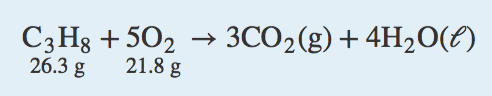Chapter 7. Stoichiometry of Chemical Reactions
7.6 End of Chapter Problems
1. How many molecules of O2 will react with 6.022 × 1023 molecules of H2 to make water?
The reaction is 2 H2(g) + O2(g) [latex]\longrightarrow[/latex] 2 H2O(ℓ).
2. How many moles are present in 6.411 kg of CO2? How many molecules is this?
3. What is the mass in milligrams of 7.22 × 1020 molecules of CO2?
4. What is the mass in grams of 1 molecule of H2O?
5. What is the volume of 3.44 mol of Ga if the density of Ga is 6.08 g/mL?
6. For the chemical reaction
2 C4H10(g) + 13 O2(g) [latex]\longrightarrow[/latex] 8 CO2(g) + 10 H2O(ℓ)
assume that 13.4 g of C4H10 reacts completely to products. The density of CO2 is 1.96 g/L. What volume in liters of CO2 is produced?
7. Calculate the mass of each product when 100.0 g of CuCl react according to the reaction
2 CuCl(aq) [latex]\longrightarrow[/latex] CuCl2(aq) + Cu(s)
What do you notice about the sum of the masses of the products? What concept is being illustrated here?
8. What mass of CO2 is produced from the combustion of 1 gal of gasoline? The chemical formula of gasoline can be approximated as C8H18. Assume that there are 2,801 g of gasoline per gallon.
9. A chemical reaction has a theoretical yield of 19.98 g and a percent yield of 88.40%. What is the actual yield?
10. Given the initial amounts listed, what is the limiting reagent, and how much of the other reactants are in excess?
2 P4 + 6 NaOH + 6 H2O [latex]\longrightarrow[/latex] 3 Na2HPO4 + 5 PH3
Initial amounts used: P4 = 35.0 g; NaOH = 12.7 g; H2O = 9.33 g
11. Verify that it does not matter which product you use to predict the limiting reagent by using both products in this combustion reaction to determine the limiting reagent and the amount of the reactant in excess. Initial amounts of each reactant are given.
12. Chlorine can be produced by the reaction of hydrochloric acid with excess manganese(IV) oxide according to the following reaction: 4HCl(aq) + MnO2(s) [latex]\longrightarrow[/latex] Cl2(g) + 2H2O(l) + MnCl2(aq). How many moles of HCl are needed to form 12.5 mol Cl2?
13. How many moles of aluminum oxide will be produced by reacting 9.5 mol of Al with O2?
How many moles of O2will react?
14. Nitrogen monoxide is oxidized in air to give brown nitrogen dioxide:
2NO(g) + O2(g) [latex]\longrightarrow[/latex] 2NO2(g)
Starting with 2.2 mol NO, how many moles and how many grams of O2 are required for complete reaction? What mass of NO2, in grams, is produced?
15. How many grams of Mg will react with 7.5 grams of H2SO4 in the reaction:
Mg + H2SO4 [latex]\longrightarrow[/latex] MgSO4 + H2
16. Zinc will react with hydrochloric acid producing hydrogen gas and zinc chloride:
Zn + 2HCl [latex]\longrightarrow[/latex] ZnCl2 + H2
How many grams of H2 will be produced if 5.0 grams of zinc are used?
17. The final step in the manufacture of platinum metal (for use in automotive catalytic converters and other products) is the reaction: 3(NH4)2PtCl6(s) [latex]\longrightarrow[/latex] 3Pt(s) + 2NH4Cl(s) + 2N2(g) + 16HCl(g)
How many grams of Pt can be produced by decomposing 12.35 g (NH4)2PtCl6?
18. How many kilograms of NH3 will be produced when 25.0 kg of H2 reacts with excess N2?
19. A 3.00 cm3 piece of aluminum reacts with a solution of HCl and produces H2 gas and AlCl3. Determine the mass of H2 formed. (Al has a density of 2.70 g/cm3)
20. One of the most important commercial reactions is the "Haber" production of ammonia:
3H2(g) + N2(g) [latex]\longrightarrow[/latex] 2NH3(g)
Chiefly by this reaction, the industrial "fixation" of nitrogen now accounts for about one-third of all the nitrogen fixed on our planet. If 3.0 kg of H2 and 1.0 kg of N2 are mixed and allowed to react until one or both of the reactants is used up,
a) how many moles of the NH3 molecules are produced?
b) how many moles of the H2 are left?
c) how many moles of the N2 are left?
21. Iron reacts with chlorine to produce FeCl3 as: 2Fe + 3Cl2 [latex]\longrightarrow[/latex] 2FeCl3
If 10.6 grams of iron are mixed with 18.9 grams of chlorine and allowed to react,
a) how many grams of FeCl3 will be produced?
b) how many grams of excess reactant will be left after the reaction is complete?
22. Silver tarnishes in the presence of hydrogen sulfide in the following reaction:
4Ag + 2H2S + O2 [latex]\longrightarrow[/latex] 2Ag2S + 2H2O
How many grams of Ag2S can be obtained by this reaction from a mixture of 0.950 g Ag, 0.140 g H2S, and 0.0800 g O2?
23. 11.92 g of Pb(NO3)2 and 20.31 g of KI react as: Pb(NO3)2+ 2 KI [latex]\longrightarrow[/latex] PbI2 + 2 KNO3
How many grams of PbI2 are produced if the yield of the reaction is 81%?
24. Reaction of H2 and N2 produces ammonia (NH3) with 65.5%yield. If 30.0 g of NH3 are required, how many grams of N2 and how many of H2 must be used?
25. Solid calcium carbonate dissolves in a solution of hydrochloric acid and reacts to form a solution of calcium chloride and water, and bubbles of carbon dioxide.
How many grams of calcium chloride will form if 40.0 g of CaCO3 is mixed with 0.500 mol of HCl?
How many grams of calcium carbonate, if any, will remain unreacted?
26. Freon-12 (CCl2F2) is a gas that has been used as a refrigerant. It is prepared by the reaction between carbon tetrachloride and antimony trifluoride. The other product that is produced is SbCl3. If the percent yield is 72.0%, how many grams of antimony trifluoride must be treated with excess carbon tetrachloride to obtain an actual yield of 25.0 grams of Freon-12?
27. Aluminum chloride (Al2Cl6) can be made by the following reaction: 2Al(s) + 3Cl2(g) [latex]\longrightarrow[/latex] Al2Cl6(s)
a) which reactant is limiting if 2.70 g Al and 4.05 g Cl2 are mixed?
b) what mass of Al2Cl6 can be produced?
c) What mass of the excess reactant will remain when the reaction is complete?
28. Iron oxide can be reduced to the metal as follows: Fe2O3(s) + 3CO(g) [latex]\longrightarrow[/latex] 2Fe(s) + 3CO2(g)
How many grams of iron can be obtained from 1.00 kg of the iron oxide?
If 654 g Fe was obtained from the reaction, what was the percent yield?
29. Disulfur dichloride can be prepared by the following reaction:
3SCl2(l) + 4NaF(s) [latex]\longrightarrow[/latex] SF4(g) + S2Cl2(l) + 4NaCl(s)
What mass of SCl2is needed to react with excess NaF to prepare 1.19 g S2Cl2, if the yield is 51%?
30. You have a 0.12 M solution of BaCl2. What ions exist in the solution, and what are their concentrations?
31. Assume that 6.73 g Na2CO3is dissolved in enough water to make 250. mL of solution,
a) what is the molarity of the sodium carbonate?
b) What are the concentrations of the Na+and CO32-ions?
32. What is the mass, in grams, of solute in 250. mL of a 0.0125 M solution of KMnO4?
33. What volume of 0.123 M NaOH, in mL, contains 25.0 g NaOH?
34. What is the maximum mass, in grams, of AgCl that can be precipitated by mixing 50.0 mL 0.025 M AgNO3solution with 100.0 mL of 0.025 M NaCl solution? Which reactant is in excess? What is the concentration of the excess reactant remaining in solution after the AgCl has precipitated?
35. How many moles of NaOH react with 7.80 mol of the acid H2SO4?
36. How many mL of 0.512 M NaOH is required to react completely with 25.0 mL of 0.234 M H2SO4?
37. What mass, in grams, of Na2CO3is required for complete reaction with 25.0 mL of 0.155 M HNO3? Na2CO3(aq)+ 2HNO3(aq) [latex]\longrightarrow[/latex] 2NaNO3(aq)+ CO2(g)+ H2O(l)
Answers
1. 3.011 × 1023 molecules of O2
2. 145.7 mol; 8.77 × 1025 molecules
3. 52.8 mg
4. 2.99 × 10−23 g
5. 39.4 mL
6. 20.7 L
7. 67.91 g of CuCl2; 32.09 g of Cu. The two masses add to 100.0 g, the initial amount of starting material, demonstrating the law of conservation of matter.
8. 8633 g
9. 17.66 g
10. The limiting reagent is NaOH; 21.9 g of P4 and 3.61 g of H2O are left over.
11. Both products predict that O2 is the limiting reagent; 20.3 g of C3H8 are left over.
12. 50.0 mol HCl
13. 4.8 mol Al2O3 7.1 mol O2
14. 1.1 mol O2 35 g O2 1.0 x 102g NO2
15. 1.9 g Mg
16. 0.15 g H2
17. 5.428 g Pt
18. 141 kg NH3
19. 0.908 g H2
20. 71 mol NH3 1.4 x 103mol H2 0 mol N2
21. 28.8 g FeCl3 0.7 g Fe
22. 1.02 g Ag2S
23. 13 g PbI2
24. 37.7 g N2 8.13 g H2
25. Must use a balanced equation to solve this:
1 CaCO3(s) + 2 HCl(aq) [latex]\longrightarrow[/latex] 1 CaCl2(aq) + 1 H2O(l) + 1 CO2(g)
27.7g CaCl2 15.0g CaCO3 unreacted
26. 34.2 g SbF3
27. a) Cl2 is limiting b) 5.08 g Al2Cl6 c) 1.67 g Al unreacted
28. 699 g 93.5%
29. 5.3 g SCl2
30. 0.12 M Ba2+, 0.24 M Cl-
31. a) 0.254 M Na2CO3 b) 0.508 M Na+, 0.254 M CO32-
32. 0.494 g KMnO4
33. 5.08 x 103mL
34. 0.18 g AgCl, NaCl, 0.0083 M NaCl
35. 15.6 mol NaOH
36. 22.9 mL NaOH solution
37. 0.205 g Na2CO3