Chapter 6. Chemical Reactions and Equations
6.2 Precipitation Reactions
Learning Objectives
By the end of this section, you will be able to:
- Define precipitation reactions
- Recognize and identify examples of precipitation reactions
- Predict the solubility of common inorganic compounds by using solubility rules
Humans interact with one another in various and complex ways, and we classify these interactions according to common patterns of behavior. When two humans exchange information, we say they are communicating. When they exchange blows with their fists or feet, we say they are fighting. Faced with a wide range of varied interactions between chemical substances, scientists have likewise found it convenient (or even necessary) to classify chemical interactions by identifying common patterns of reactivity. The following sections of this chapter (section 6.2-6.4) will provide an introduction to three of the most prevalent types of chemical reactions: precipitation, acid-base, and oxidation-reduction.
Precipitation Reactions and Solubility Rules
A precipitation reaction is one in which dissolved substances react to form one (or more) solid products. Many reactions of this type involve the exchange of ions between ionic compounds in aqueous solution and are sometimes referred to as double displacement, double replacement, or metathesis reactions. These reactions are common in nature and are responsible for the formation of coral reefs in ocean waters and kidney stones in animals. They are used widely in industry for production of a number of commodity and specialty chemicals. Precipitation reactions also play a central role in many chemical analysis techniques, including spot tests used to identify metal ions and gravimetric methods for determining the composition of matter (see the last module of this chapter).
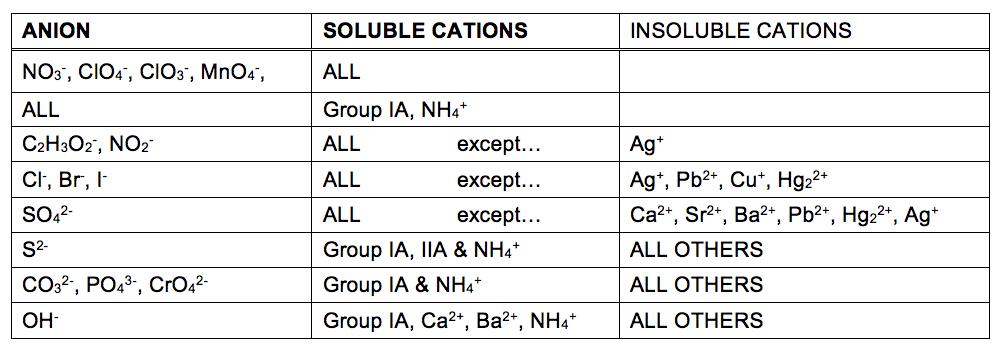
The extent to which a substance may be dissolved in water, or any solvent, is quantitatively expressed as its solubility, defined as the maximum concentration of a substance that can be achieved under specified conditions. Substances with relatively large solubilities are said to be soluble. A substance will precipitate when solution conditions are such that its concentration exceeds its solubility. Substances with relatively low solubilities are said to be insoluble, and these are the substances that readily precipitate from solution. More information on these important concepts is provided in the text chapter on solutions. For purposes of predicting the identities of solids formed by precipitation reactions, one may simply refer to patterns of solubility that have been observed for many ionic compounds (Table 1).
A vivid example of precipitation is observed when solutions of potassium iodide and lead nitrate are mixed, resulting in the formation of solid lead iodide:
This observation is consistent with the solubility guidelines: The only insoluble compound among all those involved is lead iodide, one of the exceptions to the general solubility of iodide salts.
The net ionic equation representing this reaction is:
Lead iodide is a bright yellow solid that was formerly used as an artist’s pigment known as iodine yellow (Figure 1). The properties of pure PbI2 crystals make them useful for fabrication of X-ray and gamma ray detectors.

The solubility table in Table 1 may be used to predict whether a precipitation reaction will occur when solutions of soluble ionic compounds are mixed together. One merely needs to identify all the ions present in the solution and then consider if possible cation/anion pairing could result in an insoluble compound.
For example, mixing solutions of silver nitrate and sodium fluoride will yield a solution containing Ag+, NO3−, Na+, and F− ions. Aside from the two ionic compounds originally present in the solutions, AgNO3 and NaF, two additional ionic compounds may be derived from this collection of ions: NaNO3 and AgF.
The solubility table indicate all nitrate salts are soluble but that AgF is one of the exceptions to the general solubility of fluoride salts. A precipitation reaction, therefore, is predicted to occur, as described by the following equations:
[latex]\text{NaF}(aq) + \text{AgNO}_3(aq) \longrightarrow \text{AgF}(s) + \text{NaNO}_3(aq) \;\text{(molecular)}[/latex][latex]\text{Ag}^{+}(aq) + \text{F}^{-}(aq) \longrightarrow \text{AgF}(s) \;\text{(net ionic)}[/latex]
Example 1
Predict the result of mixing reasonably concentrated solutions of the following ionic compounds. If precipitation is expected, write a balanced net ionic equation for the reaction.
a) potassium sulfate and barium nitrate
b) lithium chloride and silver acetate
c) lead nitrate and ammonium carbonate
Solution
a) The two possible products for this combination are KNO3 and BaSO4. The solubility guidelines indicate BaSO4 is insoluble, and so a precipitation reaction is expected. The net ionic equation for this reaction, derived in the manner detailed in the previous module, is
b) The two possible products for this combination are LiC2H3O2 and AgCl. The solubility guidelines indicate AgCl is insoluble, and so a precipitation reaction is expected. The net ionic equation for this reaction, derived in the manner detailed in the previous module, is
c) The two possible products for this combination are PbCO3 and NH4NO3. The solubility guidelines indicate PbCO3 is insoluble, and so a precipitation reaction is expected. The net ionic equation for this reaction, derived in the manner detailed in the previous module, is
Test Yourself
Which solution could be used to precipitate the barium ion, Ba2+, in a water sample: sodium chloride, sodium hydroxide, or sodium sulfate? What is the formula for the expected precipitate?
Answers
sodium sulfate, BaSO4
Key Concepts and Summary
Chemical reactions are classified according to similar patterns of behavior. Precipitation is one type of chemical reaction which involves the formation of one or more insoluble products. Precipitation reactions, also called double displacement reactions can be summarized with the following reaction equation:
[latex]\text{AB}(aq) + \text{CD}(aq) \longrightarrow \text{AD}(s) + \text{CB}(aq) or (s)[/latex]
The formation of the solid is the DRIVING FORCE of the reaction (the factor that makes the reaction go).
A precipitation reaction can be predicted to occur with the help of a solubility table (Table 1). There are three ways of representing a precipitation reaction, using a molecular equation, complete ionic equation or net ionic equation, as described in section 6.1.
Exercises
1. What are the general characteristics that help you recognize double replacement reactions?
a) Zn(NO3)2 + NaOH [latex]\longrightarrow[/latex] ? b) HCl + Na2S [latex]\longrightarrow[/latex] ?
3. Assuming that each double replacement reaction occurs, predict the products and write each balanced chemical equation.
a) Ca(C2H3O2)2 + HNO3 [latex]\longrightarrow[/latex] ? b) Na2CO3 + Sr(NO2)2 [latex]\longrightarrow[/latex] ?
4. Assuming that each double replacement reaction occurs, predict the products and write each balanced chemical equation.
a) Pb(NO3)2 + KBr [latex]\longrightarrow[/latex] ? b) K2O + MgCO3 [latex]\longrightarrow[/latex] ?
5. Assuming that each double replacement reaction occurs, predict the products and write each balanced chemical equation.
a) Sn(OH)2 + FeBr3 [latex]\longrightarrow[/latex] ? b) CsNO3 + KCl [latex]\longrightarrow[/latex] ?
6. Use the solubility table (Table 1) to predict if each double replacement reaction will occur and, if so, write a balanced chemical equation.
a) Pb(NO3)2 + KBr [latex]\longrightarrow[/latex] ? b) K2O + Na2CO3 [latex]\longrightarrow[/latex] ?
7. Use the solubility table (Table 1) to predict if each double replacement reaction will occur and, if so, write a balanced chemical equation.
a) Na2CO3 + Sr(NO3)2 [latex]\longrightarrow[/latex] ? b) (NH4)2SO4 + Ba(NO3)2 [latex]\longrightarrow[/latex] ?
8. Use the solubility rules to predict if each double replacement reaction will occur and, if so, write a balanced chemical equation.
a) K3PO4 + SrCl2 [latex]\longrightarrow[/latex] ? b) NaOH + MgCl2 [latex]\longrightarrow[/latex] ?
9. Use the solubility rules to predict if each double replacement reaction will occur and, if so, write a balanced chemical equation.
a) KC2H3O2 + Li2CO3 [latex]\longrightarrow[/latex] ? b) KOH + AgNO3 [latex]\longrightarrow[/latex] ?
Answers
1. A double replacement reaction occurs when parts of two ionic compounds are exchanged, making two new compounds. A characteristic of a double-replacement equation is that there are two compounds as reactants and two different compounds as products.
2. a) Zn(NO3)2 + 2 NaOH [latex]\longrightarrow[/latex] Zn(OH)2 + 2 NaNO3
b) 2 HCl + Na2S [latex]\longrightarrow[/latex] 2 NaCl + H2S
3. a) Ca(C2H3O2)2 + 2 HNO3 [latex]\longrightarrow[/latex] Ca(NO3)2 + 2 HC2H3O2
b) Na2CO3 + Sr(NO2)2 [latex]\longrightarrow[/latex] 2 NaNO2 + SrCO3
4.a) Pb(NO3)2 + 2 KBr [latex]\longrightarrow[/latex] PbBr2 + 2 KNO3
b) K2O + MgCO3 [latex]\longrightarrow[/latex] K2CO3 + MgO
5. a) 3 Sn(OH)2 + 2 FeBr3 [latex]\longrightarrow[/latex] 3 Sn(Br)2 + 2 Fe(OH)3
b) CsNO3 + KCl [latex]\longrightarrow[/latex] KNO3 + CsCl
6.a) Pb(NO3)2(aq) + 2 KBr(aq) [latex]\longrightarrow[/latex] PbBr2(s) + 2 KNO3(aq)
b) No reaction occurs.
7. a) Na2CO3(aq) + Sr(NO3)2(aq) [latex]\longrightarrow[/latex] 2 NaNO3(aq) + SrCO3(s)
b) (NH4)2SO4(aq) + Ba(NO3)2(aq) [latex]\longrightarrow[/latex] BaSO4(s) + 2 NH4NO3(aq)
8.a) 2 K3PO4(aq) + 3 SrCl2(aq) [latex]\longrightarrow[/latex] Sr3(PO4)2(s) + 6 KCl(aq)
b) 2 NaOH(aq) + MgCl2(aq) [latex]\longrightarrow[/latex] 2 NaCl(aq) + Mg(OH)2(s)
9. a) No reaction occurs.
b) KOH(aq) + AgNO3(aq) [latex]\longrightarrow[/latex] AgOH(s) + KNO3(aq)
Glossary
insoluble: of relatively low solubility; dissolving only to a slight extent
precipitate: insoluble product that forms from reaction of soluble reactants
precipitation reaction: reaction that produces one or more insoluble products; when reactants are ionic compounds, sometimes called double displacement or metathesis
salt: ionic compound that can be formed by the reaction of an acid with a base that contains a cation and an anion other than hydroxide or oxide
soluble: of relatively high solubility; dissolving to a relatively large extent
solubility: the extent to which a substance may be dissolved in water, or any solvent

