Chapter 6. Chemical Reactions and Equations
6.1 Writing and Balancing Chemical Equations
Learning Objectives
By the end of this section, you will be able to:
- Derive chemical equations from narrative descriptions of chemical reactions.
- Write and balance chemical equations in molecular, total ionic, and net ionic formats.
The preceding chapter introduced the use of element symbols to represent individual atoms. When atoms gain or lose electrons to yield ions, or combine with other atoms to form molecules, their symbols are modified or combined to generate chemical formulas that appropriately represent these species. Extending this symbolism to represent both the identities and the relative quantities of substances undergoing a chemical (or physical) change involves writing and balancing a chemical equation. Consider as an example the reaction between one methane molecule (CH4) and two diatomic oxygen molecules (O2) to produce one carbon dioxide molecule (CO2) and two water molecules (H2O). The chemical equation representing this process is provided in the upper half of Figure 1, with space-filling molecular models shown in the lower half of the figure.
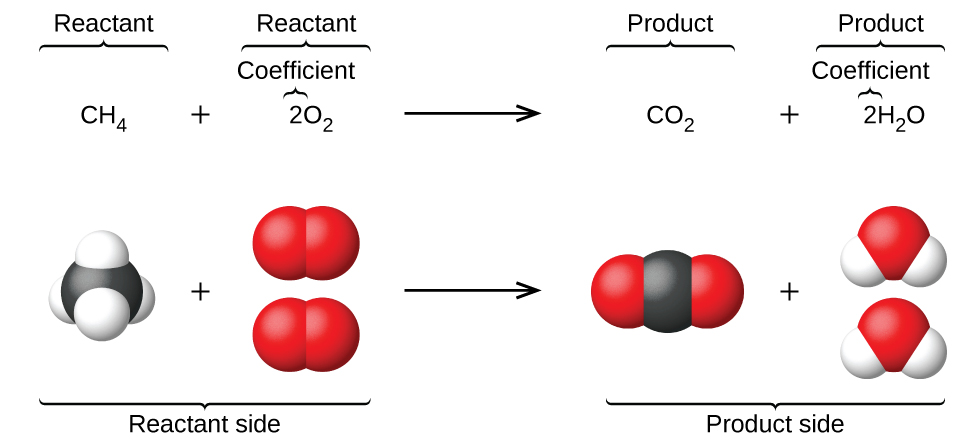
This example illustrates the fundamental aspects of any chemical equation:
- The substances undergoing reaction are called reactants, and their formulas are placed on the left side of the equation.
- The substances generated by the reaction are called products, and their formulas are placed on the right sight of the equation.
- Plus signs (+) separate individual reactant and product formulas, and an arrow ([latex]\longrightarrow[/latex]) separates the reactant and product (left and right) sides of the equation.
- The relative numbers of reactant and product species are represented by coefficients (numbers placed immediately to the left of each formula). A coefficient of 1 is typically omitted.
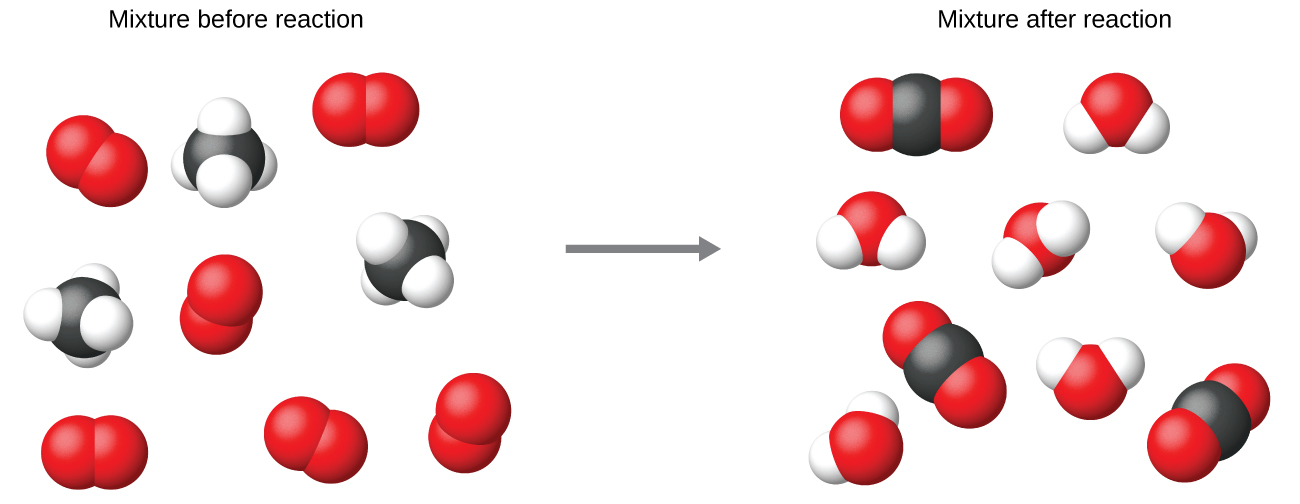
It is common practice to use the smallest possible whole-number coefficients in a chemical equation, as is done in this example. Realize, however, that these coefficients represent the relative numbers of reactants and products, and, therefore, they may be correctly interpreted as ratios. Methane and oxygen react to yield carbon dioxide and water in a 1:2:1:2 ratio. This ratio is satisfied if the numbers of these molecules are, respectively, 1-2-1-2, or 2-4-2-4, or 3-6-3-6, and so on (Figure 2). Likewise, these coefficients may be interpreted with regard to any amount (number) unit, and so this equation may be correctly read in many ways, including:
- One methane molecule and two oxygen molecules react to yield one carbon dioxide molecule and two water molecules.
- One dozen methane molecules and two dozen oxygen molecules react to yield one dozen carbon dioxide molecules and two dozen water molecules.
- One mole of methane molecules and 2 moles of oxygen molecules react to yield 1 mole of carbon dioxide molecules and 2 moles of water molecules.
Balancing Equations
The chemical equation described in Figure 1 is balanced, meaning that equal numbers of atoms for each element involved in the reaction are represented on the reactant and product sides. This is a requirement the equation must satisfy to be consistent with the law of conservation of matter. It may be confirmed by simply summing the numbers of atoms on either side of the arrow and comparing these sums to ensure they are equal. Note that the number of atoms for a given element is calculated by multiplying the coefficient of any formula containing that element by the element’s subscript in the formula. If an element appears in more than one formula on a given side of the equation, the number of atoms represented in each must be computed and then added together. For example, both product species in the example reaction, CO2 and H2O, contain the element oxygen, and so the number of oxygen atoms on the product side of the equation is
The equation for the reaction between methane and oxygen to yield carbon dioxide and water is confirmed to be balanced per this approach, as shown here:
| Element | Reactants | Products | Balanced? |
|---|---|---|---|
| C | 1 × 1 = 1 | 1 × 1 = 1 | 1 = 1, yes |
| H | 4 × 1 = 4 | 2 × 2 = 4 | 4 = 4, yes |
| O | 2 × 2 = 4 | (1 × 2) + (2 × 1) = 4 | 4 = 4, yes |
| Table 1. | |||
A balanced chemical equation often may be derived from a qualitative description of some chemical reaction by a fairly simple approach known as balancing by inspection. Consider as an example the decomposition of water to yield molecular hydrogen and oxygen. This process is represented qualitatively by an unbalanced chemical equation:
Comparing the number of H and O atoms on either side of this equation confirms its imbalance:
| Element | Reactants | Products | Balanced? |
|---|---|---|---|
| H | 1 × 2 = 2 | 1 × 2 = 2 | 2 = 2, yes |
| O | 1 × 1 = 1 | 1 × 2 = 2 | 1 ≠ 2, no |
| Table 2. | |||
The numbers of H atoms on the reactant and product sides of the equation are equal, but the numbers of O atoms are not. To achieve balance, the coefficients of the equation may be changed as needed. Keep in mind, of course, that the formula subscripts define, in part, the identity of the substance, and so these cannot be changed without altering the qualitative meaning of the equation. For example, changing the reactant formula from H2O to H2O2 would yield balance in the number of atoms, but doing so also changes the reactant’s identity (it’s now hydrogen peroxide and not water). The O atom balance may be achieved by changing the coefficient for H2O to 2.
| Element | Reactants | Products | Balanced? |
|---|---|---|---|
| H | 2 × 2 = 4 | 1 × 2 = 2 | 4 ≠ 2, no |
| O | 2 × 1 = 2 | 1 × 2 = 2 | 2 = 2, yes |
| Table 3. | |||
The H atom balance was upset by this change, but it is easily reestablished by changing the coefficient for the H2 product to 2.
| Element | Reactants | Products | Balanced? |
|---|---|---|---|
| H | 2 × 2 = 4 | 2 × 2 = 4 | 4 = 4, yes |
| O | 2 × 1 = 2 | 1 × 2 = 2 | 2 = 2, yes |
| Table 4. | |||
These coefficients yield equal numbers of both H and O atoms on the reactant and product sides, and the balanced equation is, therefore:
Example 1
Write a balanced equation for the reaction of molecular nitrogen (N2) and oxygen (O2) to form dinitrogen pentoxide.
Solution
First, write the unbalanced equation.
Next, count the number of each type of atom present in the unbalanced equation.
| Element | Reactants | Products | Balanced? |
|---|---|---|---|
| N | 1 × 2 = 2 | 1 × 2 = 2 | 2 = 2, yes |
| O | 1 × 2 = 2 | 1 × 5 = 5 | 2 ≠ 5, no |
| Table 5. | |||
Though nitrogen is balanced, changes in coefficients are needed to balance the number of oxygen atoms. To balance the number of oxygen atoms, a reasonable first attempt would be to change the coefficients for the O2 and N2O5 to integers that will yield 10 O atoms (the least common multiple for the O atom subscripts in these two formulas).
| Element | Reactants | Products | Balanced? |
|---|---|---|---|
| N | 1 ×× 2 = 2 | 2 × 2 = 4 | 2 ≠ 4, no |
| O | 5 × 2 = 10 | 2 × 5 = 10 | 10 = 10, yes |
| Table 6. | |||
The N atom balance has been upset by this change; it is restored by changing the coefficient for the reactant N2 to 2.
| Element | Reactants | Products | Balanced? |
|---|---|---|---|
| N | 2 × 2 = 4 | 2 × 2 = 4 | 4 = 4, yes |
| O | 5 × 2 = 10 | 2 × 5 = 10 | 10 = 10, yes |
| Table 7. | |||
The numbers of N and O atoms on either side of the equation are now equal, and so the equation is balanced.
Test Yourself
Write a balanced equation for the decomposition of ammonium nitrate to form molecular nitrogen, molecular oxygen, and water. (Hint: Balance oxygen last, since it is present in more than one molecule on the right side of the equation.)
Answer
[latex]2\text{NH}_4 \text{NO}_3 \longrightarrow 2\text{N}_2 + \text{O}_2 + 4\text{H}_2\text{O}[/latex]
It is sometimes convenient to use fractions instead of integers as intermediate coefficients in the process of balancing a chemical equation. When balance is achieved, all the equation’s coefficients may then be multiplied by a whole number to convert the fractional coefficients to integers without upsetting the atom balance. For example, consider the reaction of ethane (C2H6) with oxygen to yield H2O and CO2, represented by the unbalanced equation:
Following the usual inspection approach, one might first balance C and H atoms by changing the coefficients for the two product species, as shown:
This results in seven O atoms on the product side of the equation, an odd number—no integer coefficient can be used with the O2 reactant to yield an odd number, so a fractional coefficient, [latex]\frac{7}{2}[/latex], is used instead to yield a provisional balanced equation:
A conventional balanced equation with integer-only coefficients is derived by multiplying each coefficient by 2:
Finally with regard to balanced equations, recall that convention dictates use of the smallest whole-number coefficients. Although the equation for the reaction between molecular nitrogen and molecular hydrogen to produce ammonia is, indeed, balanced,
the coefficients are not the smallest possible integers representing the relative numbers of reactant and product molecules. Dividing each coefficient by the greatest common factor, 3, gives the preferred equation:
Additional Information in Chemical Equations
The physical states of reactants and products in chemical equations very often are indicated with a parenthetical abbreviation following the formulas. Common abbreviations include s for solids, l for liquids, g for gases, and aq for substances dissolved in water (aqueous solutions, as introduced in the preceding chapter). These notations are illustrated in the example equation here:
This equation represents the reaction that takes place when sodium metal is placed in water. The solid sodium reacts with liquid water to produce molecular hydrogen gas and the ionic compound sodium hydroxide (a solid in pure form, but readily dissolved in water).
Special conditions necessary for a reaction are sometimes designated by writing a word or symbol above or below the equation’s arrow. For example, a reaction carried out by heating may be indicated by the uppercase Greek letter delta (Δ) over the arrow.
Other examples of these special conditions will be encountered in more depth in later chapters.
Ionic Compounds in Solution
One important aspect about ionic compounds that differs from molecular compounds has to do with dissolving in a liquid, such as water. When molecular compounds, such as sugar, dissolve in water, the individual molecules drift apart from each other. When ionic compounds dissolve, the ions physically separate from each other. We can use a chemical equation to represent this process—for example, with NaCl:
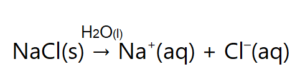
Figure 3. The dissolution of sodium chloride.
When NaCl dissolves in water, the ions separate and go their own way in solution; the ions are now written with their respective charges, and the (aq) phase label emphasizes that they are dissolved (Figure 3 "Ionic Solutions"). This process is called dissociation; we say that the ions dissociate.
When an ionic compound dissociates in water, water molecules surround each ion and separate it from the rest of the solid. Each ion goes its own way in solution.
All ionic compounds that dissolve behave this way. (This behaviour was first suggested by the Swedish chemist Svante August Arrhenius [1859–1927] as part of his PhD dissertation in 1884. Interestingly, his PhD examination team had a hard time believing that ionic compounds would behave like this, so they gave Arrhenius a barely passing grade. Later, this work was cited when Arrhenius was awarded the Nobel Prize in Chemistry.)
Keep in mind that when the ions separate, all of the ions separate. Thus, when CaCl2 dissolves, the one Ca2+ ion and the two Cl− ions separate from each other:
CaCl2(s) [latex]\longrightarrow[/latex] Ca2+(aq) + Cl-(aq) + Cl-(aq)
or
CaCl2(s) [latex]\longrightarrow[/latex] Ca2+(aq) + 2Cl-(aq)
That is, the two chloride ions go off on their own. They do not remain as Cl2 (that would be elemental chlorine; these are chloride ions); they do not stick together to make Cl2− or Cl22−. They become dissociated ions in their own right. Polyatomic ions also retain their overall identity when they are dissolved.
Example 2
Write the chemical equation that represents the dissociation of each ionic compound.
a) KBr b) Na2SO4
Solution
a) KBr(s) [latex]\longrightarrow[/latex] K+(aq) + Br−(aq)
b) Not only do the two sodium ions go their own way, but the sulfate ion stays together as the sulfate ion. The dissolving equation is
Na2SO4(s) [latex]\longrightarrow[/latex] 2Na+(aq) + SO42−(aq)
Test Yourself
Write the chemical equation that represents the dissociation of (NH4)2S.
Answer
(NH4)2S(s) [latex]\longrightarrow[/latex] 2NH4+(aq) + S2−(aq)
Equations for Ionic Reactions
Given the abundance of water on earth, it stands to reason that a great many chemical reactions take place in aqueous media. When ions are involved in these reactions, the chemical equations may be written with various levels of detail appropriate to their intended use. To illustrate this, consider a reaction between ionic compounds taking place in an aqueous solution. When aqueous solutions of CaCl2 and AgNO3 are mixed, a reaction takes place producing aqueous Ca(NO3)2 and solid AgCl:
This balanced equation, derived in the usual fashion, is called a molecular equation because it doesn’t explicitly represent the ionic species that are present in solution. When ionic compounds dissolve in water, they may dissociate into their constituent ions, which are subsequently dispersed homogenously throughout the resulting solution (a thorough discussion of this important process is provided in the chapter on solutions). Ionic compounds dissolved in water are, therefore, more realistically represented as dissociated ions, in this case:
Unlike these three ionic compounds, AgCl does not dissolve in water to a significant extent, as signified by its physical state notation, s.
Explicitly representing all dissolved ions results in a complete ionic equation. In this particular case, the formulas for the dissolved ionic compounds are replaced by formulas for their dissociated ions:
Examining this equation shows that two chemical species are present in identical form on both sides of the arrow, Ca2+(aq) and NO3−(aq).NO3−(aq). These spectator ions—ions whose presence is required to maintain charge neutrality—are neither chemically nor physically changed by the process, and so they may be eliminated from the equation to yield a more succinct representation called a net ionic equation:
Following the convention of using the smallest possible integers as coefficients, this equation is then written:
This net ionic equation indicates that solid silver chloride may be produced from dissolved chloride and silver(I) ions, regardless of the source of these ions. These molecular and complete ionic equations provide additional information, namely, the ionic compounds used as sources of Cl− and Ag+.
Example 3
Write the complete ionic equation for each chemical reaction.
a) KBr(aq) + AgC2H3O2(aq) [latex]\longrightarrow[/latex] KC2H3O2(aq) + AgBr(s)
b) MgSO4(aq) + Ba(NO3)2(aq) [latex]\longrightarrow[/latex] Mg(NO3)2(aq) + BaSO4(s)
Solution
For any ionic compound that is aqueous, we will write the compound as separated ions.
a) The complete ionic equation is
K+(aq) + Br−(aq) + Ag+(aq) + C2H3O2−(aq) [latex]\longrightarrow[/latex] K+(aq) + C2H3O2−(aq) + AgBr(s)
b) The complete ionic equation is
Mg2+(aq) + SO42−(aq) + Ba2+(aq) + 2NO3−(aq) [latex]\longrightarrow[/latex] Mg2+(aq) + 2NO3−(aq) + BaSO4(s)
Test Yourself
Write the complete ionic equation for
CaCl2(aq) + Pb(NO3)2(aq) [latex]\longrightarrow[/latex] Ca(NO3)2(aq) + PbCl2(s)
Answer
Ca2+(aq) + 2Cl−(aq) + Pb2+(aq) + 2NO3−(aq) [latex]\longrightarrow[/latex] Ca2+(aq) + 2NO3−(aq) + PbCl2(s)
Example 4
Write the net ionic equation for each chemical reaction.
a) K+(aq) + Br−(aq) + Ag+(aq) + C2H3O2−(aq) [latex]\longrightarrow[/latex] K+(aq) + C2H3O2−(aq) + AgBr(s)
b) Mg2+(aq) + SO42−(aq) + Ba2+(aq) + 2 NO3−(aq) [latex]\longrightarrow[/latex] Mg2+(aq) + 2 NO3−(aq) + BaSO4(s)
Solution
a) In the first equation, the K+(aq) and C2H3O2−(aq) ions are spectator ions, so they are canceled:
K+(aq) + Br−(aq) + Ag+(aq) + C2H3O2−(aq) [latex]\longrightarrow[/latex] K+(aq) + C2H3O2−(aq) + AgBr(s)
The net ionic equation is
Br−(aq) + Ag+(aq) [latex]\longrightarrow[/latex] AgBr(s)
b) In the second equation, the Mg2+(aq) and NO3−(aq) ions are spectator ions, so they are canceled:
Mg2+(aq) + SO42−(aq) + Ba2+(aq) + 2 NO3−(aq) [latex]\longrightarrow[/latex] Mg2+(aq) + 2 NO3−(aq) + BaSO4(s)
The net ionic equation is
SO42−(aq) + Ba2+(aq) [latex]\longrightarrow[/latex] BaSO4(s)
Test Yourself
Write the net ionic equation for
CaCl2(aq) + Pb(NO3)2(aq) [latex]\longrightarrow[/latex] Ca(NO3)2(aq) + PbCl2(s)
Answer
Pb2+(aq) + 2Cl−(aq) [latex]\longrightarrow[/latex] PbCl2(s)
Example 5
When carbon dioxide is dissolved in an aqueous solution of sodium hydroxide, the mixture reacts to yield aqueous sodium carbonate and liquid water. Write balanced molecular, complete ionic, and net ionic equations for this process.
Solution
Begin by identifying formulas for the reactants and products and arranging them properly in chemical equation form:
Balance is achieved easily in this case by changing the coefficient for NaOH to 2, resulting in the molecular equation for this reaction:
The two dissolved ionic compounds, NaOH and Na2CO3, can be represented as dissociated ions to yield the complete ionic equation:
Finally, identify the spectator ion(s), in this case Na+(aq), and remove it from each side of the equation to generate the net ionic equation:
[latex]\text{CO}_2(aq) + \rule[0.5ex]{4.25em}{0.1ex}\hspace{-4.25em} 2\text{Na}^{+}(aq) + 2\text{OH}^{-}(aq) \longrightarrow \rule[0.5ex]{4.25em}{0.1ex}\hspace{-4.25em} 2\text{Na}^{+}(aq) + {\text{CO}_3}^{2-}(aq) + \text{H}_2 \text{O}(l)[/latex][latex]\text{CO}_2(aq) + 2\text{OH}^{-}(aq) \longrightarrow {\text{CO}_3}^{2-}(aq) + \text{H}_2 \text{O}(l)[/latex]
Test Yourself
Diatomic chlorine and sodium hydroxide (lye) are commodity chemicals produced in large quantities, along with diatomic hydrogen, via the electrolysis of brine, according to the following unbalanced equation:
Write balanced molecular, complete ionic, and net ionic equations for this process.
Answers
[latex]2\text{NaCl}(aq) + 2\text{H}_2 \text{O} \longrightarrow 2 \text{NaOH}(aq) + \text{H}_2(g) + \text{Cl}_2(g) (\text{molecular})[/latex]
[latex]2\text{Na}^{+}(aq) + 2\text{Cl}^{-}(aq) + 2\text{H}_2 \text{O} \longrightarrow 2\text{Na}^{+}(aq) + 2\text{OH}^{-}(aq) + \text{H}_2(g) + \text{Cl}_2(g) (\text{complete ionic})[/latex]
[latex]2\text{Cl}^{-}(aq) + 2\text{H}_2 \text{O} \longrightarrow 2\text{OH}^{-}(aq) + 2\text{H}_2(g) + \text{Cl}_2(g) (\text{net ionic})[/latex]
Chemistry Is Everywhere: Soluble and Insoluble Ionic Compounds
The concept of solubility versus insolubility in ionic compounds is a matter of degree. Some ionic compounds are very soluble, some are only moderately soluble, and some are soluble so little that they are considered insoluble. For most ionic compounds, there is also a limit to the amount of compound can be dissolved in a sample of water. For example, you can dissolve a maximum of 36.0 g of NaCl in 100 g of water at room temperature, but you can dissolve only 0.00019 g of AgCl in 100 g of water. We consider NaCl soluble but AgCl insoluble.

One place where solubility is important is in the tank-type water heater found in many homes in the United States. Domestic water frequently contains small amounts of dissolved ionic compounds, including calcium carbonate (CaCO3). However, CaCO3 has the relatively unusual property of being less soluble in hot water than in cold water. So as the water heater operates by heating water, CaCO3 can precipitate if there is enough of it in the water. This precipitate, called limescale, can also contain magnesium compounds, hydrogen carbonate compounds, and phosphate compounds. The problem is that too much limescale can impede the function of a water heater, requiring more energy to heat water to a specific temperature or even blocking water pipes into or out of the water heater, causing dysfunction.
Another place where solubility versus insolubility is an issue is the Grand Canyon. We usually think of rock as insoluble. But it is actually ever so slightly soluble. This means that over a period of about two billion years, the Colorado River carved rock from the surface by slowly dissolving it, eventually generating a spectacular series of gorges and canyons. And all because of solubility!
Key Concepts and Summary
Chemical equations are symbolic representations of chemical and physical changes. Formulas for the substances undergoing the change (reactants) and substances generated by the change (products) are separated by an arrow and preceded by integer coefficients indicating their relative numbers. Balanced equations are those whose coefficients result in equal numbers of atoms for each element in the reactants and products. Chemical reactions in aqueous solution that involve ionic reactants or products may be represented more realistically by complete ionic equations and, more succinctly, by net ionic equations.
Exercises
1. What does it mean to say an equation is balanced? Why is it important for an equation to be balanced?
2. Balance the following equations:
a) [latex]\text{PCl}_5(s) + \text{H}_2 \text{O}(l) \longrightarrow \text{POCl}_3(l) + \text{HCl}(aq)[/latex]
b) [latex]\text{Cu}(s) + \text{HNO}_3(aq) \longrightarrow \text{Cu(NO}_3)_2(aq) + \text{H}_2 \text{O}(l) + \text{NO}(g)[/latex]
c) [latex]\text{H}_2(g) + \text{I}_2(s) \longrightarrow \text{HI}(s)[/latex]
d) [latex]\text{Fe}(s) + \text{O}_2(g) \longrightarrow \text{Fe}_2 \text{O}_3(s)[/latex]
e) [latex]\text{Na}(s) + \text{H}_2 \text{O}(l) \longrightarrow \text{NaOH}(aq) + \text{H}_2(g)[/latex]
f) [latex]\text{(NH}_4)_2 \text{Cr}_2\text{O}_7(s) \longrightarrow \text{Cr}_2\text{O}_3(s) + \text{N}_2(g) + \text{H}_2 \text{O}(g)[/latex]
g) [latex]\text{P}_4(s) + \text{Cl}_2(g) \longrightarrow \text{PCl}_3(l)[/latex]
h) [latex]\text{PtCl}_4(s) \longrightarrow \text{Pt}(s) + \text{Cl}_2(g)[/latex]
3. Balance the following equations:
a) [latex]\text{Ag}(s) + \text{H}_2 \text{S}(g) + \text{O}_2(g) \longrightarrow \text{Ag}_2 \text{S}(s) + \text{H}_2 \text{O}(l)[/latex]
b) [latex]\text{P}_4(s) + \text{O}_2(g) \longrightarrow \text{P}_4 \text{O}_{10}(s)[/latex]
c) [latex]\text{Pb}(s) + \text{H}_2 \text{O}(l) + \text{O}_2(g) \longrightarrow \text{Pb(OH)}_2(s)[/latex]
d) [latex]\text{Fe}(s) + \text{H}_2 \text{O}(l) \longrightarrow \text{Fe}_3 \text{O}_4(s) + \text{H}_2(g)[/latex]
e) [latex]\text{Sc}_2 \text{O}_3(s) + \text{SO}_3(l) \longrightarrow \text{Sc}_2 \text{(SO}_4)_3(s)[/latex]
f) [latex]\text{Ca}_3 \text{(PO}_4)_2(aq) + \text{H}_3 \text{PO}_4(aq) \longrightarrow \text{Ca(H}_2 \text{PO}_4)_2(aq)[/latex]
g) [latex]\text{Al}(s) + \text{H}_2 \text{SO}_4(aq) \longrightarrow \text{Al}_2 \text{(SO}_4)_3(s) + \text{H}_2(g)[/latex]
h) [latex]\text{TiCl}_4(s) + \text{H}_2 \text{O}(g) \longrightarrow \text{TiO}_2(s) + \text{HCl}(g)[/latex]
4. Write a balanced molecular equation describing each of the following chemical reactions.
a) Solid calcium carbonate is heated and decomposes to solid calcium oxide and carbon dioxide gas.
b) Gaseous butane, C4H10, reacts with diatomic oxygen gas to yield gaseous carbon dioxide and water vapor.
c) Aqueous solutions of magnesium chloride and sodium hydroxide react to produce solid magnesium hydroxide and aqueous sodium chloride.
d) Water vapor reacts with sodium metal to produce solid sodium hydroxide and hydrogen gas.
5. Colorful fireworks often involve the decomposition of barium nitrate and potassium chlorate and the reaction of the metals magnesium, aluminum, and iron with oxygen.
a) Write the formulas of barium nitrate and potassium chlorate.
b) The decomposition of solid potassium chlorate leads to the formation of solid potassium chloride and diatomic oxygen gas. Write an equation for the reaction.
c) The decomposition of solid barium nitrate leads to the formation of solid barium oxide, diatomic nitrogen gas, and diatomic oxygen gas. Write an equation for the reaction.
d) Write separate equations for the reactions of the solid metals magnesium, aluminum, and iron with diatomic oxygen gas to yield the corresponding metal oxides. (Assume the iron oxide contains Fe3+ ions.)
6. Fill in the blank with a single chemical formula for a covalent compound that will balance the equation: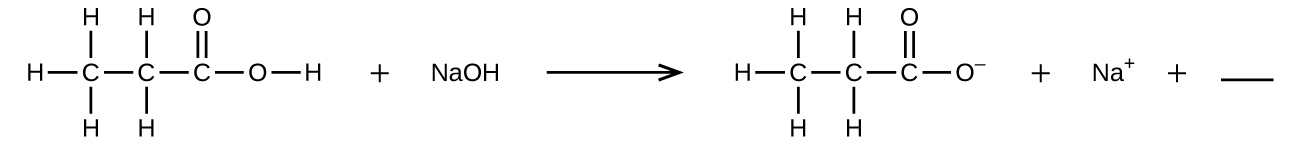
7. Aqueous hydrogen fluoride (hydrofluoric acid) is used to etch glass and to analyze minerals for their silicon content. Hydrogen fluoride will also react with sand (silicon dioxide).
a) Write an equation for the reaction of solid silicon dioxide with hydrofluoric acid to yield gaseous silicon tetrafluoride and liquid water.
b) The mineral fluorite (calcium fluoride) occurs extensively in Illinois. Solid calcium fluoride can also be prepared by the reaction of aqueous solutions of calcium chloride and sodium fluoride, yielding aqueous sodium chloride as the other product. Write complete and net ionic equations for this reaction.
8. From the balanced molecular equations, write the complete ionic and net ionic equations for the following:
a) [latex]\text{K}_2 \text{C}_2 \text{O}_4(aq) + \text{Ba(OH)}_2(aq) \longrightarrow 2\text{KOH}(aq) + \text{BaC}_2 \text{O}_2(s)[/latex]
b) [latex]{\text{Pb(NO}_3)}_2(aq) + \text{H}_2 \text{SO}_4(aq) \longrightarrow \text{PbSO}_4(s) + 2\text{HNO}_3(aq)[/latex]
c) [latex]\text{CaCO}_3(s) + \text{H}_2 \text{SO}_4(aq) \longrightarrow \text{CaSO}_4(s) + \text{CO}_2(g) + \text{H}_2\text{O}(l)[/latex]
9. From the statement “nitrogen gas and hydrogen gas react to produce ammonia gas,” identify the reactants and the products.
10. From the statement “a solution of magnesium hydroxide reacts with a solution of nitric acid to produce a solution of magnesium nitrate and water,” identify the reactants and the products.
11. Write and balance the chemical equation described by Exercise 1.
12. Write and balance the chemical equation described by Exercise 2.
13. Balance: ___NaClO3 [latex]\longrightarrow[/latex] ___NaCl + ___O2
14. Balance: ___N2 + ___H2 [latex]\longrightarrow[/latex] ___N2H4
15. Balance: ___Al + ___O2 [latex]\longrightarrow[/latex] ___Al2O3
16. Balance: ___C2H4 + ___O2 [latex]\longrightarrow[/latex] ___CO2 + ___H2O
17. Balance: ___N2(g) + ___H2(g) [latex]\longrightarrow[/latex] ___NH3(g)
18. Write a chemical equation that represents NaBr(s) dissociating in water.
19. Write a chemical equation that represents (NH4)3PO4(s) dissociating in water.
20. Write the complete ionic equation for the reaction of FeCl2(aq) and AgNO3(aq). You may have to consult the solubility rules.
21. Write the complete ionic equation for the reaction of KCl(aq) and NaC2H3O2(aq). You may have to consult the solubility rules.
22. Write the net ionic equation for the reaction of FeCl2(aq) and AgNO3(aq). You may have to consult the solubility rules.
23. Write the net ionic equation for the reaction of KCl(aq) and NaC2H3O2(aq). You may have to consult the solubility rules.
24. Identify the spectator ions in Exercises 20 and 21.
Answers
1. An equation is balanced when the same number of each element is represented on the reactant and product sides. Equations must be balanced to accurately reflect the law of conservation of matter.
2. a) [latex]\text{PCl}_5(s) + \text{H}_2 \text{O}(l) \longrightarrow \text{POCl}_3(l) + 2\text{HCl}(aq)[/latex]
b) [latex]3\text{Cu}(s) + 8\text{HNO}_3(aq) \longrightarrow 3\text{Cu(NO}_3)_2(aq) + 4\text{H}_2 \text{O}(l) + 2\text{NO}(g)[/latex]
c) [latex]\text{H}_2(g) + \text{I}_2(s) \longrightarrow 2\text{HI}(s)[/latex]
d) [latex]4\text{Fe}(s) + 3\text{O}_2(g) \longrightarrow 2\text{Fe}_2 \text{O}_3(s)[/latex]
e) [latex]2\text{Na}(s) + 2\text{H}_2 \text{O}(l) \longrightarrow 2\text{NaOH}(aq) + \text{H}_2(g)[/latex]
f) [latex]\text{(NH}_4)_2 \text{Cr}_2\text{2O}_7(s) \longrightarrow \text{Cr}_2\text{O}_3(s) + \text{N}_2(g) + 4\text{H}_2 \text{O}(l)[/latex]
g) [latex]\text{P}_4(s) + 6\text{Cl}_2(g) \longrightarrow 4\text{PCl}_3(l)[/latex]
h) [latex]\text{PtCl}_4(s) \longrightarrow \text{Pt}(s) + 2\text{Cl}_2(g)[/latex]
3. a) [latex]4\text{Ag}(s) + 2\text{H}_2 \text{S}(g) + \text{O}_2(g) \longrightarrow 2\text{Ag}_2 \text{S}(s) + 2\text{H}_2 \text{O}(l)[/latex]
b) [latex]\text{P}_4(s) + 5\text{O}_2(g) \longrightarrow \text{P}_4 \text{O}_{10}(s)[/latex]
c) [latex]2\text{Pb}(s) + 2\text{H}_2 \text{O}(l) + \text{O}_2(g) \longrightarrow 2\text{Pb(OH)}_2(s)[/latex]
d) [latex]3\text{Fe}(s) + 4\text{H}_2 \text{O}(l) \longrightarrow \text{Fe}_3 \text{O}_4(s) + 4\text{H}_2(g)[/latex]
e) [latex]\text{Sc}_2 \text{O}_3(s) + 3\text{SO}_3(l) \longrightarrow \text{Sc}_2 \text{(SO}_4)_3(s)[/latex]
f) [latex]\text{Ca}_3 \text{(PO}_4)_2(aq) + 4\text{H}_3 \text{PO}_4(aq) \longrightarrow 3\text{Ca(H}_2 \text{PO}_4)_2(aq)[/latex]
g) [latex]2\text{Al}(s) + 3\text{H}_2 \text{SO}_4(aq) \longrightarrow \text{Al}_2 \text{(SO}_4)_3(s) + 3\text{H}_2(g)[/latex]
h) [latex]\text{TiCl}_4(s) + 2\text{H}_2 \text{O}(g) \longrightarrow \text{TiO}_2(s) + 4\text{HCl}(g)[/latex]
4.a) [latex]\text{CaCO}_3(s) \longrightarrow \text{CaO}(s) + \text{CO}_2(g)[/latex]
b) [latex]2\text{C}_4 \text{H}_{10}(g) + 13 \text{O}_2(g) \longrightarrow 8\text{CO}_2(g) + 10\text{H}_2\text{O}(g)[/latex]
c) [latex]\text{MgCl}_{2}(aq) + 2 \text{NaOH}(aq) \longrightarrow \text{Mg(OH)}_2(s) + 2 \text{NaCl}(aq)[/latex]
d) [latex]2\text{H}_2 \text{O}(g) + 2 \text{Na}(s) \longrightarrow 2\text{NaOH}(s) + \text{H}_2(g)[/latex]
5. a) [latex]\text{Ba(NO}_3)_2[/latex] , [latex]\text{KClO}_3[/latex]
b) [latex]2 \text{KClO}_3(s) \longrightarrow 2 \text{KCl}(s) + 3\text{O}_2(g)[/latex]
c) [latex]2 \text{Ba(NO}_3)_2(s) \longrightarrow 2\text{BaO}(s) + 2\text{N}_2(g) + 5\text{O}_2(g)[/latex]
d) [latex]2 \text{Mg}(s) + \text{O}_2(g) \longrightarrow 2 \text{MgO}(s)[/latex]; [latex]4\text{Al}(s) + 3\text{O}_2(g) \longrightarrow 2\text{Al}_2 \text{O}_3(g)[/latex]; [latex]4\text{Fe}(s) + 3\text{O}_2(g) \longrightarrow 2\text{Fe}_2 \text{O}_3(s)[/latex]
6. H2O
7. a) [latex]4\text{HF}(aq) + \text{SiO}_2(s) \longrightarrow \text{SiF}_4(g) + 2\text{H}_2 \text{O}(l)[/latex]
b) complete: [latex]2\text{Na}^{+}(aq) + 2\text{F}^{-}(aq) + \text{Ca}^{2+}(aq) + 2\text{Cl}^{-}(aq) \longrightarrow \text{CaF}_2(s) + 2\text{Na}^{+}(aq) + 2\text{Cl}^{-}(aq)[/latex]
net: [latex]2\text{F}^{-}(aq) + \text{Ca}^{2+}(aq) \longrightarrow \text{CaF}_2(s)[/latex]
8. a) complete: [latex]2\text{K}^{+}(aq) + {\text{C}_2 \text{O}_4}^{2-}(aq) + \text{Ba}^{2+}(aq) + 2\text{OH}^{-}(aq) \longrightarrow 2\text{K}^{+}(aq) + 2\text{OH}^{-}(aq) + \text{BaC}_2 \text{O}_4(s)[/latex]
net: [latex]\text{Ba}^{2+}(aq) + {\text{C}_2 {\text{O}_4}}^{2-}(aq) \longrightarrow \text{BaC}_2 \text{O}_4(s)[/latex]
b) complete: [latex]\text{Pb}^{2+}(aq) + 2{\text{NO}_3}^{-}(aq) + 2\text{H}^{+}(aq) + {\text{SO}_4}^{2-}(aq) \longrightarrow \text{PbSO}_4(s) + 2\text{H}^{+}(aq) + 2{\text{NO}_3}^{-}(aq)[/latex]
net: [latex]\text{Pb}^{2+}(aq) + {\text{SO}_4}^{2-}(aq) \longrightarrow \text{PbSO}_4(s)[/latex]
c) complete: [latex]\text{CaCO}_3(s) + 2\text{H}^{+}(aq) + {\text{SO}_4}^{2-}(aq) \longrightarrow \text{CaSO}_4(s) + \text{CO}_2(g) + \text{H}_2 \text{O}(l)[/latex]
net: [latex]\text{CaCO}_3(s) + 2\text{H}^{+}(aq) + {\text{SO}_4}^{2-}(aq) \longrightarrow \text{CaSO}_4(s) + \text{CO}_2(g) + \text{H}_2 \text{O}(l)[/latex]
9. reactants: nitrogen and hydrogen; product: ammonia
10. reactants: magnesium hydroxide and nitric acid; products: magnesium nitrate and water
11. N2(g) + 3 H2(g) [latex]\longrightarrow[/latex] 2 NH3(g)
12. Mg(OH)2(aq) + 2 HNO3(aq) [latex]\longrightarrow[/latex] Mg(NO3)2(aq) + 2 H2O(ℓ)
13. 2 NaClO3 [latex]\longrightarrow[/latex] 2 NaCl + 3 O2
14. N2 + 2 H2 [latex]\longrightarrow[/latex] N2H4
15. 4Al + 3O2 [latex]\longrightarrow[/latex] 2Al2O3
16. C2H4 + 3 O2 [latex]\longrightarrow[/latex] 2 CO2 + 2 H2O
17. N2(g) + 3 H2(g) [latex]\longrightarrow[/latex] 2 NH3(g)
18. NaBr(s) [latex]\longrightarrow[/latex] Na+(aq) + Br−(aq)
19. (NH4)3PO4(s) [latex]\longrightarrow[/latex] 3 NH4+(aq) + PO43−(aq)
20. Fe2+(aq) + 2 Cl−(aq) + 2 Ag+(aq) + 2 NO3−(aq) [latex]\longrightarrow[/latex] Fe2+(aq) + 2 NO3−(aq) + 2 AgCl(s)
21. K+(aq) + Cl−(aq) + Na+(aq) + C2H3O2−(aq) [latex]\longrightarrow[/latex] Na+(aq) + Cl−(aq) + K+(aq) + C2H3O2−(aq)
22. 2 Cl−(aq) + 2 Ag+(aq) [latex]\longrightarrow[/latex] 2 AgCl(s)
23. There is no overall reaction.
24. In Exercise 20, Fe2+(aq) and NO3−(aq) are spectator ions; in Exercise 21, Na+(aq) and Cl−(aq) are spectator ions.
Glossary
balanced equation: chemical equation with equal numbers of atoms for each element in the reactant and product
chemical equation: symbolic representation of a chemical reaction
coefficient: number placed in front of symbols or formulas in a chemical equation to indicate their relative amount
complete ionic equation: chemical equation in which all dissolved ionic reactants and products, including spectator ions, are explicitly represented by formulas for their dissociated ions
molecular equation: chemical equation in which all reactants and products are represented as neutral substances
net ionic equation: chemical equation in which only those dissolved ionic reactants and products that undergo a chemical or physical change are represented (excludes spectator ions)
product: substance formed by a chemical or physical change; shown on the right side of the arrow in a chemical equation
reactant: substance undergoing a chemical or physical change; shown on the left side of the arrow in a chemical equation
spectator ion: ion that does not undergo a chemical or physical change during a reaction, but its presence is required to maintain charge neutrality