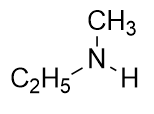Chapter 10. Organic Chemistry
10.5 Nomenclature of Amines
Learning Objectives
- Describe the structure and properties of an amine.
- Distinguish between a primary, secondary and tertiary amine.
- Name and draw structures for primary, secondary and tertiary amines.
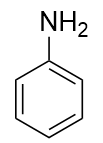
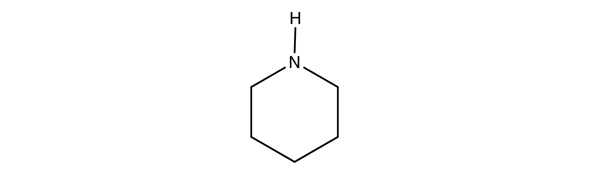
An amine is an organic derivative of ammonia (NH3). In amines, one or more of the H atoms in NH3 is substituted with an organic group.
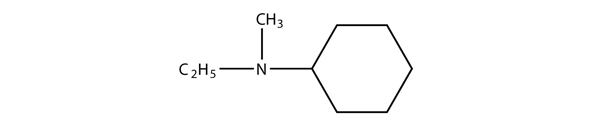
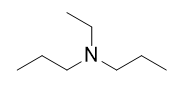
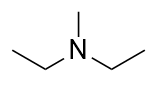
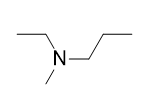
| A primary amine has one H atom substituted with an R group: | A secondary amine has two H atoms substituted with an R group: | A tertiary amine has all three H atoms substituted with R groups: |
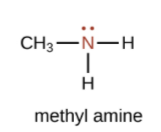 |
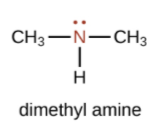 |
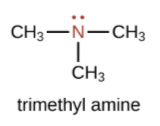 |
Like ammonia, amines are weak bases due to the lone pair of electrons on their nitrogen atoms:
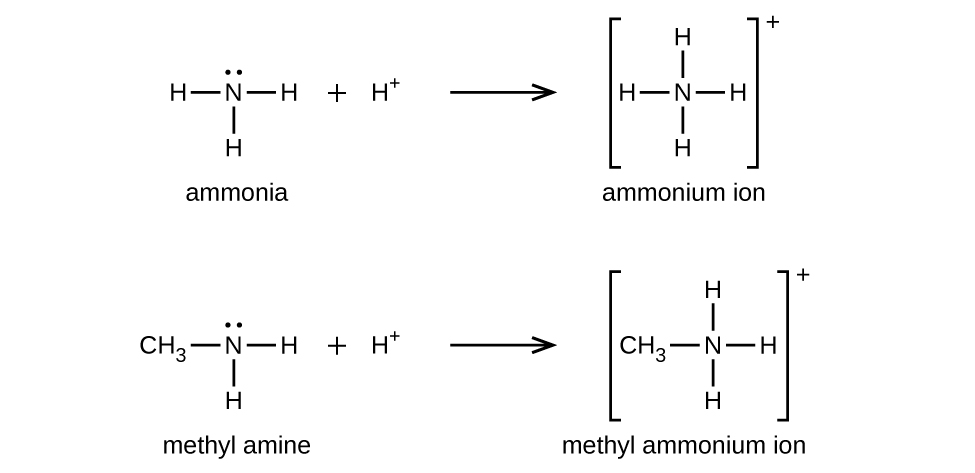
The basicity of an amine’s nitrogen atom plays an important role in much of the compound’s chemistry.
Naming Amines
The alkyl groups connected to the nitrogen atom are named separately and followed by “amine.” If some alkyl groups are the same, then a prefix is used (di or tri), as illustrated here for a few simple examples:
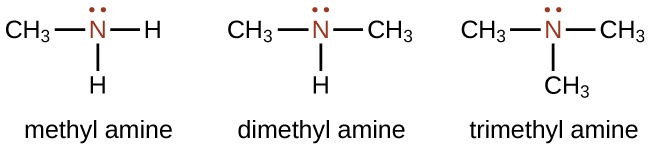
Example 1
Name the following organic compounds:
a) (CH3)2NCH2CH3 b) CH3CH2CH2NHCH3 c) CH3(CH3CH2)NCH2CH2CH3
Solution
a) ethyldimethylamine b) methylpropylamine c) ethylmethylpropylamine
Test Yourself
Give the condensed structure of the following amines:
a) butylamine b) trihexylamine c) methylpentylamine
Answer
a) CH3CH2CH2CH2NH2 b) (CH3CH2CH2CH2CH2CH2)3N c) CH3(CH3CH2CH2CH2CH2)NH
Key Concepts and Summary
Compounds containing a nitrogen atom bonded in a hydrocarbon framework are classified as amines. Amines are a basic functional group. An acid-base reaction occurs when an amine is mixed with and an acid.
Exercises
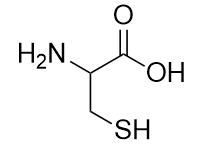
1. What are the structure and name of the smallest amine?
2. Identify each compound as a primary, secondary, or tertiary amine.
Answers
1. CH3NH2; methylamine
2. a) primary b) tertiary c) secondary
3. a) primary b) secondary
4. a) C3H3CO2HSHNH2 + HCl [latex]\longrightarrow[/latex] C3H3CO2HSHNH3Cl
b) (C6H11)(C2H5)(CH3)N + HCl [latex]\longrightarrow[/latex] (C6H11)(C2H5)(CH3)NHCl
c) (C2H5)(CH3)NH + HCl [latex]\longrightarrow[/latex] (C2H5)(CH3)NH2Cl
5. a) ethylmethylamine b) ethyldipropylamine c) diethylmethylamine
Glossary
amine: organic molecule in which a nitrogen atom is bonded to one or more alkyl group