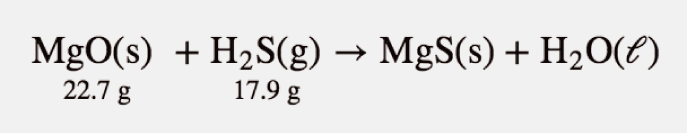Chapter 7. Stoichiometry of Chemical Reactions
7.2 Limiting Reagent and Reaction Yields
Learning Objectives
By the end of this section, you will be able to:
- Explain the concepts of theoretical yield and limiting reactants/reagents.
- Derive the theoretical yield for a reaction under specified conditions.
- Calculate the percent yield for a reaction.
The relative amounts of reactants and products represented in a balanced chemical equation are often referred to as stoichiometric amounts. All the exercises of the preceding module involved stoichiometric amounts of reactants. For example, when calculating the amount of product generated from a given amount of reactant, it was assumed that any other reactants required were available in stoichiometric amounts (or greater). In this module, more realistic situations are considered, in which reactants are not present in stoichiometric amounts.
Limiting Reactant
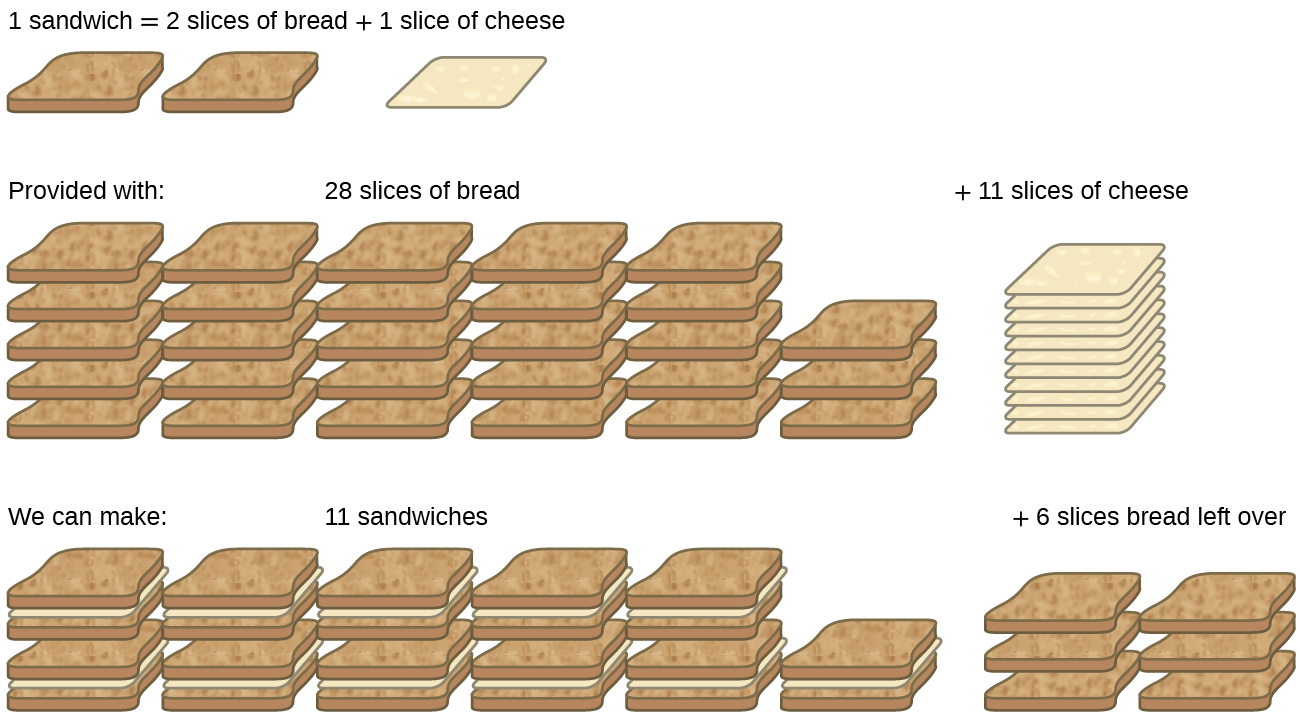
Consider another food analogy, making grilled cheese sandwiches (Figure 1):
Stoichiometric amounts of sandwich ingredients for this recipe are bread and cheese slices in a 2:1 ratio. Provided with 28 slices of bread and 11 slices of cheese, one may prepare 11 sandwiches per the provided recipe, using all the provided cheese and having six slices of bread left over. In this scenario, the number of sandwiches prepared has been limited by the number of cheese slices, and the bread slices have been provided in excess.
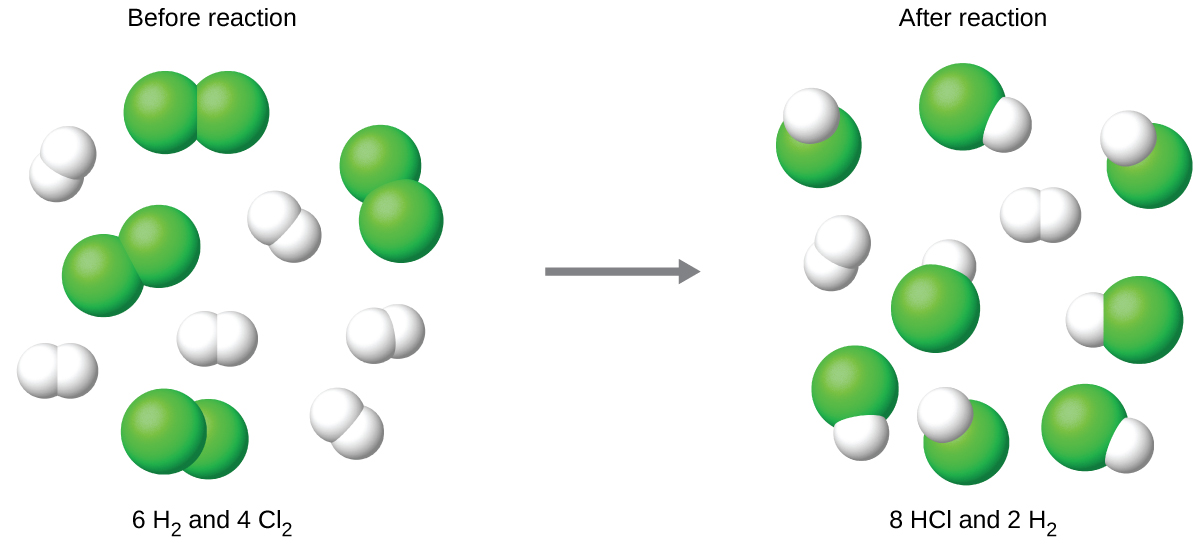
Consider this concept now with regard to a chemical process, the reaction of hydrogen with chlorine to yield hydrogen chloride:
The balanced equation shows the hydrogen and chlorine react in a 1:1 stoichiometric ratio. If these reactants are provided in any other amounts, one of the reactants will nearly always be entirely consumed, thus limiting the amount of product that may be generated. This substance is the limiting reactant, and the other substance is the excess reactant. Identifying the limiting and excess reactants for a given situation requires computing the molar amounts of each reactant provided and comparing them to the stoichiometric amounts represented in the balanced chemical equation. For example, imagine combining 3 moles of H2 and 2 moles of Cl2. This represents a 3:2 (or 1.5:1) ratio of hydrogen to chlorine present for reaction, which is greater than the stoichiometric ratio of 1:1. Hydrogen, therefore, is present in excess, and chlorine is the limiting reactant. Reaction of all the provided chlorine (2 mol) will consume 2 mol of the 3 mol of hydrogen provided, leaving 1 mol of hydrogen unreacted.
An alternative approach to identifying the limiting reactant involves comparing the amount of product expected for the complete reaction of each reactant. Each reactant amount is used to separately calculate the amount of product that would be formed per the reaction’s stoichiometry. The reactant yielding the lesser amount of product is the limiting reactant. For the example in the previous paragraph, complete reaction of the hydrogen would yield
Complete reaction of the provided chlorine would produce
The chlorine will be completely consumed once 4 moles of HCl have been produced. Since enough hydrogen was provided to yield 6 moles of HCl, there will be unreacted hydrogen remaining once this reaction is complete. Chlorine, therefore, is the limiting reactant and hydrogen is the excess reactant (Figure 2).
Example 1
Silicon nitride is a very hard, high-temperature-resistant ceramic used as a component of turbine blades in jet engines. It is prepared according to the following equation:
Which is the limiting reactant when 2.00 g of Si and 1.50 g of N2 react?
Solution
Compute the provided molar amounts of reactants, and then compare these amounts to the balanced equation to identify the limiting reactant.
The provided Si:N2 molar ratio is:
The stoichiometric Si:N2 ratio is:
Comparing these ratios shows that Si is provided in a less-than-stoichiometric amount, and so is the limiting reactant.
Alternatively, compute the amount of product expected for complete reaction of each of the provided reactants. The 0.0712 moles of silicon would yield
[latex]\text{mol Si}_3 \text{N}_4 \;\text{produced} = 0.0\underline{712}11 \;\text{mol Si} \times \frac{1 \;\text{mol Si}_3 \text{N}_4}{3 \;\text{mol Si}} = 0.0237 \;\text{mol Si}_3 \text{N}_4[/latex]
while the 0.0535 moles of nitrogen would produce
Since silicon yields the lesser amount of product, it is the limiting reactant.
Test Yourself
Which is the limiting reactant when 5.00 g of H2 and 10.0 g of O2 react and form water?
Answer
O2
Percent Yield
The amount of product that may be produced by a reaction under specified conditions, as calculated per the stoichiometry of an appropriate balanced chemical equation, is called the theoretical yield of the reaction. In practice, the amount of product obtained is called the actual yield, and it is often less than the theoretical yield for a number of reasons. Some reactions are inherently inefficient, being accompanied by side reactions that generate other products. Others are, by nature, incomplete (consider the partial reactions of weak acids and bases discussed earlier in this chapter). Some products are difficult to collect without some loss, and so less than perfect recovery will reduce the actual yield. The extent to which a reaction’s theoretical yield is achieved is commonly expressed as its percent yield:
Actual and theoretical yields may be expressed as masses or molar amounts (or any other appropriate property; e.g., volume, if the product is a gas). As long as both yields are expressed using the same units, these units will cancel when percent yield is calculated.
Example 2
Upon reaction of 1.274 g of copper sulfate with excess zinc metal, 0.392 g copper metal was obtained according to the equation:
What is the percent yield?
Solution
The provided information identifies copper sulfate as the limiting reactant, and so the theoretical yield is found by the approach illustrated in the previous module, as shown here:
[latex]1.274 \;\rule[0.5ex]{3.75em}{0.1ex}\hspace{-3.75em}\text{g CuSO}_4 \times \frac{1 \;\rule[0.25ex]{4em}{0.1ex}\hspace{-4em}\text{mol CuSO}_4}{159.610 \;\rule[0.25ex]{3em}{0.1ex}\hspace{-3em}\text{g CuSO}_4} \times \frac{1 \;\rule[0.25ex]{2.5em}{0.1ex}\hspace{-2.5em}\text{mol Cu}}{1 \rule[0.25ex]{3.5em}{0.1ex}\hspace{-3.5em}\text{mol CuSO}_4} \times \frac{63.546 \;\text{g Cu}}{1 \;\rule[0.25ex]{2.5em}{0.1ex}\hspace{-2.5em}\text{mol Cu}} = 0.\underline{5072}21 \;\text{g Cu with 4 sig figs}[/latex]
Using this theoretical yield and the provided value for actual yield, the percent yield is calculated to be
[latex]\text{percent yield} = (\frac{0.392 \;\text{g Cu}}{0.\underline{5072}21 \;\text{g Cu}}) \times 100\%[/latex]
[latex]= 77.3\%[/latex]
Test Yourself
What is the percent yield of a reaction that produces 12.5 g of the gas Freon CF2Cl2 from 32.9 g of CCl4 and excess HF?
Green Chemistry and Atom Economy
The purposeful design of chemical products and processes that minimize the use of environmentally hazardous substances and the generation of waste is known as green chemistry. Green chemistry is a philosophical approach that is being applied to many areas of science and technology, and its practice is summarized by guidelines known as the “Twelve Principles of Green Chemistry” (see details at this website). One of the 12 principles is aimed specifically at maximizing the efficiency of processes for synthesizing chemical products. The atom economy of a process is a measure of this efficiency, defined as the percentage by mass of the final product of a synthesis relative to the masses of all the reactants used:
Though the definition of atom economy at first glance appears very similar to that for percent yield, be aware that this property represents a difference in the theoretical efficiencies of different chemical processes. The percent yield of a given chemical process, on the other hand, evaluates the efficiency of a process by comparing the yield of product actually obtained to the maximum yield predicted by stoichiometry.
The synthesis of the common nonprescription pain medication, ibuprofen, nicely illustrates the success of a green chemistry approach (Figure 3). First marketed in the early 1960s, ibuprofen was produced using a six-step synthesis that required 514 g of reactants to generate each mole (206 g) of ibuprofen, an atom economy of 40%.
![This figure is labeled, “a,” and, “b.” Part a shows an open bottle of ibuprofen and a small pile of ibuprofen tablets beside it. Part b shows a reaction along with line structures. The first line structure looks like a diagonal line pointing down and to the right, then up and to the right and then down and to the right. At this point it connects to a hexagon with alternating double bonds. At the first trough there is a line that points straight down. From this structure, there is an arrow pointing downward. The arrow is labeled, “H F,” on the left and “( C H subscript 3 C O ) subscript 2 O,” on the right. The next line structure looks exactly like the first line structure, but it has a line angled down and to the right from the lower right point of the hexagon. This line is connected to another line which points straight down. Where these two lines meet, there is a double bond to an O atom. There is another arrow pointing downward, and it is labeled, “H subscript 2, Raney N i.” The next structure looks very similar to the second, previous structure, except in place of the double bonded O, there is a singly bonded O H group. There is a final reaction arrow pointing downward, and it is labeled, “C O, [ P d ].” The final structure is similar to the third, previous structure except in place of the O H group, there is another line that points down and to the right to an O H group. At these two lines, there is a double bonded O.](https://pressbooks.bccampus.ca/chem1114langaracollege/wp-content/uploads/sites/387/2018/04/CNX_Chem_04_04_GreenChem-2.jpg)
In the 1990s, an alternative process was developed by the BHC Company (now BASF Corporation) that requires only three steps and has an atom economy of ~80%, nearly twice that of the original process. The BHC process generates significantly less chemical waste; uses less-hazardous and recyclable materials; and provides significant cost-savings to the manufacturer (and, subsequently, the consumer). In recognition of the positive environmental impact of the BHC process, the company received the Environmental Protection Agency’s Greener Synthetic Pathways Award in 1997.
More Worked Out Problems
Example 3
A 5.00 g quantity of Rb are combined with 3.44 g of MgCl2 according to this chemical reaction:
2 Rb(s) + MgCl2(s) [latex]\longrightarrow[/latex] Mg(s) + 2 RbCl(s)
What mass of Mg is formed, and what mass of what reactant is left over?
Solution
Because the question asks what mass of magnesium is formed, we can perform two mass-mass calculations and determine which amount is less.
[latex]5.00 \;\rule[0.5ex]{0.5em}{0.1ex}\hspace{-0.5em}\text{g Rb} \times \frac{1 \;\rule[0.25ex]{1.25em}{0.1ex}\hspace{-1.25em}\text{mol Rb}}{85.4678 \;\rule[0.25ex]{0.5em}{0.1ex}\hspace{-0.5em}\text{g Rb}} \times \frac{1 \;\rule[0.25ex]{1.25em}{0.1ex}\hspace{-1.25em}\text{mol Mg}}{2 \rule[0.25ex]{1.25em}{0.1ex}\hspace{-1.25em}\text{mol Rb}} \times \frac{24.3050 \;\text{g Mg}}{1 \;\rule[0.25ex]{1.25em}{0.1ex}\hspace{-1.25em}\text{mol Mg}} = 0.\underline{711}53 \;\text{g Mg with 3 sig figs}[/latex]
[latex]3.44 \;\rule[0.5ex]{0.5em}{0.1ex}\hspace{-0.5em}\text{g MgCl}_2 \times \frac{1 \;\rule[0.25ex]{1.25em}{0.1ex}\hspace{-1.25em}\text{mol MgCl}_2}{95.2104 \;\rule[0.25ex]{0.5em}{0.1ex}\hspace{-0.5em}\text{g MgCl}_2} \times \frac{1 \;\rule[0.25ex]{1.25em}{0.1ex}\hspace{-1.25em}\text{mol Mg}}{1 \rule[0.25ex]{1.25em}{0.1ex}\hspace{-1.25em}\text{mol MgCl}_2} \times \frac{24.3050 \;\text{g Mg}}{1 \;\rule[0.25ex]{1.25em}{0.1ex}\hspace{-1.25em}\text{mol Mg}} = 0.\underline{878}15 \;\text{g Mg with 3 sig figs}[/latex]
The 0.712 g of Mg is the lesser quantity, so the associated reactant—5.00 g of Rb—is the limiting reagent. To determine how much of the other reactant is left, we have to do one more mass-mass calculation to determine what mass of MgCl2 reacted with the 5.00 g of Rb and then subtract the amount reacted from the original amount.
[latex]5.00 \;\rule[0.5ex]{0.5em}{0.1ex}\hspace{-0.5em}\text{g Rb} \times \frac{1 \;\rule[0.25ex]{1.25em}{0.1ex}\hspace{-1.25em}\text{mol Rb}}{85.4678 \;\rule[0.25ex]{0.5em}{0.1ex}\hspace{-0.5em}\text{g Rb}} \times \frac{1 \;\rule[0.25ex]{1.25em}{0.1ex}\hspace{-1.25em}\text{mol MgCl}_2}{2 \rule[0.25ex]{1.25em}{0.1ex}\hspace{-1.25em}\text{mol Rb}} \times \frac{95.2104 \;\text{g MgCl}_2}{1 \;\rule[0.25ex]{1.25em}{0.1ex}\hspace{-1.25em}\text{mol MgCl}_2} = \underline{2.78}498 \;\text{g magnesium chloride with 3 sig figs}[/latex]
Because we started with 3.44 g of MgCl2, we have
3.44 g MgCl2 − 2.78498 g MgCl2 reacted = 0.66 g MgCl2 left
Test Yourself
Given the initial amounts listed, what is the limiting reagent, and what is the mass of the leftover reagent?
Answer
H2S is the limiting reagent; 1.5 g of MgO are left over.
Example 4
A worker reacts 30.5 g of Zn with nitric acid and evapourates the remaining water to obtain 65.2 g of Zn(NO3)2. What are the theoretical yield, the actual yield, and the percent yield?
Zn(s) + 2 HNO3(aq) [latex]\longrightarrow[/latex] Zn(NO3)2(aq) + H2(g)
Solution
A mass-mass calculation can be performed to determine the theoretical yield. We need the molar masses of Zn (65.37 g/mol) and Zn(NO3)2 (189.38 g/mol). In three steps, the mass-mass calculation is
[latex]30.5 \;\rule[0.5ex]{0.5em}{0.1ex}\hspace{-0.5em}\text{g Zn} \times \frac{1 \;\rule[0.25ex]{1.25em}{0.1ex}\hspace{-1.25em}\text{mol Zn}}{65.37 \;\rule[0.25ex]{0.5em}{0.1ex}\hspace{-0.5em}\text{g Zn}} \times \frac{1 \;\rule[0.25ex]{1.25em}{0.1ex}\hspace{-1.25em}\text{mol Zn(NO}_3 \text{)}_2}{1 \rule[0.25ex]{1.25em}{0.1ex}\hspace{-1.25em}\text{mol Zn}} \times \frac{189.38 \;\text{g Zn(NO}_3 \text{)}_2}{1 \;\rule[0.25ex]{1.25em}{0.1ex}\hspace{-1.25em}\text{mol Zn(NO}_3 \text{)}_2} = \underline{88.3}6 \;\text{g zing nitrate with 3 sig figs}[/latex]
Thus, the theoretical yield is 88.4 g of Zn(NO3)2. The actual yield is the amount that was actually made, which was 65.2 g of Zn(NO3)2. To calculate the percent yield, we take the actual yield and divide it by the theoretical yield and multiply by 100:
[latex]\text{percent yield} = (\frac{65.2 \;\text{g Zn(NO}_3 \text{)}_2}{\underline{88.3}6 \;\text{g Zn(NO}_3 \text{)}_2}) \times 100\%[/latex]
[latex]= 73.8\%[/latex]
The worker achieved almost three-fourths of the possible yield.
Test Yourself
A synthesis produced 2.05 g of NH3 from 16.5 g of N2. What is the theoretical yield and the percent yield?
N2(g) + 3 H2(g) [latex]\longrightarrow[/latex] 2 NH3(g)
Answer
theoretical yield = 20.1 g; percent yield = 10.2%
Chemistry Is Everywhere: Actual Yields in Drug Synthesis and Purification
Many drugs are the product of several steps of chemical synthesis. Each step typically occurs with less than 100% yield, so the overall percent yield might be very small. The general rule is that the overall percent yield is the product of the percent yields of the individual synthesis steps. For a drug synthesis that has many steps, the overall percent yield can be very tiny, which is one factor in the huge cost of some drugs. For example, if a 10-step synthesis has a percent yield of 90% for each step, the overall yield for the entire synthesis is only 35%. Many scientists work every day trying to improve percent yields of the steps in the synthesis to decrease costs, improve profits, and minimize waste.
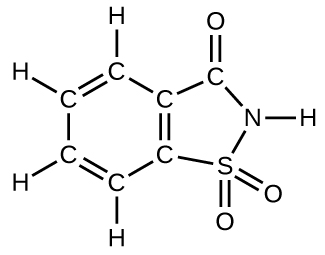
Even purifications of complex molecules into drug-quality purity are subject to percent yields. Consider the purification of impure albuterol. Albuterol (C13H21NO2; accompanying figure) is an inhaled drug used to treat asthma, bronchitis, and other obstructive pulmonary diseases. It is synthesized from norepinephrine, a naturally occurring hormone and neurotransmitter. Its initial synthesis makes very impure albuterol that is purified in five chemical steps. The details of the steps do not concern us; only the percent yields do:
| impure albuterol → intermediate A | percent yield = 70% |
| intermediate A → intermediate B | percent yield = 100% |
| intermediate B → intermediate C | percent yield = 40% |
| intermediate C → intermediate D | percent yield = 72% |
| intermediate D → purified albuterol | percent yield = 35% |
| overall percent yield = 70% × 100% × 40% × 72% × 35% = 7.5% | |
That is, only about one-fourteenth of the original material was turned into the purified drug. This gives you one reason why some drugs are so expensive; a lot of material is lost in making a high-purity pharmaceutical.
Key Concepts and Summary
When reactions are carried out using less-than-stoichiometric quantities of reactants, the amount of product generated will be determined by the limiting reactant. The amount of product generated by a chemical reaction is its actual yield. This yield is often less than the amount of product predicted by the stoichiometry of the balanced chemical equation representing the reaction (its theoretical yield). The extent to which a reaction generates the theoretical amount of product is expressed as its percent yield.
Key Equations
- [latex]\text{percent yield} = (\frac{\text{actual yield}}{\text{theoretical yield}}) \times 100\%[/latex]
Exercises
1. What is the limiting reactant in a reaction that produces sodium chloride from 8 g of sodium and 8 g of diatomic chlorine?
2. A student isolated 25 g of a compound following a procedure that would theoretically yield 81 g. What was his percent yield?
3. Freon-12, CCl2F2, is prepared from CCl4 by reaction with HF. The other product of this reaction is HCl. Outline the steps needed to determine the percent yield of a reaction that produces 12.5 g of CCl2F2 from 32.9 g of CCl4. Freon-12 has been banned and is no longer used as a refrigerant because it catalyzes the decomposition of ozone and has a very long lifetime in the atmosphere. Determine the percent yield.
4. Toluene, C6H5CH3, is oxidized by air under carefully controlled conditions to benzoic acid, C6H5CO2H, which is used to prepare the food preservative sodium benzoate, C6H5CO2Na. What is the percent yield of a reaction that converts 1.000 kg of toluene to 1.21 kg of benzoic acid?
[latex]2\text{C}_6\text{H}_5\text{CH}_3\;+\;3\text{O}_2\;{\longrightarrow}\;2\text{C}_6\text{H}_5\text{CO}_2\text{H}\;+\;2\text{H}_2\text{O}[/latex]
5. Outline the steps needed to solve the following problem, then do the calculations. Ether, (C2H5)2O, which was originally used as an anesthetic but has been replaced by safer and more effective medications, is prepared by the reaction of ethanol with sulfuric acid.
[latex]2\text{C}_2\text{H}_5\text{OH}\;+\;\text{H}_2\text{SO}_4\;{\longrightarrow}\;(\text{C}_2\text{H}_5)_2\;+\;\text{H}_2\text{SO}_4{\cdot}\text{H}_2\text{O}[/latex]
What is the percent yield of ether if 1.17 L (d = 0.7134 g/mL) is isolated from the reaction of 1.500 L of C2H5OH (d = 0.7894 g/mL)?
6. Outline the steps needed to determine the limiting reactant when 0.50 mol of Cr and 0.75 mol of H3PO4 react according to the following chemical equation.
[latex]2\text{Cr}\;+\;2\text{H}_3\text{PO}_4\;{\longrightarrow}\;2\text{CrPO}_4\;+\;3\text{H}_2[/latex]
Determine the limiting reactant.
7. Uranium can be isolated from its ores by dissolving it as UO2(NO3)2, then separating it as solid UO2(C2O4)·3H2O. Addition of 0.4031 g of sodium oxalate, Na2C2O4, to a solution containing 1.481 g of uranyl nitrate, UO2(NO3)2, yields 1.073 g of solid UO2(C2O4)·3H2O.
[latex]\text{Na}_2\text{C}_2\text{O}_4\;+\;\text{UO}_2(\text{NO}_3)_2\;+\;3\text{H}_2\text{O}\;{\longrightarrow}\;\text{UO}_2(\text{C}_2\text{O}_4){\cdot}3\text{H}_2\text{O}\;+\;2\text{NaNO}_3[/latex]
Determine the limiting reactant and the percent yield of this reaction.
8. How many molecules of the sweetener saccharin can be prepared from 30 C atoms, 25 H atoms, 12 O atoms, 8 S atoms, and 14 N atoms?
9. Would you agree to buy 1 trillion (1,000,000,000,000) gold atoms for $5? Explain why or why not. Find the current price of gold at http://money.cnn.com/data/commodities/ (1 troy ounce = 31.1 g)
10. The box below shows a group of nitrogen and hydrogen molecules that will react to produce ammonia, NH3. What is the limiting reagent?
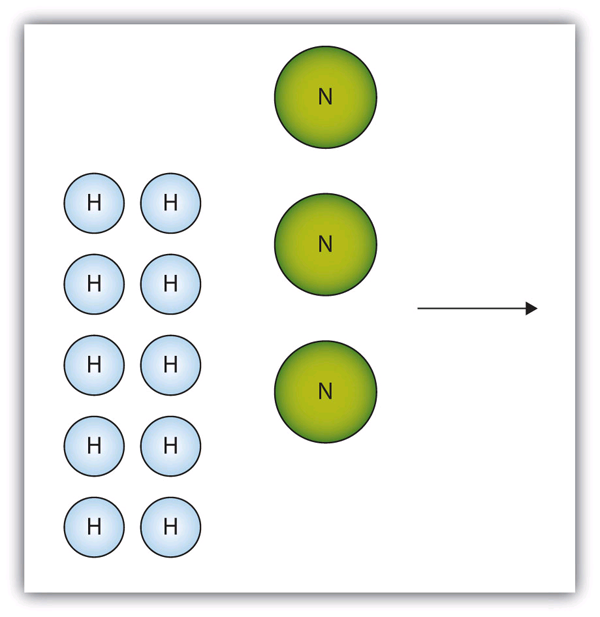
2 C(s) + H2(g) [latex]\longrightarrow[/latex] C2H2(g)
What is the limiting reagent? How much of the other reactant is in excess?
SiO2(s) + 4 HF(g) [latex]\longrightarrow[/latex] SiF4(g) + 2 H2O(ℓ)
17. A chemist decomposes 1.006 g of NaHCO3 and obtains 0.0334 g of Na2CO3. What are the theoretical yield and the actual yield?
2 NaHCO3(s) [latex]\longrightarrow[/latex] Na2CO3(s) + H2O(ℓ) + CO2(g)
18. What is the percent yield in Exercise 16?
19. What is the percent yield in Exercise 17?
Answers
1. The limiting reactant is Cl2.
2. Percent yield = 31%
3. g CCl4 [latex]\longrightarrow[/latex] mol CCl4 [latex]\longrightarrow[/latex] mol CCl2F2 [latex]\longrightarrow[/latex] g CCl2F2, percent yield = 48.3%
4. percent yield = 91.3%
5. Convert mass of ethanol to moles of ethanol; relate the moles of ethanol to the moles of ether produced using the stoichiometry of the balanced equation. Convert moles of ether to grams; divide the actual grams of ether (determined through the density) by the theoretical mass to determine the percent yield; 87.6%
6. The conversion needed is [latex]\text{mol\;Cr}\;{\longrightarrow}\;\text{mol\;H}_3\text{PO}_4[/latex]. Then compare the amount of Cr to the amount of acid present. Cr is the limiting reactant.
7. Na2C2O4 is the limiting reactant. percent yield = 86.6%
8. Only four molecules can be made.
9. This amount cannot be weighted by ordinary balances and is worthless.
10. Nitrogen is the limiting reagent.
11. Yes; methane is the limiting reagent.
12. C is the limiting reagent; 4.33 g of H2 are left over.
13. H2O is the limiting reagent; 25.9 g of P4O6 are left over.
14. 6.06 g of PbCl2 are formed; 0.33 g of NaCl is left over.
15. Theoretical yield is what you expect stoichiometrically from a chemical reaction; actual yield is what you actually get from a chemical reaction.
16. theoretical yield = 4.052 g; actual yield = 2.675 g
17. theoretical yield = 0.635 g; actual yield = 0.0334 g
18. 66.02%
19. 5.26%
Glossary
actual yield: amount of product formed in a reaction
excess reactant: reactant present in an amount greater than required by the reaction stoichiometry
limiting reactant: reactant present in an amount lower than required by the reaction stoichiometry, thus limiting the amount of product generated
percent yield: measure of the efficiency of a reaction, expressed as a percentage of the theoretical yield
theoretical yield: amount of product that may be produced from a given amount of reactant(s) according to the reaction stoichiometry