Chapter 1. Chemistry: An Experimental Science
1.4 Laboratory Techniques for Separation of Mixtures
Learning Objectives
By the end of this section, you will be able to:
- Describe different methods of separation.
- Identify which separation method is most suited for a given mixture.
- Identify what physical change occurs during the separation process.
A mixture is composed of two or more types of matter that can be present in varying amounts and can be physically separated by using methods that use physical properties to separate the components of the mixture, such as evaporation, distillation, filtration and chromatography.
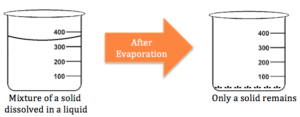
Evaporation can be used as a separation method to separate components of a mixture with a dissolved solid in a liquid. The liquid is evaporated, meaning it is convert from its liquid state to gaseous state. This often requires heat. Once the liquid is completely evaporated, the solid is all that is left behind.
Distillation is a separation technique used to separate components of a liquid mixture by a process of heating and cooling, which exploits the differences in the volatility of each of the components.
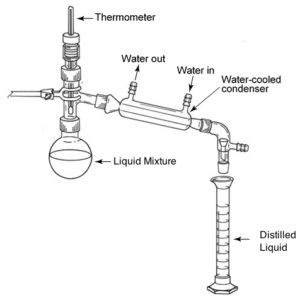
Distillation procedure: 1) the round bottom flask contains the liquid mixture which must be heated to a vigorous boil, 2) the component with the lower boiling point will change into its gaseous state, 3) upon contact with the water-cooled condenser, the gas will condense, 4) trickle down into the graduated cylinder where the chemist can them recuperate the final distilled liquid, and 5) the other liquid component remains in the round bottom flask.
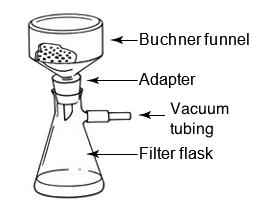
Filtration procedure: 1) the mixture is pored through a funnel lined with a filter paper, 2) the filtrate (liquid) drips through to the filter flask, 3) the solid remains in the funnel.
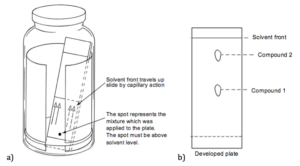
Though chromatography is a simple technique in principle, it remains the most important method for the separation of mixtures into its components. It is quite versatile for it can be used to separate mixtures of solids, or of liquids, or mixtures of solids and liquids combined, or in the case of gas chromatography, can separate mixtures of gases. The two elements of chromatography are the stationary phase and the mobile phase. There are many choices of stationary phases, some being alumina, silica, and even paper. The mobile phase, in liquid chromatography, can also vary. It is often either a solvent or a mixture of solvents and is often referred to as the eluant.. A careful choice of eluting solvent helps to make the separation more successful. The mixture is placed on the stationary phase. The eluant passes over the mixture and continues to pass through the stationary phase carrying along the components of the mixture. If a component in the mixture has greater affinity for the mobile phase (eluant) than the stationary phase, it will tend to be carried along easily with the eluant. If another component in the mixture has a greater affinity for the stationary phase than the mobile phase then it will not be carried along so easily. A separation is thus obtained when the different components in a mixture have different affinity for the stationary and mobile phase. Three important types of chromatography based on the principles discussed above are: 1) thin layer chromatography (TLC), 2) column chromatography, and 3) gas chromatography.
Example 1
Identify which separation method is most suited for the following mixtures:
| Separation methods: | A mixture of solids | A mixture of liquids | A mixture of a solid dissolved in a liquid | A mixture of solid and liquid |
| Evaporation | ||||
| Distillation | ||||
| Filtration | ||||
| Chromatography |
Solution
| Separation methods: | A mixture of solids | A mixture of liquids | A mixture of a solid dissolved in a liquid | A mixture of solid and liquid |
| Evaporation | NO | NO | YES* | YES** |
| Distillation | NO | YES* | YES* | NO |
| Filtration | NO | NO | NO | YES |
| Chromatography | YES* | YES* | YES* | YES* |
* Success depends on the physical properties of the components in the mixture.
** Would work but filtration is so much faster.
Test Yourself
What method of separation would be most effective on the following mixtures:
a) Sea water
b) Gold nuggets in water.
c) A solution of alcohol (liquid) and water.
Answers
a) evaporation or distillation (chromatography not effective here)
b) filtration
c) distillation
Key Concepts and Summary
Mixtures can be physically separated by using methods that use differences in physical properties to separate the components of the mixture, such as evaporation, distillation, filtration and chromatography. Which separation method used when attempting to separate a mixture depends on what kind of mixture it is (what states of matter are present) and on the physical properties of the components.
Exercises
1. What method of separation would be most effective on the following mixtures:
a) Vinegar (a solution of acetic acid (liquid) in water)
b) Loose tea leaves in tea.
c) Copper sulfate (solid) in water.
2. Identify what physical change occurs during the following separation processes.
a) Distillation of a solution comprising of 50:50 acetone and water
b) Filtration to remove tea leaves from tea.
c) Evaporation for water from a sugar solution to obtain sugar crystals.
d) Taking a sand and salt mixing, mixing it with water, followed by filtration to remove the sand, then evaporating the salt solution to retrieve salt crystals.
3. Propose a method of separate the following complex mixtures:
a) A mixture of sand, sea water (water and salt)
b) A mixture of marbles, small gold nuggets, and sugar
Answers
1. a) distillation; b) filtration; c) evaporation
2. a) The lower boiling liquid (acetone) would undergo a phase change (evaporation) upon heating, then once the gaseous acetone comes in contact with the condenser it would under another phase change (condensation). b) No phase changes, this simply involves physical removal of the leaves via filtration. c) Water would undergo a phase change (evaporation) upon heating.
c) The salt would dissolve in the water, then during the evaporation step, water would undergo a phase change (evaporation) upon heating.
3. a) Filtration to remove the sand, then evaporating the salt solution to retrieve salt crystals.
b) Manual separation of the marbles (removing by using your fingers), dissolve the rest in water, then filtration to remove the gold nuggets, and then evaporation of the water to retrieve sugar crystals.
Glossary
chromatography: is a separation technique based on how the different components in a mixture have different affinity for the stationary and mobile phase
distillation: is a separation technique used to separate components of a liquid mixture by a process of heating and cooling
evaporation: is a separation method used to separate of a mixture of a liquid with a dissolved solid, involving removal of a liquid by evaporating it and leaving behind a solid
filtration: is a separation technique used to separate the components of a mixture containing an undissolved solid in a liquid by using a funnel lined with filter paper to retain the solids while letting the liquid through.

