Units and Measurement
The Scope and Scale of Physics
Learning Objectives
By the end of this section, you will be able to:
- Describe the scope of physics.
- Calculate the order of magnitude of a quantity.
- Compare measurable length, mass, and timescales quantitatively.
- Describe the relationships among models, theories, and laws.
Physics is devoted to the understanding of all natural phenomena. In physics, we try to understand physical phenomena at all scales—from the world of subatomic particles to the entire universe. Despite the breadth of the subject, the various subfields of physics share a common core. The same basic training in physics will prepare you to work in any area of physics and the related areas of science and engineering. In this section, we investigate the scope of physics; the scales of length, mass, and time over which the laws of physics have been shown to be applicable; and the process by which science in general, and physics in particular, operates.
The Scope of Physics
Take another look at the chapter-opening image. The Whirlpool Galaxy contains billions of individual stars as well as huge clouds of gas and dust. Its companion galaxy is also visible to the right. This pair of galaxies lies a staggering billion trillion miles [latex]\left(1.4\phantom{\rule{0.2em}{0ex}}×\phantom{\rule{0.2em}{0ex}}{10}^{21}\text{mi}\right)[/latex] from our own galaxy (which is called the Milky Way). The stars and planets that make up the Whirlpool Galaxy might seem to be the furthest thing from most people’s everyday lives, but the Whirlpool is a great starting point to think about the forces that hold the universe together. The forces that cause the Whirlpool Galaxy to act as it does are thought to be the same forces we contend with here on Earth, whether we are planning to send a rocket into space or simply planning to raise the walls for a new home. The gravity that causes the stars of the Whirlpool Galaxy to rotate and revolve is thought to be the same as what causes water to flow over hydroelectric dams here on Earth. When you look up at the stars, realize the forces out there are the same as the ones here on Earth. Through a study of physics, you may gain a greater understanding of the interconnectedness of everything we can see and know in this universe.
Think, now, about all the technological devices you use on a regular basis. Computers, smartphones, global positioning systems (GPSs), MP3 players, and satellite radio might come to mind. Then, think about the most exciting modern technologies you have heard about in the news, such as trains that levitate above tracks, “invisibility cloaks” that bend light around them, and microscopic robots that fight cancer cells in our bodies. All these groundbreaking advances, commonplace or unbelievable, rely on the principles of physics. Aside from playing a significant role in technology, professionals such as engineers, pilots, physicians, physical therapists, electricians, and computer programmers apply physics concepts in their daily work. For example, a pilot must understand how wind forces affect a flight path; a physical therapist must understand how the muscles in the body experience forces as they move and bend. As you will learn in this text, the principles of physics are propelling new, exciting technologies, and these principles are applied in a wide range of careers.
The underlying order of nature makes science in general, and physics in particular, interesting and enjoyable to study. For example, what do a bag of chips and a car battery have in common? Both contain energy that can be converted to other forms. The law of conservation of energy (which says that energy can change form but is never lost) ties together such topics as food calories, batteries, heat, light, and watch springs. Understanding this law makes it easier to learn about the various forms energy takes and how they relate to one another. Apparently unrelated topics are connected through broadly applicable physical laws, permitting an understanding beyond just the memorization of lists of facts.
Science consists of theories and laws that are the general truths of nature, as well as the body of knowledge they encompass. Scientists are continuously trying to expand this body of knowledge and to perfect the expression of the laws that describe it. Physics, which comes from the Greek phúsis, meaning “nature,” is concerned with describing the interactions of energy, matter, space, and time to uncover the fundamental mechanisms that underlie every phenomenon. This concern for describing the basic phenomena in nature essentially defines the scope of physics.
Physics aims to understand the world around us at the most basic level. It emphasizes the use of a small number of quantitative laws to do this, which can be useful to other fields pushing the performance boundaries of existing technologies. Consider a smartphone ((Figure)). Physics describes how electricity interacts with the various circuits inside the device. This knowledge helps engineers select the appropriate materials and circuit layout when building a smartphone. Knowledge of the physics underlying these devices is required to shrink their size or increase their processing speed. Or, think about a GPS. Physics describes the relationship between the speed of an object, the distance over which it travels, and the time it takes to travel that distance. When you use a GPS in a vehicle, it relies on physics equations to determine the travel time from one location to another.
Knowledge of physics is useful in everyday situations as well as in nonscientific professions. It can help you understand how microwave ovens work, why metals should not be put into them, and why they might affect pacemakers. Physics allows you to understand the hazards of radiation and to evaluate these hazards rationally and more easily. Physics also explains the reason why a black car radiator helps remove heat in a car engine, and it explains why a white roof helps keep the inside of a house cool. Similarly, the operation of a car’s ignition system as well as the transmission of electrical signals throughout our body’s nervous system are much easier to understand when you think about them in terms of basic physics.
Physics is a key element of many important disciplines and contributes directly to others. Chemistry, for example—since it deals with the interactions of atoms and molecules—has close ties to atomic and molecular physics. Most branches of engineering are concerned with designing new technologies, processes, or structures within the constraints set by the laws of physics. In architecture, physics is at the heart of structural stability and is involved in the acoustics, heating, lighting, and cooling of buildings. Parts of geology rely heavily on physics, such as radioactive dating of rocks, earthquake analysis, and heat transfer within Earth. Some disciplines, such as biophysics and geophysics, are hybrids of physics and other disciplines.
Physics has many applications in the biological sciences. On the microscopic level, it helps describe the properties of cells and their environments. On the macroscopic level, it explains the heat, work, and power associated with the human body and its various organ systems. Physics is involved in medical diagnostics, such as radiographs, magnetic resonance imaging, and ultrasonic blood flow measurements. Medical therapy sometimes involves physics directly; for example, cancer radiotherapy uses ionizing radiation. Physics also explains sensory phenomena, such as how musical instruments make sound, how the eye detects color, and how lasers transmit information.
It is not necessary to study all applications of physics formally. What is most useful is knowing the basic laws of physics and developing skills in the analytical methods for applying them. The study of physics also can improve your problem-solving skills. Furthermore, physics retains the most basic aspects of science, so it is used by all the sciences, and the study of physics makes other sciences easier to understand.
The Scale of Physics
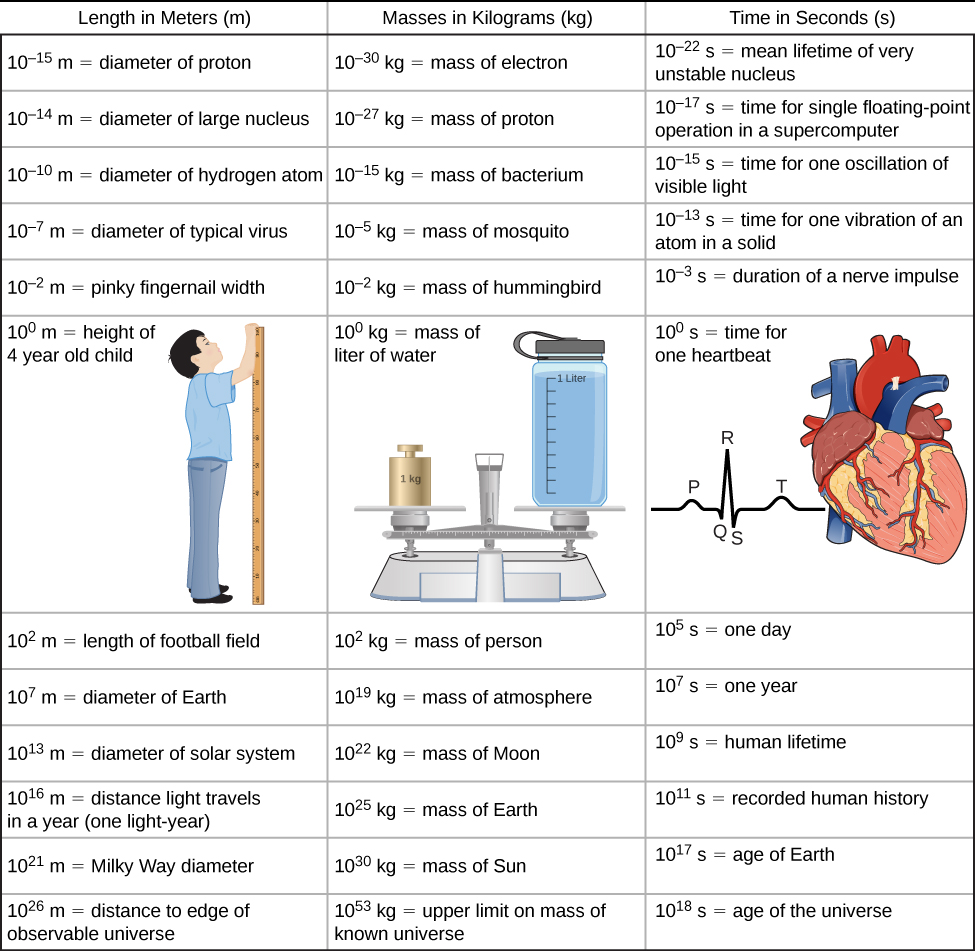
From the discussion so far, it should be clear that to accomplish your goals in any of the various fields within the natural sciences and engineering, a thorough grounding in the laws of physics is necessary. The reason for this is simply that the laws of physics govern everything in the observable universe at all measurable scales of length, mass, and time. Now, that is easy enough to say, but to come to grips with what it really means, we need to get a little bit quantitative. So, before surveying the various scales that physics allows us to explore, let’s first look at the concept of “order of magnitude,” which we use to come to terms with the vast ranges of length, mass, and time that we consider in this text ((Figure)).

Order of magnitude
The order of magnitude of a number is the power of 10 that most closely approximates it. Thus, the order of magnitude refers to the scale (or size) of a value. Each power of 10 represents a different order of magnitude. For example, [latex]{10}^{1},{10}^{2},{10}^{3},[/latex] and so forth, are all different orders of magnitude, as are [latex]{10}^{0}=1,{10}^{-1},{10}^{-2},[/latex] and [latex]{10}^{-3}.[/latex] To find the order of magnitude of a number, take the base-10 logarithm of the number and round it to the nearest integer, then the order of magnitude of the number is simply the resulting power of 10. For example, the order of magnitude of 800 is 103 because [latex]{\text{log}}_{10}800\approx 2.903,[/latex] which rounds to 3. Similarly, the order of magnitude of 450 is 103 because [latex]{\text{log}}_{10}450\approx 2.653,[/latex] which rounds to 3 as well. Thus, we say the numbers 800 and 450 are of the same order of magnitude: 103. However, the order of magnitude of 250 is 102 because [latex]{\text{log}}_{10}250\approx 2.397,[/latex] which rounds to 2.
An equivalent but quicker way to find the order of magnitude of a number is first to write it in scientific notation and then check to see whether the first factor is greater than or less than [latex]\sqrt{10}={10}^{0.5}\approx 3.[/latex] The idea is that [latex]\sqrt{10}={10}^{0.5}[/latex] is halfway between [latex]1={10}^{0}[/latex] and [latex]10={10}^{1}[/latex] on a log base-10 scale. Thus, if the first factor is less than [latex]\sqrt{10},[/latex] then we round it down to 1 and the order of magnitude is simply whatever power of 10 is required to write the number in scientific notation. On the other hand, if the first factor is greater than [latex]\sqrt{10},[/latex] then we round it up to 10 and the order of magnitude is one power of 10 higher than the power needed to write the number in scientific notation. For example, the number 800 can be written in scientific notation as [latex]8\phantom{\rule{0.2em}{0ex}}×\phantom{\rule{0.2em}{0ex}}{10}^{2}.[/latex] Because 8 is bigger than [latex]\sqrt{10}\approx 3,[/latex] we say the order of magnitude of 800 is [latex]{10}^{2+1}={10}^{3}.[/latex] The number 450 can be written as [latex]4.5\phantom{\rule{0.2em}{0ex}}×\phantom{\rule{0.2em}{0ex}}{10}^{2},[/latex] so its order of magnitude is also 103 because 4.5 is greater than 3. However, 250 written in scientific notation is [latex]2.5\phantom{\rule{0.2em}{0ex}}×\phantom{\rule{0.2em}{0ex}}{10}^{2}[/latex] and 2.5 is less than 3, so its order of magnitude is [latex]{10}^{2}.[/latex]
The order of magnitude of a number is designed to be a ballpark estimate for the scale (or size) of its value. It is simply a way of rounding numbers consistently to the nearest power of 10. This makes doing rough mental math with very big and very small numbers easier. For example, the diameter of a hydrogen atom is on the order of 10−10 m, whereas the diameter of the Sun is on the order of 109 m, so it would take roughly [latex]{10}^{9}\text{/}{10}^{-10}={10}^{19}[/latex] hydrogen atoms to stretch across the diameter of the Sun. This is much easier to do in your head than using the more precise values of [latex]1.06\phantom{\rule{0.2em}{0ex}}×\phantom{\rule{0.2em}{0ex}}{10}^{-10}\text{m}[/latex] for a hydrogen atom diameter and [latex]1.39\phantom{\rule{0.2em}{0ex}}×\phantom{\rule{0.2em}{0ex}}{10}^{9}\text{m}[/latex] for the Sun’s diameter, to find that it would take [latex]1.31\phantom{\rule{0.2em}{0ex}}×\phantom{\rule{0.2em}{0ex}}{10}^{19}[/latex] hydrogen atoms to stretch across the Sun’s diameter. In addition to being easier, the rough estimate is also nearly as informative as the precise calculation.
Known ranges of length, mass, and time
The vastness of the universe and the breadth over which physics applies are illustrated by the wide range of examples of known lengths, masses, and times (given as orders of magnitude) in (Figure). Examining this table will give you a feeling for the range of possible topics in physics and numerical values. A good way to appreciate the vastness of the ranges of values in (Figure) is to try to answer some simple comparative questions, such as the following:
- How many hydrogen atoms does it take to stretch across the diameter of the Sun?
(Answer: 109 m/10–10 m = 1019 hydrogen atoms)
- How many protons are there in a bacterium?
(Answer: 10–15 kg/10–27 kg = 1012 protons)
- How many floating-point operations can a supercomputer do in 1 day?
(Answer: 105 s/10–17 s = 1022 floating-point operations)
In studying (Figure), take some time to come up with similar questions that interest you and then try answering them. Doing this can breathe some life into almost any table of numbers.
Visit this site to explore interactively the vast range of length scales in our universe. Scroll down and up the scale to view hundreds of organisms and objects, and click on the individual objects to learn more about each one.
Building Models
How did we come to know the laws governing natural phenomena? What we refer to as the laws of nature are concise descriptions of the universe around us. They are human statements of the underlying laws or rules that all natural processes follow. Such laws are intrinsic to the universe; humans did not create them and cannot change them. We can only discover and understand them. Their discovery is a very human endeavor, with all the elements of mystery, imagination, struggle, triumph, and disappointment inherent in any creative effort ((Figure)). The cornerstone of discovering natural laws is observation; scientists must describe the universe as it is, not as we imagine it to be.

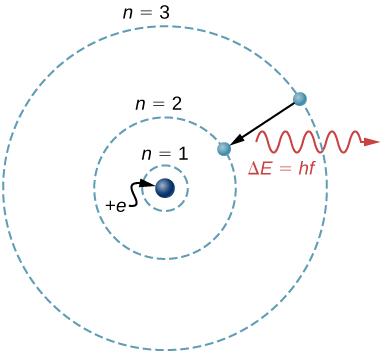
A model is a representation of something that is often too difficult (or impossible) to display directly. Although a model is justified by experimental tests, it is only accurate in describing certain aspects of a physical system. An example is the Bohr model of single-electron atoms, in which the electron is pictured as orbiting the nucleus, analogous to the way planets orbit the Sun ((Figure)). We cannot observe electron orbits directly, but the mental image helps explain some of the observations we can make, such as the emission of light from hot gases (atomic spectra). However, other observations show that the picture in the Bohr model is not really what atoms look like. The model is “wrong,” but is still useful for some purposes. Physicists use models for a variety of purposes. For example, models can help physicists analyze a scenario and perform a calculation or models can be used to represent a situation in the form of a computer simulation. Ultimately, however, the results of these calculations and simulations need to be double-checked by other means—namely, observation and experimentation.
The word theory means something different to scientists than what is often meant when the word is used in everyday conversation. In particular, to a scientist a theory is not the same as a “guess” or an “idea” or even a “hypothesis.” The phrase “it’s just a theory” seems meaningless and silly to scientists because science is founded on the notion of theories. To a scientist, a theory is a testable explanation for patterns in nature supported by scientific evidence and verified multiple times by various groups of researchers. Some theories include models to help visualize phenomena whereas others do not. Newton’s theory of gravity, for example, does not require a model or mental image, because we can observe the objects directly with our own senses. The kinetic theory of gases, on the other hand, is a model in which a gas is viewed as being composed of atoms and molecules. Atoms and molecules are too small to be observed directly with our senses—thus, we picture them mentally to understand what the instruments tell us about the behavior of gases. Although models are meant only to describe certain aspects of a physical system accurately, a theory should describe all aspects of any system that falls within its domain of applicability. In particular, any experimentally testable implication of a theory should be verified. If an experiment ever shows an implication of a theory to be false, then the theory is either thrown out or modified suitably (for example, by limiting its domain of applicability).
A law uses concise language to describe a generalized pattern in nature supported by scientific evidence and repeated experiments. Often, a law can be expressed in the form of a single mathematical equation. Laws and theories are similar in that they are both scientific statements that result from a tested hypothesis and are supported by scientific evidence. However, the designation law is usually reserved for a concise and very general statement that describes phenomena in nature, such as the law that energy is conserved during any process, or Newton’s second law of motion, which relates force (F), mass (m), and acceleration (a) by the simple equation [latex]F=ma.[/latex] A theory, in contrast, is a less concise statement of observed behavior. For example, the theory of evolution and the theory of relativity cannot be expressed concisely enough to be considered laws. The biggest difference between a law and a theory is that a theory is much more complex and dynamic. A law describes a single action whereas a theory explains an entire group of related phenomena. Less broadly applicable statements are usually called principles (such as Pascal’s principle, which is applicable only in fluids), but the distinction between laws and principles often is not made carefully.
The models, theories, and laws we devise sometimes imply the existence of objects or phenomena that are as yet unobserved. These predictions are remarkable triumphs and tributes to the power of science. It is the underlying order in the universe that enables scientists to make such spectacular predictions. However, if experimentation does not verify our predictions, then the theory or law is wrong, no matter how elegant or convenient it is. Laws can never be known with absolute certainty because it is impossible to perform every imaginable experiment to confirm a law for every possible scenario. Physicists operate under the assumption that all scientific laws and theories are valid until a counterexample is observed. If a good-quality, verifiable experiment contradicts a well-established law or theory, then the law or theory must be modified or overthrown completely.
The study of science in general, and physics in particular, is an adventure much like the exploration of an uncharted ocean. Discoveries are made; models, theories, and laws are formulated; and the beauty of the physical universe is made more sublime for the insights gained.
Summary
- Physics is about trying to find the simple laws that describe all natural phenomena.
- Physics operates on a vast range of scales of length, mass, and time. Scientists use the concept of the order of magnitude of a number to track which phenomena occur on which scales. They also use orders of magnitude to compare the various scales.
- Scientists attempt to describe the world by formulating models, theories, and laws.
Conceptual Questions
What is physics?
Physics is the science concerned with describing the interactions of energy, matter, space, and time to uncover the fundamental mechanisms that underlie every phenomenon.
Some have described physics as a “search for simplicity.” Explain why this might be an appropriate description.
If two different theories describe experimental observations equally well, can one be said to be more valid than the other (assuming both use accepted rules of logic)?
No, neither of these two theories is more valid than the other. Experimentation is the ultimate decider. If experimental evidence does not suggest one theory over the other, then both are equally valid. A given physicist might prefer one theory over another on the grounds that one seems more simple, more natural, or more beautiful than the other, but that physicist would quickly acknowledge that he or she cannot say the other theory is invalid. Rather, he or she would be honest about the fact that more experimental evidence is needed to determine which theory is a better description of nature.
What determines the validity of a theory?
Certain criteria must be satisfied if a measurement or observation is to be believed. Will the criteria necessarily be as strict for an expected result as for an unexpected result?
Probably not. As the saying goes, “Extraordinary claims require extraordinary evidence.”
Can the validity of a model be limited or must it be universally valid? How does this compare with the required validity of a theory or a law?
Problems
Find the order of magnitude of the following physical quantities. (a) The mass of Earth’s atmosphere: [latex]5.1\phantom{\rule{0.2em}{0ex}}×\phantom{\rule{0.2em}{0ex}}{10}^{18}\text{kg;}[/latex] (b) The mass of the Moon’s atmosphere: 25,000 kg; (c) The mass of Earth’s hydrosphere: [latex]1.4\phantom{\rule{0.2em}{0ex}}×\phantom{\rule{0.2em}{0ex}}{10}^{21}\text{kg;}[/latex] (d) The mass of Earth: [latex]5.97\phantom{\rule{0.2em}{0ex}}×\phantom{\rule{0.2em}{0ex}}{10}^{24}\text{kg;}[/latex] (e) The mass of the Moon: [latex]7.34\phantom{\rule{0.2em}{0ex}}×\phantom{\rule{0.2em}{0ex}}{10}^{22}\text{kg;}[/latex] (f) The Earth–Moon distance (semimajor axis): [latex]3.84\phantom{\rule{0.2em}{0ex}}×\phantom{\rule{0.2em}{0ex}}{10}^{8}\text{m;}[/latex] (g) The mean Earth–Sun distance: [latex]1.5\phantom{\rule{0.2em}{0ex}}×\phantom{\rule{0.2em}{0ex}}{10}^{11}\text{m;}[/latex] (h) The equatorial radius of Earth: [latex]6.38\phantom{\rule{0.2em}{0ex}}×\phantom{\rule{0.2em}{0ex}}{10}^{6}\text{m;}[/latex] (i) The mass of an electron: [latex]9.11\phantom{\rule{0.2em}{0ex}}×\phantom{\rule{0.2em}{0ex}}{10}^{-31}\text{kg;}[/latex] (j) The mass of a proton: [latex]1.67\phantom{\rule{0.2em}{0ex}}×\phantom{\rule{0.2em}{0ex}}{10}^{-27}\text{kg;}[/latex] (k) The mass of the Sun: [latex]1.99\phantom{\rule{0.2em}{0ex}}×\phantom{\rule{0.2em}{0ex}}{10}^{30}\text{kg.}[/latex]
Use the orders of magnitude you found in the previous problem to answer the following questions to within an order of magnitude. (a) How many electrons would it take to equal the mass of a proton? (b) How many Earths would it take to equal the mass of the Sun? (c) How many Earth–Moon distances would it take to cover the distance from Earth to the Sun? (d) How many Moon atmospheres would it take to equal the mass of Earth’s atmosphere? (e) How many moons would it take to equal the mass of Earth? (f) How many protons would it take to equal the mass of the Sun?
a. 103; b. 105; c. 102; d. 1015; e. 102; f. 1057
For the remaining questions, you need to use (Figure) to obtain the necessary orders of magnitude of lengths, masses, and times.
Roughly how many heartbeats are there in a lifetime?
A generation is about one-third of a lifetime. Approximately how many generations have passed since the year 0 AD?
102 generations
Roughly how many times longer than the mean life of an extremely unstable atomic nucleus is the lifetime of a human?
Calculate the approximate number of atoms in a bacterium. Assume the average mass of an atom in the bacterium is 10 times the mass of a proton.
1011 atoms
(a) Calculate the number of cells in a hummingbird assuming the mass of an average cell is 10 times the mass of a bacterium. (b) Making the same assumption, how many cells are there in a human?
Assuming one nerve impulse must end before another can begin, what is the maximum firing rate of a nerve in impulses per second?
103 nerve impulses/s
About how many floating-point operations can a supercomputer perform each year?
Roughly how many floating-point operations can a supercomputer perform in a human lifetime?
1026 floating-point operations per human lifetime
Glossary
- law
- description, using concise language or a mathematical formula, of a generalized pattern in nature supported by scientific evidence and repeated experiments
- model
- representation of something often too difficult (or impossible) to display directly
- order of magnitude
- the size of a quantity as it relates to a power of 10
- physics
- science concerned with describing the interactions of energy, matter, space, and time; especially interested in what fundamental mechanisms underlie every phenomenon
- theory
- testable explanation for patterns in nature supported by scientific evidence and verified multiple times by various groups of researchers

