Sim Labs
Objective
To operate the MC-90 ship model fully loaded on “At sea” mode and compare the NOx and SOx emissions when the plant is operating with :
- SCR unit by passed while on “At sea” passage mode at full speed
- SCR unit by passe while on “At sea” passage mode at reduced speed of 45 RPM
- SCR in use while on “At sea” passage mode at full speed
- SCR in use while on “At sea” passage mode at reduced sped of 45 RPM
- SCR unit by passed, but with Fuel emulsion of 20% while “At sea” passage mode at full speed
- SCR unit by passed, but with Fuel emulsion of 20% while “At sea” passage mode at reduced speed of 45 RPM
- SCR unit and Fuel emulsion by passed during maneuvering (various RPM) – 58, 48, 37 and 30
DeliverableYour lab report is to include the following:
|
Theory
NOx: What is it? Where does it come from?
Nitrogen oxides (NOx), are the generic term for a group of highly reactive gases, all of which contain nitrogen and oxygen in varying amounts. Many of the nitrogen oxides are colorless and odorless. However, one common pollutant, nitrogen dioxide (NO2) along with particles in the air can often be seen as a reddish-brown layer over many densely populated industrial and urban areas.
Nitrogen oxides form when fuel is burned at high temperatures, as in a combustion process in a Marine Diesel Engines. The primary sources of NOx are ships, motor vehicles, electric utilities, and other industrial, commercial, and residential sources that burn fuels.
Advances in thermal efficiency have directly contributed to a rise in NOx emission. Dominating factors in the formation of NOx are temperature and oxygen concentration – the higher the temperature and the higher the residence time at the high temperature in the cylinder the greater the amount of NOx that will be generated. A longer combustion time span means that the low speed engines generate more NOx than medium or high speed engines.
NOx and the pollutants formed from NOx can be transported over long distances, following the pattern of prevailing winds. This means that problems associated with NOx are not confined to areas where NOx are emitted. Therefore, controlling NOx is often most effective if done from a regional perspective, rather than focusing on sources in one local area.
NOx emissions are increasing and since 1970, EPA has tracked emissions of the six principal air pollutants – carbon monoxide, lead, nitrogen oxides, particulate matter, sulfur dioxide, and volatile organic compounds. Emissions of all of these pollutants have decreased significantly except for NOx which has increased approximately 10 percent over this period.
Harmful effects of NOx
- contributes to the formation of ground-level ozone, which can trigger serious respiratory problems.
- reacts to form nitrate particles, acid aerosols, as well as NO2, which also cause respiratory problems.
- contributes to formation of acid rain.
- contributes to nutrient overload that deteriorates water quality.
- contributes to atmospheric particles, that cause visibility impairment most noticeable in national parks.
- reacts to form toxic chemicals.
- contributes to global warming.
Ground-level Ozone (Smog) – is formed when NOx and volatile organic compounds (VOCs) react in the presence of heat and sunlight. Children, people with lung diseases such as asthma, and people who work or exercise outside are susceptible to adverse effects such as damage to lung tissue and reduction in lung function. Ozone can be transported by wind currents and cause health impacts far from original sources. Millions of people live in areas that do not meet the health standards for ozone. Other impacts from ozone include damaged vegetation and reduced crop yields
Acid Rain – NOx and sulfur dioxide react with other substances in the air to form acids which fall to earth as rain, fog, snow or dry particles. Some may be carried by wind for hundreds of miles. Acid rain damages; causes deterioration of cars, buildings and historical monuments; and causes lakes and streams to become acidic and unsuitable for many fish.
Particles – NOx reacts with ammonia, moisture, and other compounds to form nitric acid and related particles. Human health concerns include effects on breathing and the respiratory system, damage to lung tissue, and premature death. Small particles penetrate deeply into sensitive parts of the lungs and can cause or worsen respiratory disease such as emphysema and bronchitis, and aggravate existing heart disease.
Water Quality Deterioration – Increased nitrogen loading in water bodies, particularly coastal estuaries, upsets the chemical balance of nutrients used by aquatic plants and animals. Additional nitrogen accelerates “eutrophication,” which leads to oxygen depletion and reduces fish and shellfish populations. NOx emissions in the air are one of the largest sources of nitrogen pollution in the Chesapeake Bay.
Global Warming – One member of the Nitrous oxide (NOx), is a greenhouse gas. It accumulates in the atmosphere with other greenhouse gasses causing a gradual rise in the earth’s temperature. This will lead to increased risks to human health, a rise in the sea level, and other adverse changes to plant and animal habitat.
Toxic Chemicals – In the air, NOx reacts readily with common organic chemicals and even ozone, to form a wide variety of toxic products, some of which may cause biological mutations. Examples of these chemicals include the nitrate radical, nitroarenes, and nitrosamines.
Visibility Impairment – Nitrate particles and nitrogen dioxide can block the transmission of light, reducing visibility in urban areas and on a regional scale in national parks.
Regulations applying to the Marine industry:
The Marpol Protocol of 1997 (Annex VI – Regulations for the Prevention of Air Pollution from Ships). Adoption: 26 September 1997
“Subject to the provision of regulation 3 of this Annex, the operation of each diesel engine to which this regulation applies is prohibited, except when the emission of nitrogen oxides (calculated as the total weighted emission of NO2) from the engine is within the following limits:
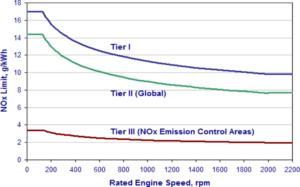
Limits for NOx Emission from a Merchant Vessel
Tier 2 – From 1st Jan 2011
- 14.4g/kWh when n is less than 130 RPM
- 40*n(-0.23) g/kWh when n is 130 or more but less than 2000 RPM
- 7.7 g/kWh when n is 2000 RPM or more
where n = rated engine RPM
Tier 3 (for Emission control areas only) – From 1st Jan 2016, applies only in ECA (not in SECA).
- 3.4g/kWh when n is less than 130 RPM
- 9.0*n(-0.2) g/kWh when n is 130 or more but less than 2000 RPM
- 2.0 g/kWh when n is 2000 RPM or more
- Injection Retardation: By injecting fuel later in the compression cycle of the piston, the fuel has less time to burn, creating less NOx. However, it still leaves the other byproducts. This is usually combined with higher injection pressures, which cause finer fuel droplets, which burn more quickly, leaving the opposite problem of too much NOx. Despite this, there is a measurable drop in both levels when compared to unmodified engines.
- Charge Air Cooling: The use of a cooling agent to cool the air before it enters the piston, lowering the burn temperature, and creating less NOx.
- Catalytic Converter: Due to the cost of materials, this is exclusively for small land based units and automobiles. It utilizes rhodium as a catalyst to change nitrogen oxides back to nitrogen gas and water.
- Direct Water Injection (Wartsila): The key element in the DWI system is the combined injection nozzle which has one needle valve for the fuel and another one for the fresh water. Water to fuel
Injection nozzle injects both water and fuel in the ratio 0.4 to 0.7, coating droplets of water with fuel. This increases atomization of the fuel, and creates a low temperature combustion which reduces NOx emissions by 50%-60%, with unaffected or slightly improved specific fuel consumption. - Selective Catalytic Reduction System (Wartsila): By spraying the exhaust gases with a mist of ammonia, and passing it through a catalyst, the ammonia and NOx react, and change to nitrogen gas and water. 85%-95% emissions reductions are possible. This equipment can be retrofitted; however it is bulky and has to be fitted before the exhaust boiler.
- Exhaust gas recirculation
50 to 60% reduction of NOx is possible for 15% recirculation of exhaust gases into the inlet manifold. The exhaust gases being recirculated are cleaned and cooled before recirculation to the scavenge air side. It reduces NOx by lowering the oxygen concentration in combustion zone. - Fuel water emulsion
Adding water to the fuel dramatically sinks both soot emissions and nitrogen oxide emissions from diesel engines. This equipment can be retrofitted on all diesel engines, regardless of whether they are pre-chamber/swirl chamber engines or unit injector/common rail direct injection engines. The emulsion is produced immediately before the injection pump; no intervention in the engine or the direct injection system is required.
The MC-90 Ships model SCR Plant Description
This ship is fitted with two options for reducing NOx
1.Selective Catalytic Reduction unit which treats the exhaust gases before they enter the turbocharger. Pre-programmed quantity of Ammonia is added to the exhaust gas stream, and the mixture passed through a catalyst at a temperature between 300°C and 400°C. Within the SCR Reactor the hot exhaust gases containing NOx gases are mixed with the ammonia stream. This reduces the NOx to N2 and H2O.
If the temperature of reaction is too high (above 490°C), the ammonia burns and does not react, and at low temperatures (below 250°C) the reaction rate is low and the catalyst can be damaged therefore the SCR is not used for lower engine RPMs
2. Fuel water emulsion situated between the fuel oil meter and the venting box is the Fuel-Water Emulsion Control Unit which is designed for emulsification of the fuel to reduce the NOx values in the exhaust gas from the engines.
SULPHUR OXIDES
Sulphur oxides (SOx) are the generic term for Sulphur dioxide (SO2) and sulphur trioxide (SO3). The sulfur oxides emission in the Diesel Engine exhaust gases is due to the presence of Sulphur in the marine fuels used on Ships. Better the grade of the fuel the lower will be the sulfur content as it is removed by refining of the fuel. All sulphur entering the engine combustion chamber is oxidized forming SOx, which is emitted to the atmosphere with the exhaust gases. The sulphur absorbed by the alkaline cylinder lubricant being negligible the SOx emissions from the engine are directly proportional to the fuel sulphur content and fuel consumption.
For regulatory purposes the sulphur oxides concentration in the Engine exhaust gases are generally calculated from the sulphur content of the fuel, as its physical measurement is challenging and expensive.
Environmental Impact
• Acid rain.
• Contributes to formation of small size particulates as part of SOx forms SO3 and sulphates
which condense to particulates
• Contributes to detrimental effect on human health, vegetation, and buildings
Methods for Reducing SOx Emissions
- Using low-sulphur fuel (or LNG and other cleaner fuels)
- Installing Exhaust gas Cleaning (EGC) systems which include closed-loop, open-loop, and hybrid systems.
Exhaust Gas Cleaning systems:
Based on the natural alkalinity of seawater,the open-loop system uses sea water to scrub and neutralize the SO2 in the exhaust gases in the scrubber tower. After scrubbing, this water is discharged into the sea, however, this can cause secondary pollution and contribute to the greenhouse effect due to the release of carbon dioxide. Furthermore, it has high energy consumption.
The closed-loop system uses fresh water or seawater as the scrubbing liquid dosed with caustic soda (NaOH) as the absorbent to remove SO2 in exhaust gases. This system removes 95% of SOx has low energy consumption, and eliminates CO2 emissions as by product. The scrubbing water with dissolved SOx is sent to a water treatment effluent emulsion breaking plant after which it can be discharged overboard or pumped ashore .
- Compatibility with waste heat recovery units and SCR systems
All wet SOx scrubbers significantly cool the exhaust gas. Therefore, they are not suitable for installation before a waste heat recovery unit. For the same reason, it would not be possible to install a wet SOx scrubber before an SCR system unless a reheater was fitted after the wet scrubber to raise the exhaust gas temperature back up to around 300oC
Limits for SOx Emission from a Merchant Vessel
Outside the emission controlled area, the sulphur content of any oil used on board ship must not exceed 3.5% m/m.
When the ship is within emission controlled area, the sulphur content of any fuel oil used on board ships must not exceed 0.1% m/m., alternatively the ship should be fitted with an exhaust gas cleaning system or any other technological process to reduce the sulphur emission from ships including auxiliary and main propulsion engine to 6.0g SOx/kWh or less
Lab Instructions
Press F6 and from right hand view box select ” Initial condition”
Select Box 10 – “ready to start” exercise. Ensure it is CPP model
Suggested pen recorder variables that need to be plotted and monitored are:
Z00518 – g/kwhr ME exh SOx content
E03760 – Propeller power output in MW
N02015 – ME RPM
Z02013 – ME exhaust gas smoke content
N06312 – Ships speed in Knots
Z01970 – g/kwhr ME exh NOx content final
Tabulate your data as shown below: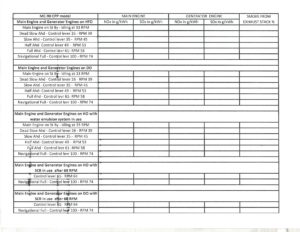
Deliverables
Your lab report is to include the following:
- Trend plots: Supply all plots taken for this lab (make sure plots are labeled properly)
- Data table : with all findings entered
- Conclusion: Write a summary (max. 500 words in a text box, if using Excel) comparing your results and suggestions for further study.
Further Reading:

