare the composition of glomerular filtrate and urine in humans with respect to the following: glucose, proteins, salts, urea, uric acid, water.
IV. Describe the hormonal control of urine production.
- For aldosterone and anti-diuretic hormone, describe (where applicable):
- The stimulus or stimuli that causes their release (or increased release)
- The effect of the hormone on urine composition and volume
- The effect of the hormone on blood composition and volume
V. Describe the nervous control of micturition.
- Describe the involuntary and voluntary pathways that control the process of micturition, and the interaction between the two pathways.
VI. Describe the physical properties and chemical composition of urine.
- Describe normal urine in terms of the following:
- Volume voided in single day
- Specific gravity
- pH
- Chemical composition
Part 1: The Urinary Tract
Urine is a fluid of variable composition that requires specialized structures to remove it from the body safely and efficiently. Blood is filtered, and the filtrate is transformed into urine at a relatively constant rate throughout the day. This processed liquid is stored until a convenient time for excretion. All structures involved in the transport and storage of the urine are large enough to be visible to the naked eye.
Gross Anatomy of the Kidney: The kidneys lie on either side of the spine in the retroperitoneal space between the parietal peritoneum and the posterior abdominal wall, well protected by muscle, fat, and ribs. They are roughly the size of your fist. The kidneys are well vascularized, receiving about 25 percent of the cardiac output at rest.
External Anatomy: The left kidney is located at about the T12 to L3 vertebrae, whereas the right is lower due to slight displacement by the liver. Upper portions of the kidneys are somewhat protected by the eleventh and twelfth ribs (Figure 1). Each kidney weighs about 125–175 g in males and 115–155 g in females. They are about 11–14 cm in length, 6 cm wide, and 4 cm thick, and are directly covered by a fibrous capsule composed of dense, irregular connective tissue that helps to hold their shape and protect them.
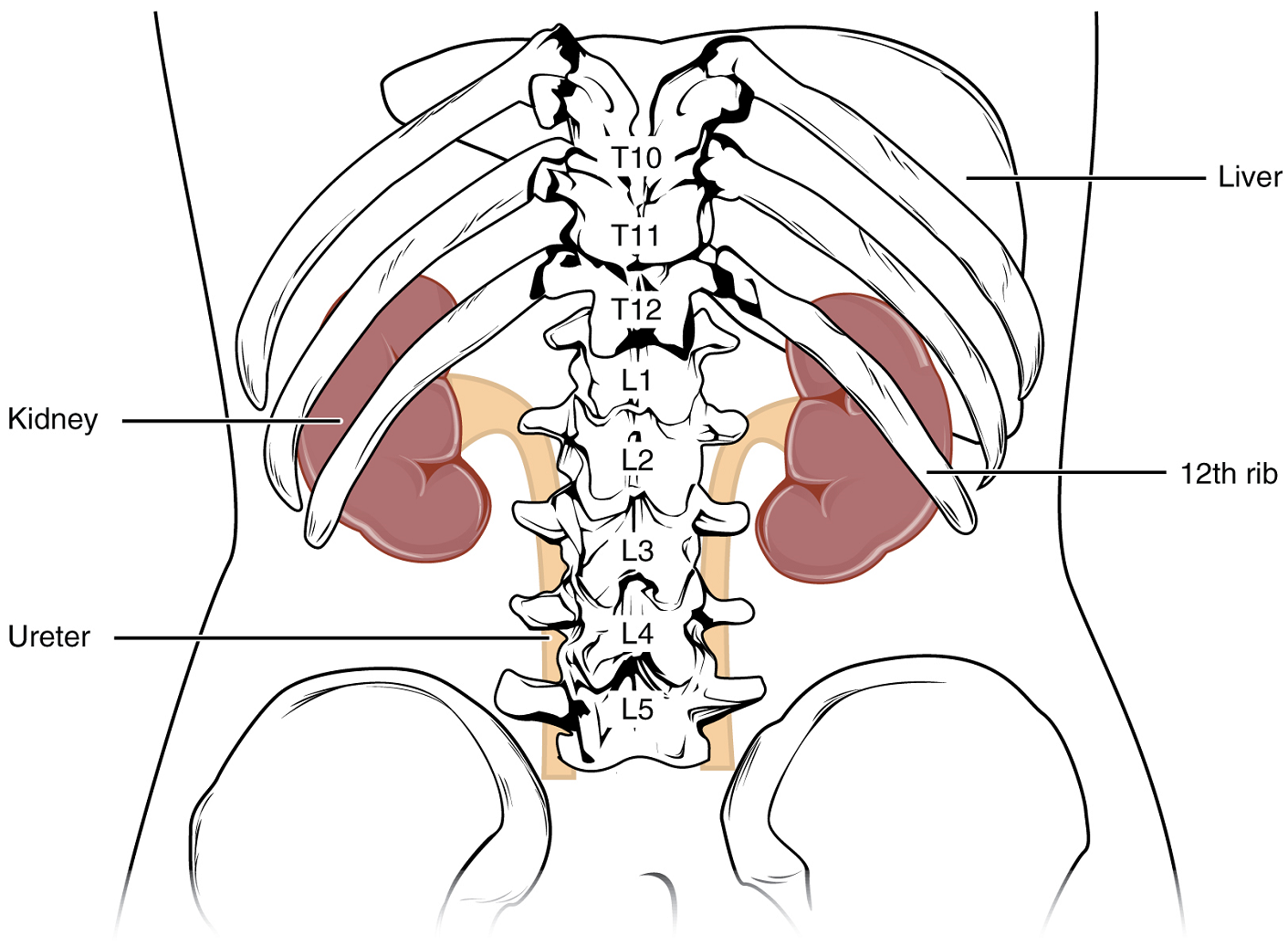
This capsule is covered by a shock-absorbing layer of adipose tissue called the renal fat pad. On the superior aspect of each kidney is the adrenal gland.
Internal Anatomy: A frontal section through the kidney reveals an outer region called the renal cortex and an inner region called the medulla (Figure 2). The renal columns are connective tissue extensions that radiate downward from the cortex through the medulla to separate the most characteristic features of the medulla, the renal pyramids and renal papillae. The papillae are bundles of collecting ducts that transport urine made by nephrons to the calyces of the kidney for excretion. The renal columns also serve to divide the kidney into 6–8 lobes and provide a supportive framework for vessels that enter and exit the cortex. The pyramids and renal columns taken together constitute the kidney lobes.
Renal Hilum: The renal hilum is the entry and exit site for structures serving the kidneys: blood vessels, nerves, lymphatics, and ureters. Emerging from the hilum is the renal pelvis, which is formed from the major and minor calyces in the kidney. The smooth muscle in the renal pelvis funnels urine via peristalsis into the ureter. The renal arteries form directly from the descending aorta, whereas the renal veins return cleansed blood directly to the inferior vena cava. The artery, vein, and renal pelvis are arranged in an anterior-to-posterior order.
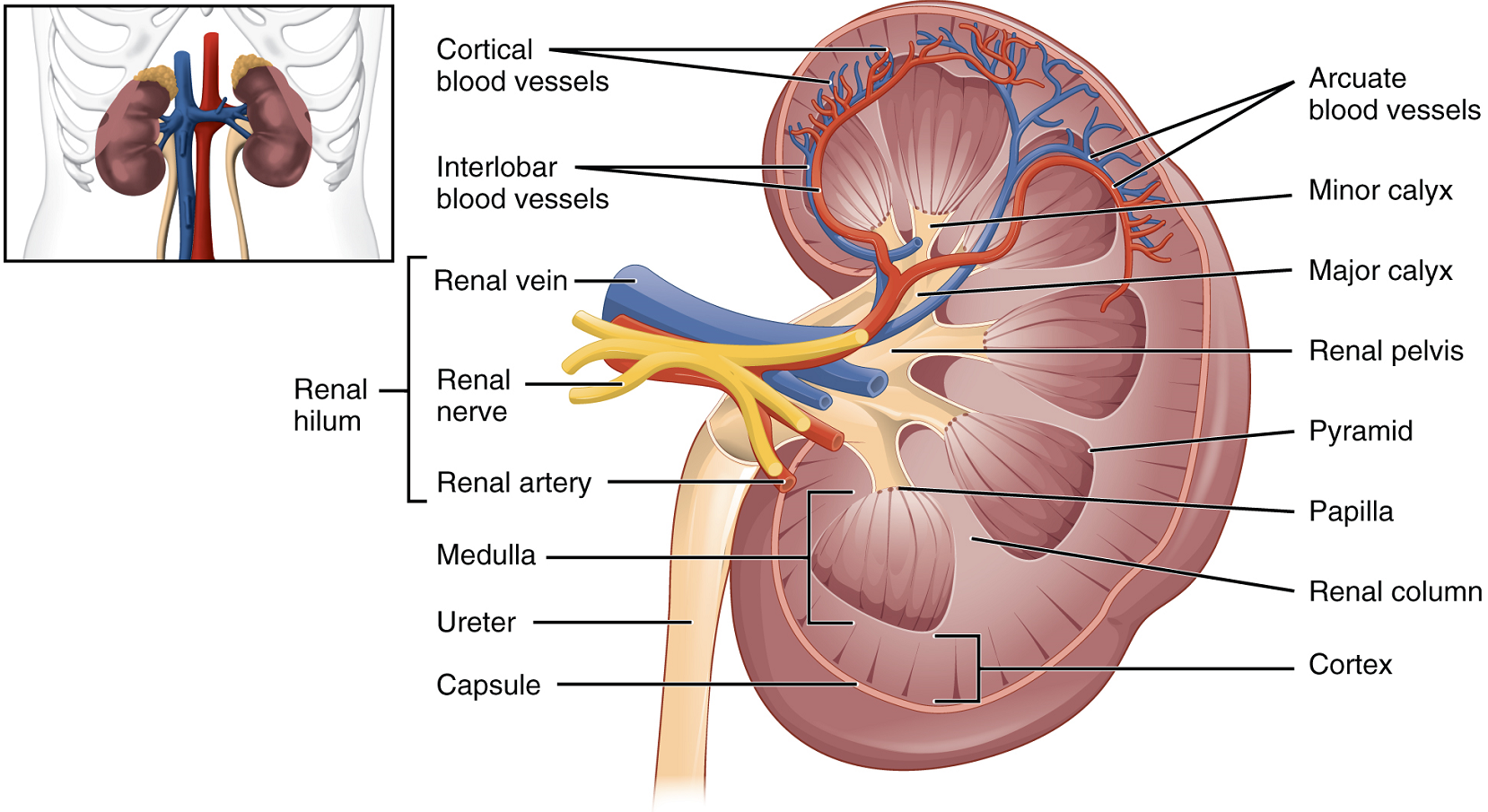
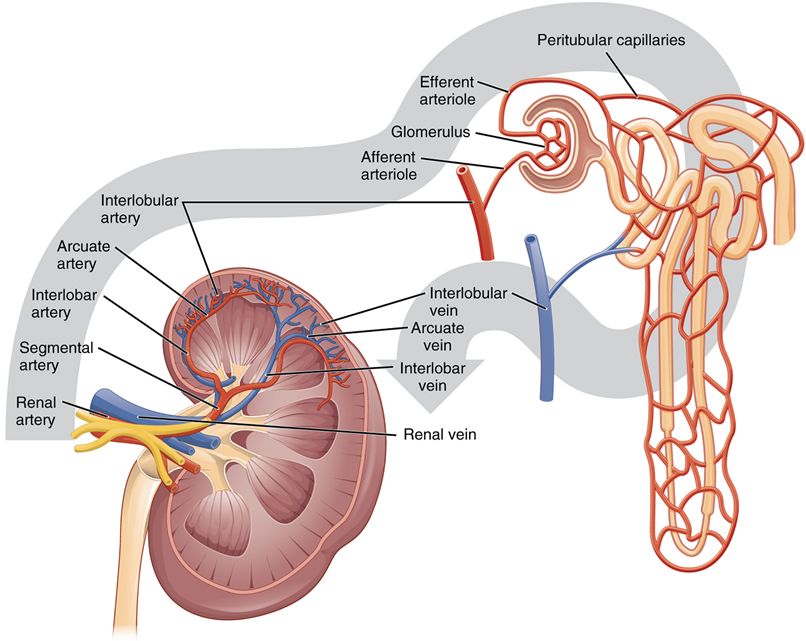
Nephrons and Vessels: The renal artery first divides into segmental arteries, followed by further branching to form interlobar arteries that pass through the renal columns to reach the cortex (Figure 3). The interlobar arteries, in turn, branch into arcuate arteries, cortical radiate arteries, and then into afferent arterioles. The afferent arterioles supply blood to about 1.3 million nephrons in each kidney.
Ureters: The kidneys and ureters are completely retroperitoneal, and the bladder has a peritoneal covering only over the dome. As urine is formed, it drains into the calyces of the kidney, which merge to form the funnel-shaped renal pelvis in the hilum of each kidney. The hilum narrows to become the ureter of each kidney. As urine passes through the ureter, it does not passively drain into the bladder but rather is propelled by waves of peristalsis.
As they approach the bladder, they turn medially and pierce the bladder wall obliquely. This is important because it creates a one-way valve (a physiological sphincter rather than an anatomical sphincter) that allows urine into the bladder but prevents reflux of urine from the bladder back into the ureter.
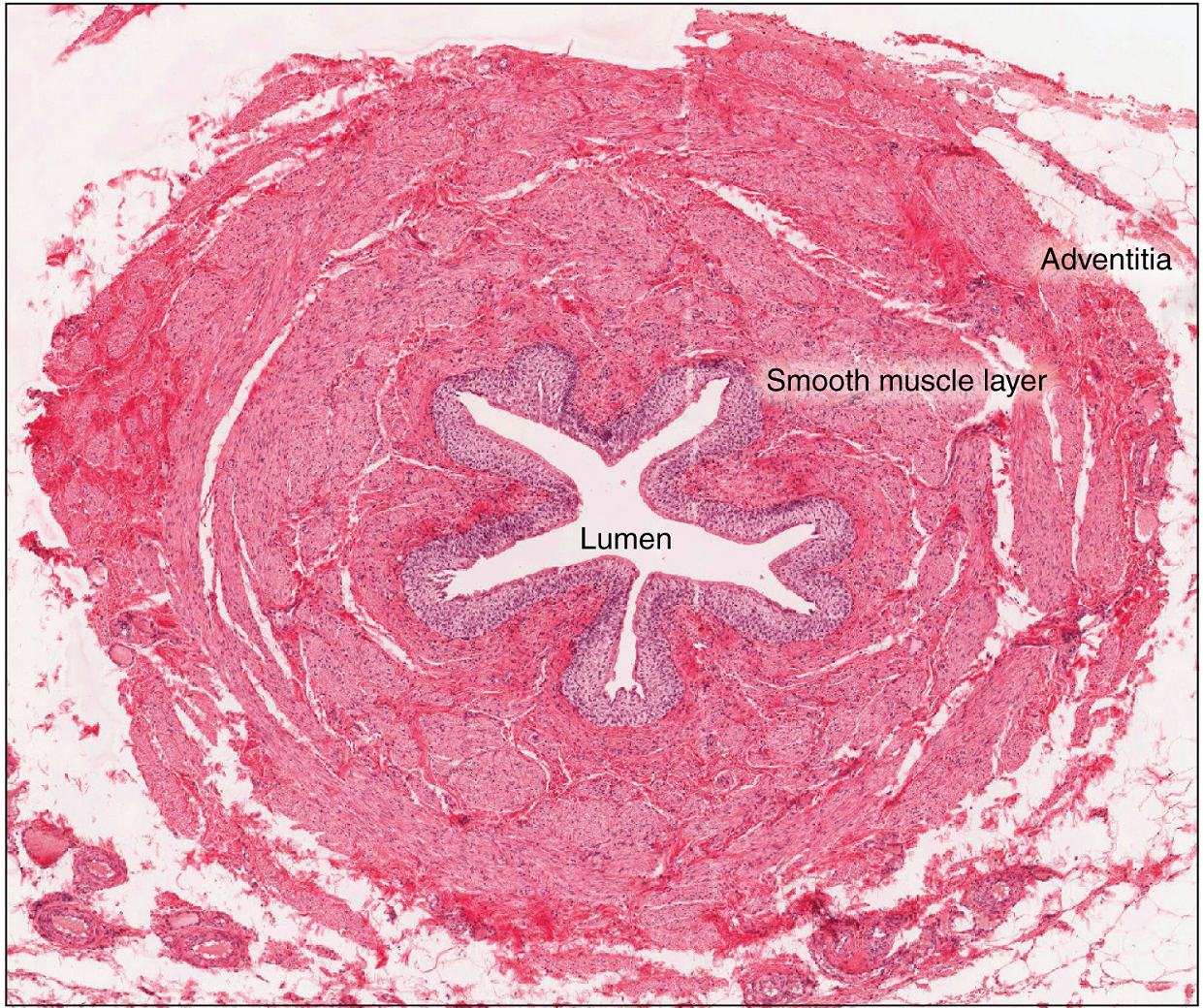
The ureters are approximately 30 cm long. The inner mucosa is lined with transitional epithelium (Figure 5) and scattered goblet cells that secrete protective mucus. The muscular layer of the ureter consists of longitudinal and circular smooth muscles that create the peristaltic contractions to move the urine into the bladder without the aid of gravity (Figure 4).
Bladder: The urinary bladder collects urine from both ureters (Figure 5). The bladder lies anterior to the uterus in females, posterior to the pubic bone and anterior to the rectum. In males, the anatomy is similar, minus the uterus, and with the addition of the prostate inferior to the bladder. The bladder is partially retroperitoneal (outside the peritoneal cavity).
The bladder is a highly distensible organ comprised of irregular crisscrossing bands of smooth muscle collectively called the detrusor muscle. The interior surface is made of transitional epithelium that is structurally suited for the large volume fluctuations of the bladder. Volumes in adults can range from nearly zero to 500–600 mL.
The detrusor muscle contracts with significant force in the young. The bladder’s strength diminishes with age, but voluntary contractions of abdominal skeletal muscles can increase intra-abdominal pressure to promote more forceful bladder emptying.
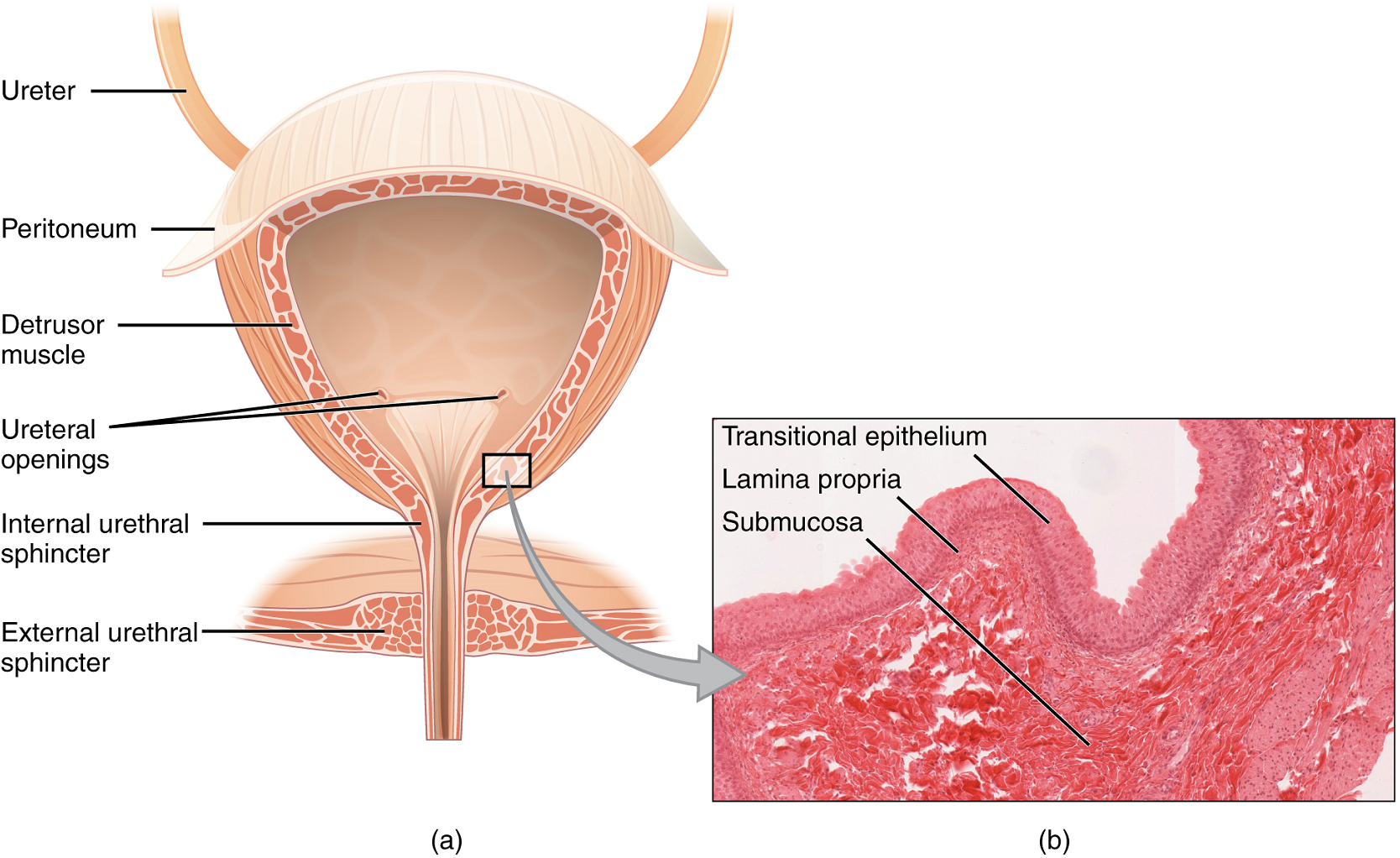
Urethra: The urethra transports urine from the bladder to the outside of the body for disposal. The urethra shows significant anatomic difference between males and females; all other urine transport structures are identical (Figure 6).
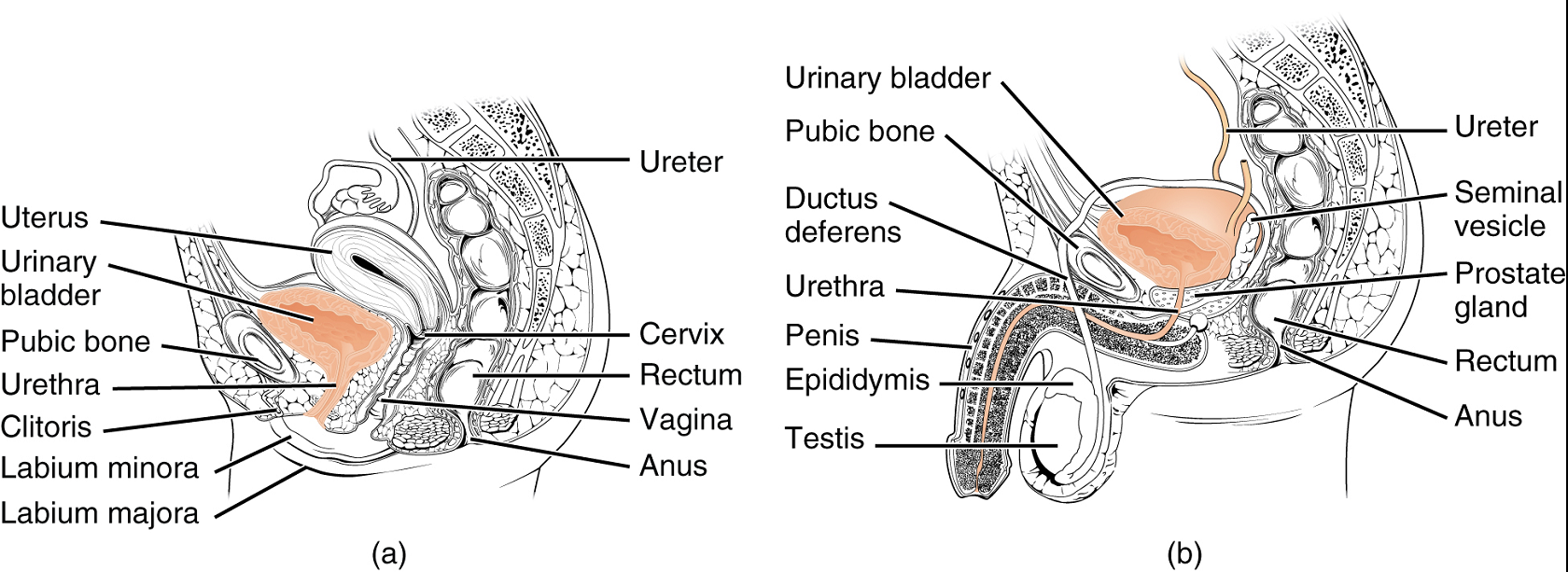
The urethra in both males and females begins inferior and central to the two ureteral openings. (Figure 5a). In both males and females, the proximal urethra is lined by transitional (stratified epithelium which can contract and expand) epithelium, whereas the terminal portion is a nonkeratinized, stratified squamous epithelium. Voiding is regulated by an involuntary autonomic nervous system-controlled internal urinary sphincter, consisting of smooth muscle and voluntary skeletal muscle that forms the external urinary sphincter below it.
Female Urethra: Its short length, about 4 cm, is less of a barrier to fecal bacteria than the longer male urethra and the best explanation for the greater incidence of urinary tract infections in women.
Male Urethra: The male urethra passes through the prostate gland immediately inferior to the bladder before passing below the pubic symphysis (Figure 6b). The urethra passes through the deep muscles of the perineum and exits at the tip (external urethral orifice) of the penis. Mucous glands are found along much of the length of the urethra and protect the urethra from extremes of urine pH. Male urethra also serves as the duct through which semen (fluid containing sperms) is discharged.
Part 2: Microscopic Anatomy of the Kidney
The renal structures that conduct the essential work of the kidney cannot be seen by the naked eye.
Cortex: In a dissected kidney, it is easy to identify the cortex; it appears lighter in color compared to the rest of the kidney. All of the renal corpuscles as well as both the proximal convoluted tubules and distal convoluted tubules are found here. Some nephrons have a short loop of Henle that does not dip beyond the cortex. These nephrons are called cortical nephrons. About 15 percent of nephrons have long loops of Henle that extend deep into the medulla and are called juxtamedullary nephrons.
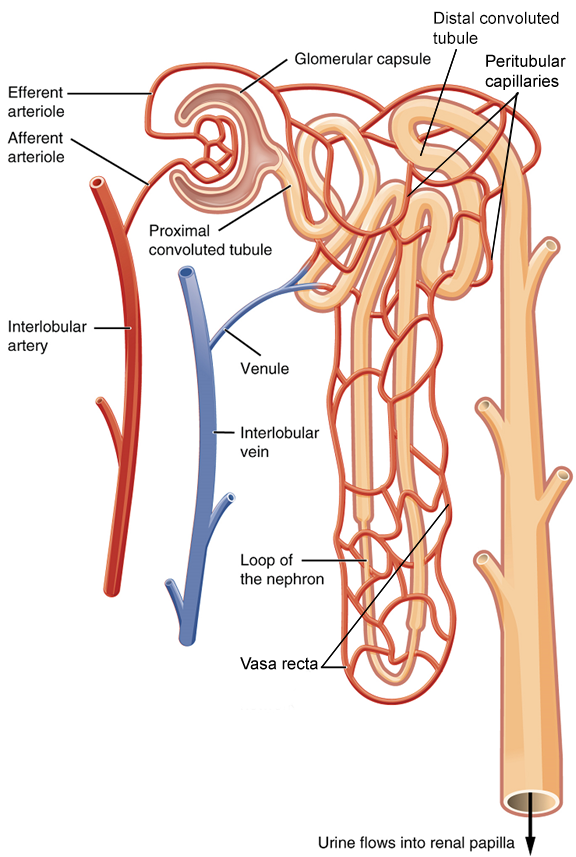
Nephrons: Nephrons are the functional unit of the kidney; they take a simple filtrate of the blood and modify it into urine. They cleanse the blood and maintain the levels of blood chemical components within physiological values. The afferent arterioles form a tuft of high-pressure capillaries, the glomerulus. The rest of the nephron consists of a continuous tubule, the proximal end of which surrounds the glomerulus—this is Bowman’s capsule or glomerular capsule. As mentioned earlier, these glomerular capillaries filter the blood based on particle size.
Renal Corpuscle: The glomerulus and Bowman’s capsule together form the renal corpuscle. After passing through the renal corpuscle, the capillaries form a second arteriole, the efferent arteriole (Figure 7). These will next form a capillary network around the more distal portions of the nephron tubule, the peritubular capillaries and vasa recta, before returning to the venous system. As the glomerular filtrate progresses through the nephron, these capillary networks regain most of the solutes and water and return them to the circulation.
The glomerulus is a high-pressure capillary bed between afferent and efferent arterioles. Bowman’s capsule surrounds the glomerulus to form a lumen, and captures and directs this filtrate to the proximal convoluted tubule. As blood passes through the glomerulus, 10 to 20 percent of the fluid that moves into the glomerulus is captured by Bowman’s capsule and funneled to the proximal convoluted tubule.
Proximal Convoluted Tubule (PCT): Filtered fluid collected by Bowman’s capsule enters into the proximal convoluted tubule. It is called convoluted due to its tortuous path. Simple cuboidal cells form this tubule with prominent microvilli on the luminal surface, forming a brush border. These microvilli create a large surface area to maximize the absorption and secretion of solutes (Na+, Cl–, glucose, etc.), the most essential function of this portion of the nephron. These cells actively transport ions across their membranes.
Loop of Henle: The descending and ascending portions of the loop of Henle (nephron loop) are, the continuations of the same tubule. The descending loop of Henle consists of an initial short, thick portion and long, thin portion, whereas the ascending loop consists of an initial short, thin portion followed by a long, thick portion. The descending thick portion consists of simple cuboidal epithelium similar to that of the proximal convoluted tubule. The descending and ascending thin portions consist of simple squamous epithelium. these are important differences, since different portions of the loop have different permeabilities for solutes and water. The ascending thick portion consists of simple cuboidal epithelium similar to the distal convoluted tubule.
Distal Convoluted Tubule (DCT): The distal convoluted tubule, like the proximal convoluted tubule, is very tortuous and formed by simple cuboidal epithelium, but it is shorter than the proximal convoluted tubule.
Collecting Ducts: The collecting ducts are continuous with the nephron but not generally considered part of it. In fact, each duct collects filtrate from several nephrons for final modification. Collecting ducts merge as they descend deeper in the medulla to form about 30 terminal ducts, which empty at a papilla. They are lined with simple squamous epithelium with receptors for antidiuretic hormone (ADH). When stimulated by antidiuretic hormone, these cells will allow water to pass from the duct lumen through the cells and into the interstitial spaces to be reabsorbed by the vasa recta. This process allows for the recovery of large amounts of water from the filtrate back into the blood.

Part 3: Physiology of Urine Formation – Filtration, Tubular Reabsorption, and Secretion
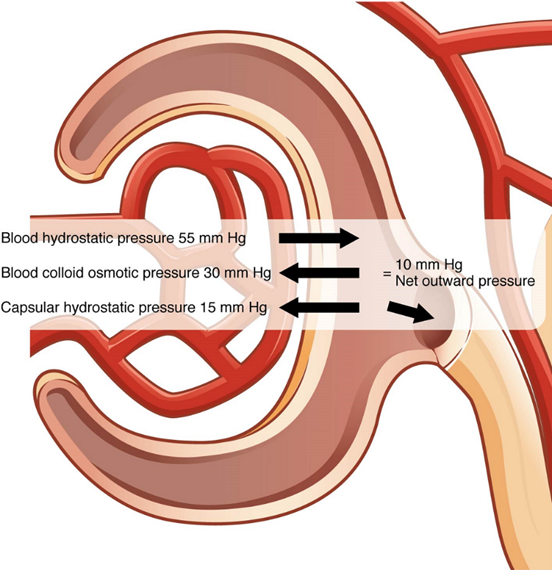
Different parts of the nephron utilize specific processes to produce urine: filtration, reabsorption, and secretion. The volume of filtrate formed by both kidneys per minute is termed the glomerular filtration rate (GFR). The heart pumps about 5 L blood per min under resting conditions. Approximately 20 percent or one liter enters the kidneys to be filtered. Ninety-nine percent of this filtrate is returned to the circulation by reabsorption so that only about 1–2 liters of urine are produced per day. The glomerular filtration rate is influenced by the hydrostatic pressure and colloid osmotic pressure on either side of the capillary membrane of the glomerulus (Figure 8). Filtration occurs as pressure forces fluid and solutes through a semipermeable barrier with the solute movement constrained by particle size. Hydrostatic pressure is the pressure produced by a fluid against a surface. If you have a fluid on both sides of a barrier, both fluids exert a pressure in opposing directions. Net fluid movement will be in the direction of the lower pressure.
Up to 180 liters per day passes through the nephrons of the kidney. The renal corpuscle filters the blood to create a filtrate that differs from blood mainly in the absence of cells and large proteins. From this point to the ends of the collecting ducts, the filtrate or forming urine is undergoing modification through secretion and reabsorption before true urine is produced. The first point at which the forming urine is modified is in the proximal convoluted tubule. Here, some substances are reabsorbed, whereas others are secreted. Note the use of the term “reabsorbed.” All of these substances were “absorbed” in the digestive tract—99 percent of the water and most of the solutes filtered by the nephron must be reabsorbed. Water and substances that are reabsorbed are returned to the circulation by the peritubular and vasa recta capillaries (Figure 7). Movement of water into the peritubular capillaries and vasa recta will be influenced primarily by osmolarity and concentration gradients. Sodium is actively pumped out of the proximal convoluted tubule into the interstitial spaces between cells and diffuses down its concentration gradient into the peritubular capillaries. As it does so, water will follow passively to maintain an isotonic fluid environment inside the capillary. Most of that fluid and its contents are reabsorbed. That recovery occurs in the proximal convoluted tubule, loop of Henle, distal convoluted tubule, and the collecting ducts (Table 1 and Figure 9). Various portions of the nephron differ in their capacity to reabsorb water and specific solutes. While much of the reabsorption and secretion occur passively based on concentration gradients, the amount of water that is reabsorbed or lost is tightly regulated. This control is exerted directly by antidiuretic hormone and another hormone aldosterone. Most water is recovered in the proximal convoluted tubule, loop of Henle, and distal convoluted tubule. About 10 percent (about 18 L) reaches the collecting ducts. The collecting ducts, under the influence of antidiuretic hormone, can recover almost all of the water passing through them.
Mechanisms by which substances move across membranes for reabsorption or secretion include active transport, diffusion, facilitated diffusion, secondary active transport, and osmosis. These were discussed in an earlier chapter, and you may wish to review them.
Most of the Ca2+, Na+, glucose, and amino acids must be reabsorbed by the nephron to maintain homeostatic plasma concentrations. Other substances, such as urea, K+, ammonia (NH3), creatinine, and some drugs are secreted into the filtrate as waste products. Acid–base balance is maintained through actions of the lungs and kidneys. In the case of urea, about 50 percent is passively reabsorbed by the proximal convoluted tubule.
A few of the substances that are transported with Na+ include Cl–, Ca2+, amino acids, glucose, and PO43−. Sodium is actively exchanged for K+ using ATP on the basal membrane.
About 67 percent of the water, Na+, and K+ entering the nephron is reabsorbed in the proximal convoluted tubule and returned to the circulation. Almost 100 percent of glucose, amino acids, and other organic substances such as vitamins are normally recovered here.
More substances move across the membranes of the proximal convoluted tubule than any other portion of the nephron.
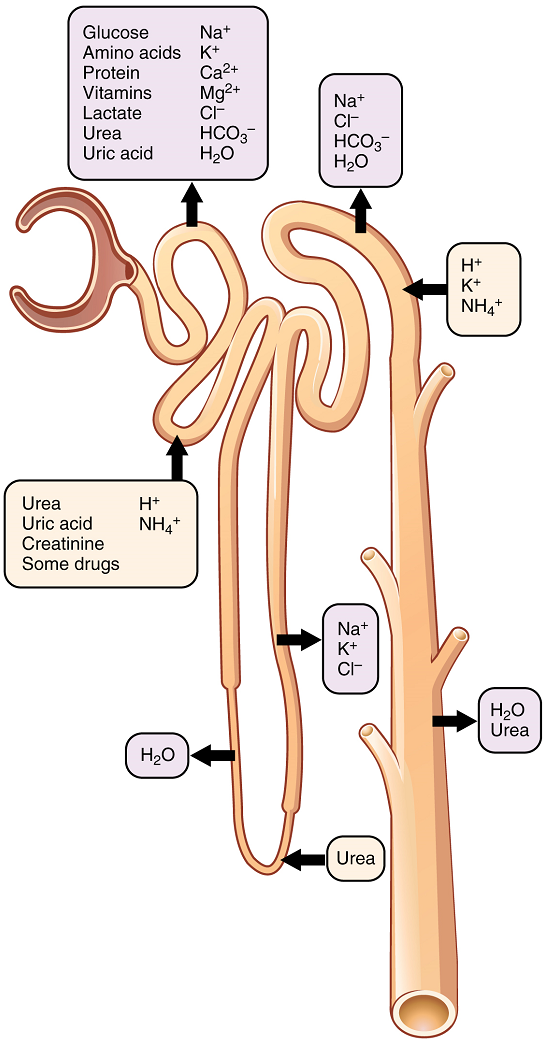
| Substance | Proximal convoluted tubule | Loop of Henle | Distal convoluted tubule | Collecting ducts |
|---|---|---|---|---|
| Glucose | ~100% reabsorbed; secondary active transport with Na+ | |||
| Amino acids, oligopeptides, proteins | ~100% reabsorbed; symport with Na+ | |||
| Vitamins | Reabsorbed | |||
| Lactate | Reabosorbed | |||
| Creatinine | Secreted | |||
| Urea | 50% reabsorbed by diffusion; also secreted | Secretion by diffusion in descending limb | Reabsorption by diffusion in medullary collecting ducts | |
| Sodium | 65% reabsorbed | 25% reabsorbed by active transport in thick ascending limb | 5% reabsorbed by active transport | 5% reabsorbed by active transport, stimulated by aldosterone |
| Chloride | Reabsorbed by symport with Na+ and diffusion | Reabsorbed by diffusion in thin & thick ascending limb | Reabsorbed by diffusion | Reabsorbed by symport |
| Water | 67% reabsorbed by osmosis (follows solutes) | 15% reabsorbed by osmosis in descending limb | In the presence of antidiuretic hormone, 8% reabsorbed by osmosis | Variable amounts reabsorbed by osmosis; regulated by antidiuretic hormone |
| Bicarbonate | 80-90% reabsorbed by symport with Na+ | Reabsorbed in ascending limb by symport with Na+ and antiport with Cl– | Reabsorbed by antiport with Cl– | |
| Hydrogen ions (H+) | Secreted by diffusion | Secreted by active transport | Secreted by active transport | |
| Ammonium (NH4+) | Secreted by diffusion | Secreted by diffusion | Secreted by diffusion | |
| Bicarbonate (HCO3–) | Reabsorbed by diffusion | Reabsorbed by diffusion in ascending limb | Reabsorbed by diffusion | Reabsorbed by antiport with Na+ |
| Some drugs | Secreted | Secreted by active transport | Secreted by active transport | |
| Potassium | 65% reabsorbed by diffusion | 20% reabsorbed by symport in thick ascending limb | Secreted by active transport | Secreted by active transport, regulated by aldosterone |
| Calcium | Reabsorbed by diffusion | Reabsorbed by diffusion in thick ascending limb | In the presence of parathyroid hormone, reabsorbed by active transport | |
| Magnesium | Reabsorbed by diffusion | Reabsorbed by diffusion in thick ascending limb | Reabsorbed | |
| Phosphate | 85% reabsorbed by diffusion; inhibited by parathyroid hormone | Reabsorbed by diffusion |
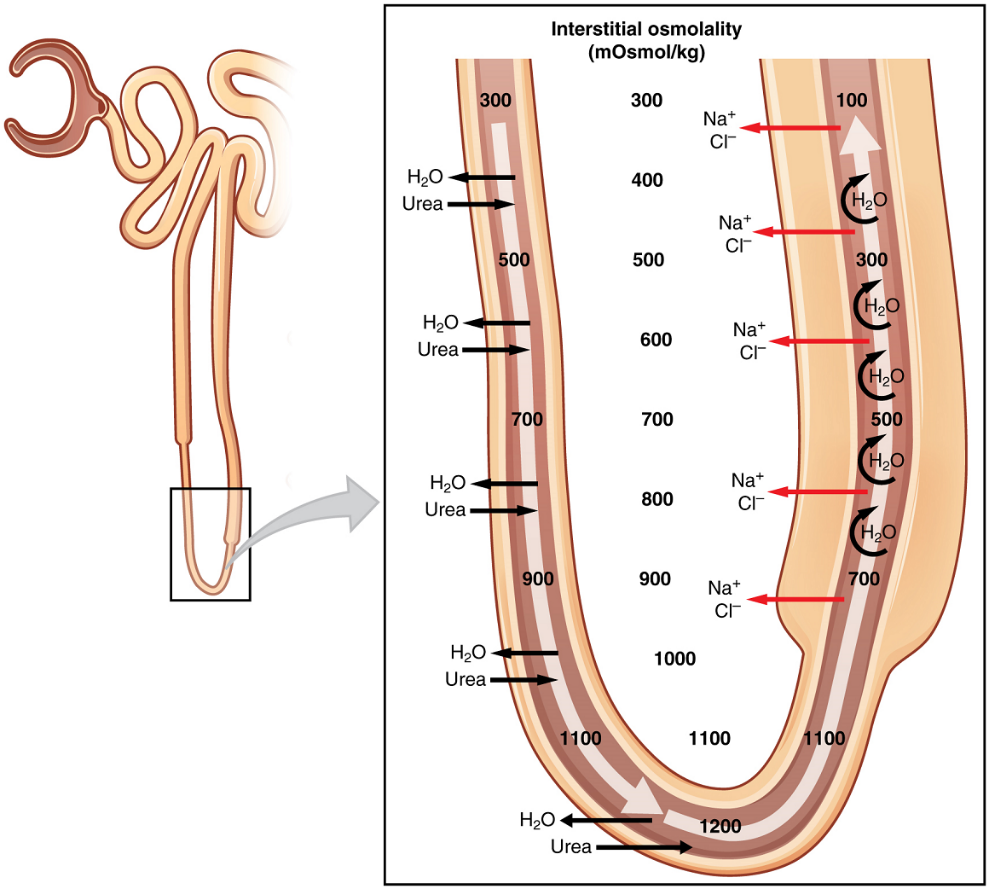
Reabsorption and Secretion in the Loop of Henle: The descending and ascending portions of the loop are highly specialized to enable recovery of much of the Na+ and water that were filtered by the glomerulus. As the forming urine moves through the loop, the osmolarity will change from isosmotic with blood to both a very hypertonic solution and a very hypotonic solution. These changes are accomplished by osmosis in the descending limb and active transport in the ascending limb. Solutes and water recovered from these loops are returned to the circulation by way of the vasa recta.
Descending Loop: The majority of the descending loop is comprised of simple squamous epithelial cells; to simplify the function of the loop, this discussion focuses on these cells. These membranes have permanent aquaporin channel proteins that allow unrestricted movement of water from the descending loop into the surrounding interstitial fluid. Approximately 15 percent of the water and modest amounts of urea, Na+, and other ions are recovered here.
Most of the solutes that were filtered in the glomerulus have now been recovered along with a majority of water, about 82 percent. As the forming urine enters the ascending loop, major adjustments will be made to the concentration of solutes to create urine.
Ascending Loop: The thick portion is lined with simple cuboidal epithelium It is completely impermeable to water due to the absence of aquaporin proteins, but ions, mainly Na+, are actively pumped out of the loop by large numbers of the Na+/K+ ATPase pump. This causes the removal of Na+ while retaining water leading to a hypotonic filtrate by the time it reaches the distal convoluted tubule.
The Na+/K+ ATPase pumps allow Na+ to be actively pumped into the interstitial fluid, Cl– follows the Na+ from the lumen into the interstitial fluid.
In addition, collecting ducts have urea pumps that actively pump urea into the interstitial spaces. This results in the recovery of Na+ to the circulation via the vasa recta and creates a high osmolar environment in the depths of the medulla.
At the same time that water is freely diffusing out of the descending loop through aquaporin channels into the interstitial spaces of the medulla, urea freely diffuses into the lumen of the descending loop as it descends deeper into the medulla, much of it to be reabsorbed from the forming urine when it reaches the collecting duct. Thus, the movement of Na+ and urea into the interstitial spaces by these mechanisms creates the hyperosmotic environment of the medulla. The net result of this is to recover both water from the descending limb of the loop of Henle and Na+ in the circulation (Figure 10).
Reabsorption and Secretion in the Distal Convoluted Tubule: Approximately 80 percent of filtered water has been recovered by the time the dilute forming urine enters the distal convoluted tubule. The distal convoluted tubule will recover another 10–15 percent before the forming urine enters the collecting ducts. Aldosterone increases the amount of Na+/K+ ATPase in the basal membrane of the distal convoluted tubule and collecting duct. The movement of Na+ out of the lumen of the collecting duct creates a negative charge that promotes the movement of Cl– out of the lumen into the interstitial space. Peritubular capillaries receive the solutes and water, returning them to the circulation.
Cells of the distal convoluted tubule also recover Ca2+ from the filtrate. In addition, as Na+ is pumped out of the cell, the resulting electrochemical gradient attracts Ca2+ into the cell. Any Ca2+ not reabsorbed at this point is lost in the urine.
Collecting Ducts and Recovery of Water: Regulation of urine volume and osmolarity are major functions of the collecting ducts. By varying the amount of water that is recovered, the collecting ducts play a major role in maintaining the body’s normal osmolarity. If the blood becomes hyperosmotic, the collecting ducts recover more water to dilute the blood; if the blood becomes hyposmotic, the collecting ducts recover less of the water, leading to concentration of the blood. Another way of saying this is: If plasma osmolarity rises, more water is recovered and urine volume decreases; if plasma osmolarity decreases, less water is recovered and urine volume increases. This function is regulated by antidiuretic hormone (vasopressin), a hypothalamic hormone that is stored and released by the posterior pituitary. With mild dehydration, plasma osmolarity rises slightly. This increase is detected by osmoreceptors in the hypothalamus, which stimulates the release of antidiuretic hormone from the posterior pituitary. If plasma osmolarity decreases slightly, the opposite occurs.
When stimulated by antidiuretic hormone, aquaporin channels are inserted into the apical membrane of the cells lining the collecting ducts. As the ducts descend through the medulla, the osmolarity surrounding them increases. If aquaporin water channels are present, water will be osmotically pulled from the collecting duct into the surrounding interstitial space and into the peritubular capillaries. Therefore, the final urine will be more concentrated. If less antidiuretic hormone is secreted, fewer aquaporin channels are inserted and less water is recovered, resulting in dilute urine. By altering the number of aquaporin channels, the volume of water recovered or lost is altered. This, in turn, regulates the blood osmolarity, blood pressure, and osmolarity of the urine.
Aldosterone is a hormone produced by the cortex of the adrenal glands. The control of aldosterone is complex (Figure 11). One mechanism involves detection of a decrease in blood pressure by certain cells in the kidney, resulting in the release of the enzyme renin. This ultimately leads to the production of Angiotensin II, which stimulates the adrenal cortex to produce aldosterone. A second mechanism involves potassium ion concentration. An increased in K+ concentration in extracellular fluid directly stimulates aldosterone secretion by the adrenal cortex.
Aldosterone acts on the cells lining the distal convoluted tubule and the collecting duct to promote, simultaneously, active reabsorption of Na+ from the tubules and active secretion of K+ into the tubules. As Na+ is pumped from the forming urine, water is passively recaptured for the circulation; this preservation of vascular volume is critically important for the maintenance of a normal blood pressure. As an extremely potent vasoconstrictor, angiotensin II functions immediately to increase blood pressure. It also stimulates aldosterone production, which provides a longer-lasting mechanism to support blood pressure by maintaining vascular volume (water recovery).
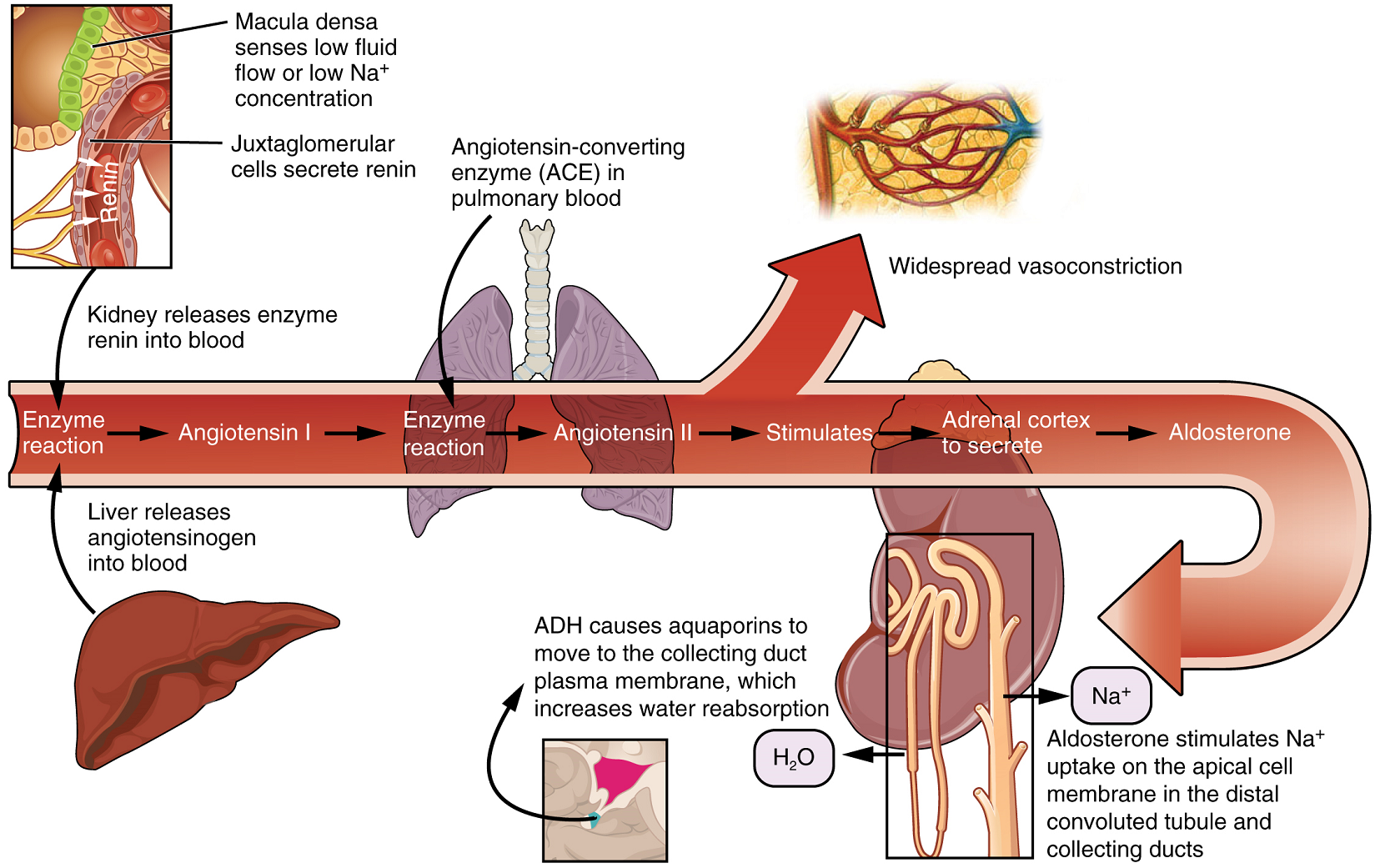
While antidiuretic hormone is primarily involved in the regulation of water recovery, aldosterone regulates Na+ recovery. When aldosterone output increases, more Na+ is recovered from the forming urine and water follows the Na+ passively. As the pump recovers Na+ for the body, it is also pumping K+ into the forming urine, since the pump moves K+ in the opposite direction. When aldosterone decreases, more Na+ remains in the forming urine and more K+ is recovered in the circulation.
Part 4: Micturition Reflex
Micturition is a term for urination or voiding. It results from an interplay of involuntary and voluntary actions by the internal and external urethral sphincters. When bladder volume reaches about 150 mL, an urge to void is sensed but is easily overridden. Voluntary control of urination relies on consciously preventing relaxation of the external urethral sphincter to maintain urinary continence. As the bladder fills, subsequent urges become harder to ignore. Ultimately, voluntary constraint fails with resulting incontinence, which will occur as bladder volume approaches 300 to 400 mL.
Normal micturition is a result of stretch receptors in the bladder wall that transmit nerve impulses to the sacral region of the spinal cord to generate a spinal reflex. The resulting parasympathetic neural outflow causes contraction of the detrusor muscle and relaxation of the involuntary internal urethral sphincter. At the same time, the spinal cord inhibits somatic motor neurons, resulting in the relaxation of the skeletal muscle of the external urethral sphincter. The micturition reflex is active in infants but with maturity, children learn to override the reflex by asserting external sphincter control, thereby delaying voiding.
Voluntary micturition requires an intact spinal cord and functional pudendal nerve arising from the sacral micturition center. Since the external urinary sphincter is voluntary skeletal muscle, it remains contracted (and thereby maintains continence) during filling of the bladder. At the same time, sympathetic nervous activity suppresses contraction of the detrusor muscle. With further bladder stretch, afferent signals traveling over sacral pelvic nerves activate parasympathetic neurons. This activates efferent neurons to release acetylcholine at the neuromuscular junctions, producing detrusor contraction and bladder emptying.
Part 5: Physical Characteristics of Urine
Characteristics of the urine change, depending on influences such as water intake, exercise, environmental temperature, nutrient intake, and other factors (Table 2). Some of the characteristics such as color and odor are rough descriptors of your state of hydration. For example, if you exercise or work outside, and sweat a great deal, your urine will turn darker and produce a slight odor, even if you drink plenty of water.
Urinalysis (urine analysis) often provides clues to renal disease. Normally, only traces of protein are found in urine, and when higher amounts are found, damage to the glomeruli is the likely basis. Unusually large quantities of urine may point to diseases like diabetes mellitus or hypothalamic tumors that cause diabetes insipidus. The color of urine is determined mostly by the breakdown products of red blood cell destruction (Figure 12). The “heme” of hemoglobin is converted by the liver into water-soluble forms that can be excreted indirectly into the urine. Urine color may also be affected by certain foods like beets, berries, and fava beans. A kidney stone or a cancer of the urinary system may produce enough bleeding to manifest as pink or even bright red urine.
| Characteristic | Normal values |
|---|---|
| Colour | Pale yellow to deep amber |
| Odour | Odourless |
| Volume | 750-2000 mL / 24 h |
| pH | 4.5-8.0 |
| Specific gravity | 1.003-1.032 |
| Osmolarity | 40-1350 mOsmol / kg |
| Urobilinogen | 0.2-1.0 mg / 100 mL |
| Leukocytes | 0-2 HPF (per “high-power field” of microscope) |
| Leukocyte esterase | None |
| Protein | None or trace |
| Bilirubin | < 0.3 mg / 100 mL |
| Ketones | None |
| Nitrites | None |
| Erythrocytes | None |
| Glucose | None |
Diseases of the liver or obstructions of bile drainage from the liver impart a dark “tea” or “cola” hue to the urine. Dehydration produces darker, concentrated urine that may also possess the slight odor of ammonia. Most of the ammonia produced from protein breakdown is converted into urea by the liver, so ammonia is rarely detected in fresh urine. Certain foods such as onions, garlic, and fish can impart odor to the urine.
Urine volume varies considerably. The normal range is one to two liters per day (Table 3). The kidneys must produce a minimum urine volume of about 500 mL/day to rid the body of wastes. Output below this level may be caused by severe dehydration or renal disease and is termed oliguria. The virtual absence of urine production is termed anuria.
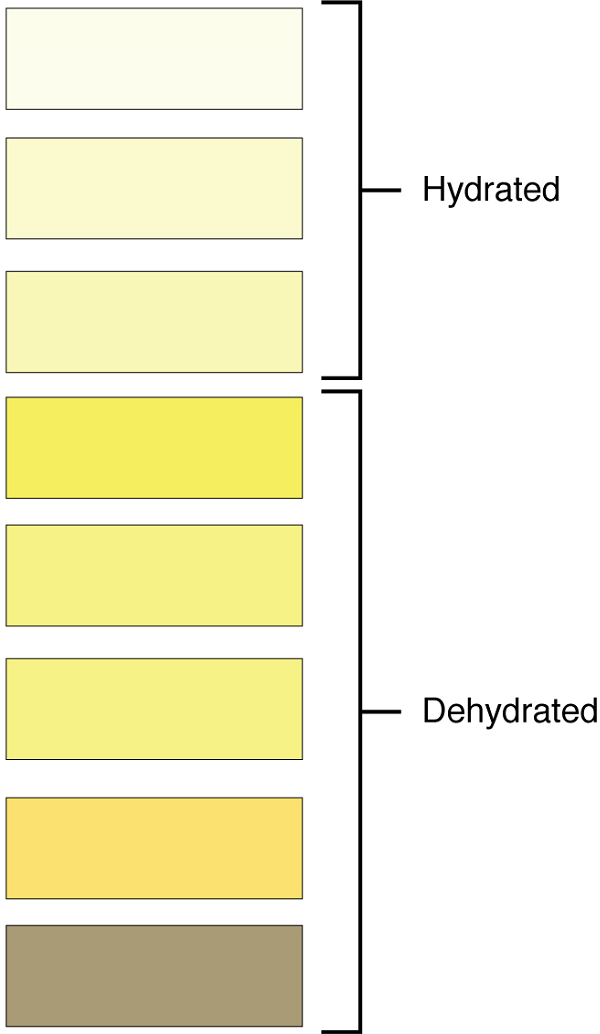
Excessive urine production is polyuria, which may be due to diabetes mellitus or diabetes insipidus. In diabetes mellitus, blood glucose levels are high resulting in the appearance of glucose in the urine. Glucose attracts water osmotically, leading to its loss in the urine. In the case of diabetes insipidus, defficiency of antidiuretic hormone (ADH) leads to high volumes of very dilute urine.
| Volume condition | Volume | Causes |
|---|---|---|
| Normal | 1-2 L/day | |
| Polyuria | >2.5 L/day | Diabetes mellitus; diabetes insipidus; excess caffeine or alcohol; kidney disease; certain drugs, e.g. diuretics; sickle cell anemia, excessive water intake |
| Oliguria | 300-500 mL/day | Dehydration; blood loss; diarrhea; cardiogenic shock; kidney disease; enlarged prostate |
| Anuria | <50 mL/day | Kidney failure; obstruction, e.g. kidney stone or tumor; enlarged prostate |
The pH (hydrogen ion concentration) of the urine can vary more than 1000-fold, from a normal low of 4.5 to a maximum of 8.0. Diet can influence pH; meats lower the pH, whereas citrus fruits, vegetables, and dairy products raise the pH. Chronically high or low pH can lead to disorders, such as the development of kidney stones or osteomalacia (softening of the bones).
Specific gravity is a measure of the quantity of solutes per unit volume of a solution. Urine will always have a specific gravity greater than pure water (water = 1.0) due to the presence of solutes.
Cells are not normally found in the urine. The presence of leukocytes may indicate a urinary tract infection.
Protein does not normally leave the glomerular capillaries, so only trace amounts of protein should be found in the urine, approximately 10 mg/100 mL in a random sample. If excessive protein is detected in the urine, it usually means that the glomerulus is damaged and is allowing protein to “leak” into the filtrate.
Ketones are byproducts of fat metabolism. Finding ketones in the urine suggests that the body is using fat as an energy source in preference to glucose. In diabetes mellitus when there is not enough insulin (type I diabetes mellitus) or because of insulin resistance (type II diabetes mellitus), there is plenty of glucose, but without the action of insulin, the cells cannot take it up, so it remains in the bloodstream. Instead, the cells are forced to use fat as their energy source, and fat consumed at such a level produces excessive ketones as byproducts. These excess ketones will appear in the urine. Ketones may also appear if there is a severe deficiency of proteins or carbohydrates in the diet. Nitrates (NO3–) occur normally in the urine.


