Chapter 10. Organic Chemistry
10.7 Summary of Nomenclature Rules
It is important to note that to be able to name an organic compounds, you must be able to easily identify the functional groups: alkane, alkene, alkyne, arene, alcohol, ether, amine, aldehyde, ketone, carboxylic acid, ester, amide.
The IUPAC systematic methods of naming most of the functional groups seen follow a similar procedure described in the nomenclature guide.
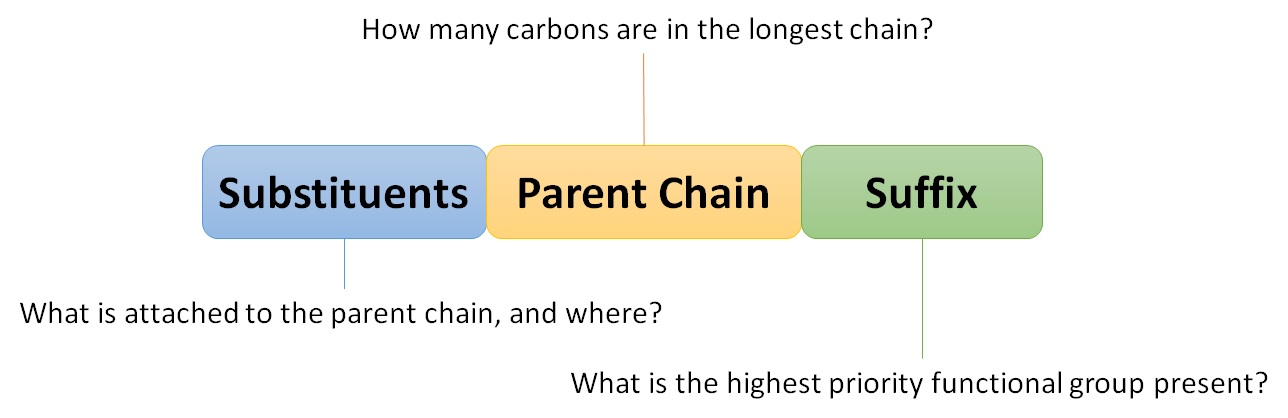
The names are made of three main parts: 1) specifying the information about the substituents; 2) specifying the information about the parent chain (or ring); and 3) the ending which specifies what functional group is present in the structure being named.
| Two Rules | |
|
Alkanes: #-substituents–PREFIX+ANE |
Rule 1. Identify the longest chain of carbon atoms.
Rule 2. Names and position of the substituents. *** |
|
Alkenes: #-substituents–PREFIX–#-ENE |
Rule 1. Identify the longest chain of carbons which contains the double bond and its position. And when numbering the main chain, the double gets the lowest possible number.Rule 2. Names and position of the substituents.
|
|
Alkynes: #-substituents–PREFIX–#-YNE |
Rule 1. Identify the longest chain of carbons which contains the triple bond and its position. And when numbering the main chain, the triple gets the lowest possible number.Rule 2. Names and position of the substituents. |
| Arenes (specifically benzene derivatives):
#-substituents–BENZENE |
Rule 1. Identify the arene ring (BENZENE).
Rule 2. Names and position (if more than one) of the substituents: If there are two or more substituents on a benzene molecule, the relative positions must be numbered. The substituent that is first alphabetically is assigned position 1, and the ring is numbered in a circle to give the other substituents the lowest possible number(s). |
| Alcohols:
#-substituents–PREFIX–#-AN |
Rule 1. Identify the longest chain of carbons which contains the OH group and its position. And when numbering the parent chain, the hydroxyl group gets the lowest possible number.
Rule 2. Names and position of the substituents.*** |
| Ethers:
PREFIX+OXY–PREFIX–ANE |
Rule 1. Identify the longest carbon branch
Rule 2. Names of the substituent, the other carbon branch (PREFIX+OXY) |
| Aldehydes:
#-substituents–PREFIX–AN |
Rule 1. Identify the longest chain of carbons which contains the carbonyl group (PREFIX-AN Rule 2. Names and position of the substituents.*** |
| Ketones:
#-substituents–PREFIX–AN |
Rule 1. Identify the longest chain of carbons which contains the carbonyl group. And when numbering the parent chain, the carbonyl group gets the lowest possible number. In the smaller ketones (propanone and butanone), the locant number is not used because there is no alternative placement in these smaller ketones.
Rule 2. Names and position of the substituents.*** |
| Carboxylic Acids:
#-substituents–PREFIX–AN |
Rule 1. Identify the longest chain of carbons which contains the carbonyl group. And when numbering the parent chain, the carbonyl group gets the lowest possible number, therefore it is always 1 and therefore is not included in the name.
Rule 2. Names and position of the substituents.*** |
| Esters:
ALKYL #-substituents–PREFIX–AN |
Rule 1. Identify the longest chain of carbons which contains the carbonyl group. And when numbering the parent chain, the carbonyl group gets the lowest possible number, therefore it is always 1 and therefore is not included in the name. AND then name the other carbon chain (PREFIX+YL).
Rule 2. Names and position of the substituents.*** |
| Amides:
#-substituents–PREFIX–AN |
Rule 1. Identify the longest chain of carbons which contains the carbonyl group (PREFIX-AN Rule 2. Names and position of the substituents.*** |
***If there are substituents on the parent chain, their names and position on the chain must be included at the front of the name. The position of a substituent or branch is identified by the number of the carbon atom it is bonded to in the chain. Multiple substituents are named individually and placed in alphabetical order at the front of the name.
It is helpful to recognize the similarities between the rules of alkenes and alkynes, and between the rules of alcohols and the carbonyl functional groups. Ethers and amines have their own unique naming procedures.

