Describing and Naming Chemical and Biochemical Sedimentary Rocks
Goals
By the end of the chemical/biochemical rock section you will be able to accomplish the following:
1. Determine if a carbonate rock is comprised of dolomite or calcite.
2. Identify typical fragments contained within carbonate rocks.
3. Identify the nature of the cement that binds fragments together.
4. Name a carbonate rock based on mineralogy, the clasts it contains, and type of cement.
5. Identify and name other chemical and biochemical sedimentary rocks.
Chemical Sedimentary Rocks: Carbonates vs. The Others
The classification of chemical and biochemical rocks starts with identifying the dominant mineral. This identification is made easier because carbonates formed of calcite or dolomite will react to dilute hydrochloric acid (HCl) by “fizzing” when exposed to a drop of acid. This makes the first major distinction between carbonates and non-carbonates, or other chemical/biochemical sedimentary rocks.
How to Perform an Acid Test Using Dilute HCl
Equipment: Dilute HCl in a dropper bottle, a porcelain plate, and paper towels.
Safety note: Geoscientists use dilute HCl as a means of testing for the presence of calcite or dolomite in a rock. This is a solution of 10% HCl carried in a small plastic dropper bottle. Handle the acid with care and avoid getting it on your skin or clothing. Wash your hands after use. Acid and carbonate will neutralize to be harmless, but if a rock does not react with the acid it will still be on the surface. Rinse the acid off with water. If in doubt, ask for assistance.
Scrape the rock sample on the porcelain plate so that some rock dust is left behind. Watching closely, place one drop of acid on the rock dust. Look for bubbles of carbon dioxide that are formed when carbonate and acid react. You can also try putting a drop on the fresh face of rock created by scraping on the porcelain plate. The vigorousness of the fizz will tell you if the sample is dolomite (low fizz) or calcite (high fizz). If the dust does not cause a reaction, it is not a carbonate. Wipe the plate clean with the paper towel or rinse with water.
Non-Carbonate Chemical and Biochemical Rocks
These chemical sedimentary rocks do not react to a dilute acid test. We will cover the “Other” category of chemical and biochemical rocks first because these are fairly simple to describe and name. The “Other” category is based entirely on mineralogy. Few descriptive terms are needed to narrow down to a rock name as mineralogy is the dominant feature.
A non-carbonate chemical or biochemical rock description should include:
Colour, Name, Other Features
These rocks are named following Table 1.5. An example descriptive name is:
Dull black coal with occasional plant fossils
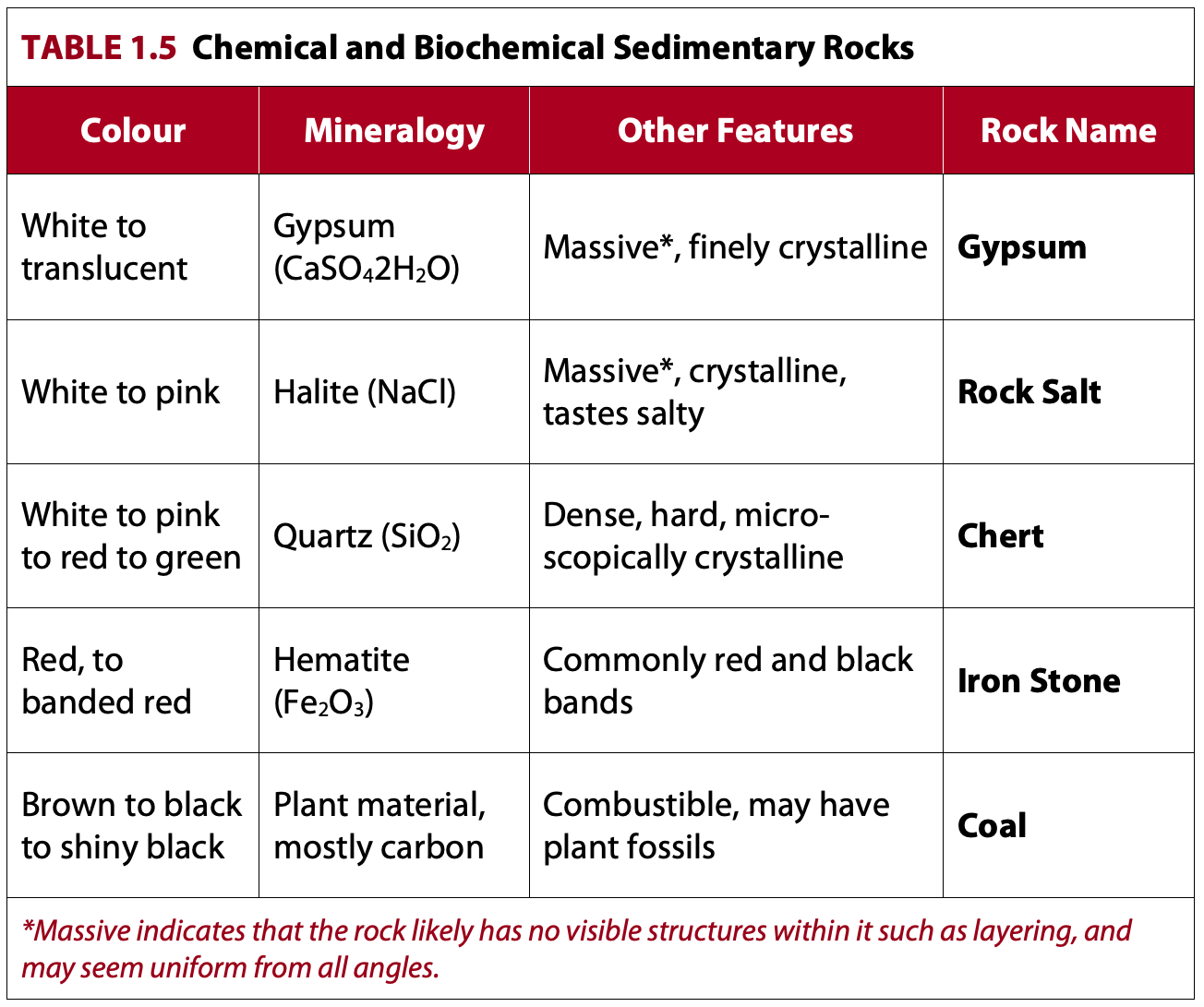
Evaporites
Gypsum and halite are often formed in environments where sea water is being evaporated. These two minerals (as well as other minerals formed this way) are therefore sometimes called evaporites, which is a term we will see in later labs.
Carbonate Chemical and Biochemical Rocks
Those rocks that do react with the HCl acid are called carbonates. Carbonate sediments and carbonate rocks are the most abundant form of chemical/biochemical sediments, comprising an estimated 20-25% of all sedimentary rock.
Carbonates rocks have two main components: a finer-grained matrix; and larger grains. For our purposes, the matrix is the finer grained material that makes up the “background” part of the carbonate. The matrix has no individual features identifiable by eye only (i.e., with no magnifiers or microscopes) because the material is too fine-grained. In carbonates, grains are objects that are large enough to be visible to the eye that are found within the otherwise finer-grained calcareous matrix.
You will use a simple flow chart (Figure 1.4) to describe and name carbonate rocks which will include:
Colour, Name, Other Features
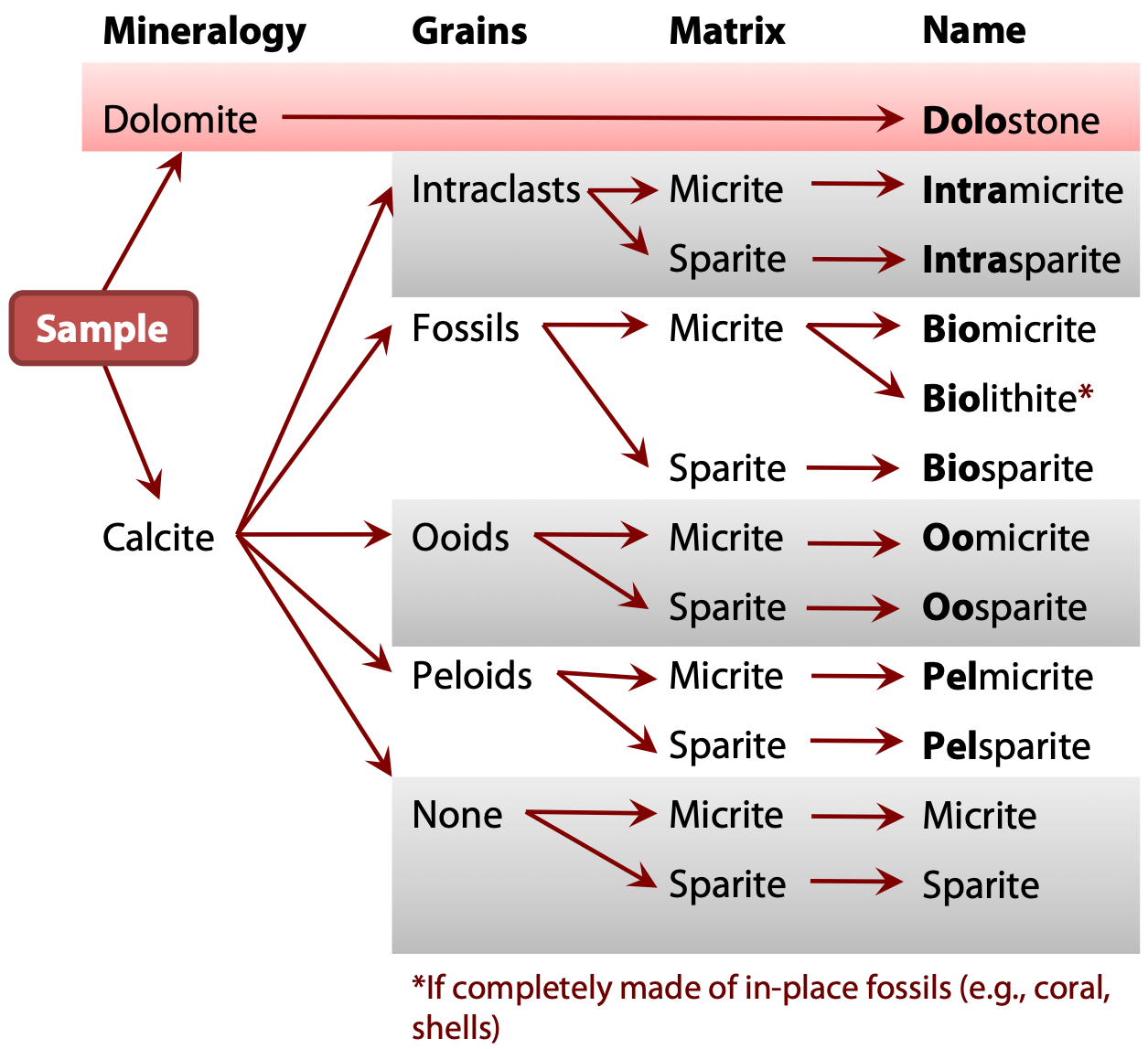
Step 1 – Mineralogy
The primary mineral in the finer-grained matrix of a carbonate rock is first identified based on a dilute acid test. The carbonate rock is thus identified as either a limestone or a dolomite:
Calcite (CaCO3) is highly reactive to HCl. It will likely bubble when acid is place on the rock, and will bubble vigorously when powdered. Carbonates in which the dominant mineral is calcite are called limestones.
Dolomite (CaMg(CO3)2) is slightly reactive to HCl. It may not bubble at all when acid is placed on the rock, but will react more readily to HCl when powdered. It is the dominant mineral in dolomites.
Other minerals may also be present in carbonates in smaller amounts.
If you identify the matrix as dolomite: Classifying carbonate rocks in which the dominant mineral is dolomite is not covered in this course. For this course, all dolomites are considered to be dolostones. If your sample is a dolostone, you have come to the end of the flow chart.
If you identify the matrix as calcite: Carry on with Steps 2 and 3.
Step 2 – Grain Type
Visible grains may or may not be present. The size of carbonate grains is less important than what the grains are composed of or their form. Four forms are possible:
Intraclasts (Carbonate Clasts) – Fragments of previously lithified carbonate rocks that are included in a younger matrix. This is analogous to a clastic conglomerate or breccia in which clastic rock fragments are suspended in a matrix of finer clasts. Prefix: Intra-
Fossil Skeletal Particles – Fragments of previously living organisms and/or their shells. The type of organism contained in the carbonate will not affect the name, and in this lab, do not try to identify the fossils. Fossils can give important insights into the age of the rock and the environment in which it formed. Generally, the fossil type should be included in the final rock description under other features, and we will do this in later labs. Prefix: Bio-
Ooids – Ooids are smooth, usually moderate- to high-sphericity, visible grains formed when a small carbonate grain is rolled back and forth on the sediment surface in an environment in which carbonates are forming. New carbonate is gradually added to the outer surface of the ooid creating finely layered spheres that have a tree-ring like appearance when cut in half. These spherical nodules may appear at first glance to be similar to some skeletal materials. Prefix: Oo-
Peloids – These are small particles, generally silt-sized, which are most often fecal pellets produced by small marine organisms that ingest calcareous material while feeding. Other forms of peloids are possible but they are beyond the scope of this course. Prefix: Pel-
None – Grains are not present in all carbonate rocks. They may be composed entirely of cementing material.
This classification system is very simplistic. It is possible for a carbonate to be composed of all larger carbonate grains, a mixture of grains and fine grained cement, or to be completely composed of fine cement. For the purposes of this course, however, this classification is appropriate. It is based on the classification proposed by R.L. Folk (1962) and from the text Principles of Sedimentology and Stratigraphy (4th Edition, S. Boggs Jr.).
Step 3 – Matrix
The matrix is the fine grained material that hosts the carbonate grains. Remember: sediments undergo lithification to become a sedimentary rock. (Click here to review lithification). Cementation or re-crystallization during lithification can transform the fine grained matrix into larger crystals of calcite that are visible to the eye. The matrix can therefore be either very fine-grained and mud-like, or a generally coarser grained/crystalline calcite. Both can be present between the grains of a carbonate, but the rock name is based upon which form is dominant.
Micrite is a fine-grained calcareous material which can either act as the cement for carbonate clasts or may comprise the entire rock. Often micrite will contain impurities which results in an off-white or grey colour. The word micrite is a shortened form of “microcrystalline calcite.”
Sparite is comprised of coarse crystals of calcite generally near 1 mm in size. These crystals are distinguishable from micrite due to their size and clarity (the crystals are typically clear).
With mineralogy, grain form and cement identified, the name of the carbonate can be found using Figure 1.4. A full description of the carbonate rock also includes colour and other features.
Other features is a descriptor is used to include other characteristics that may be useful for distinguishing one carbonate rock from another similar rock, or features that help provide further clues as to age, or depositional environment of the rock. For example, two limestone of different origin might be identical in other parts of the description, but the presence of “oyster shell fragments” in one and not in the other can be used to tell them apart.
“Other features” might include the following:
- Organic matter or fossils. Any organic matter is described with its colour, nature (type) and distinguishing properties. You will learn how to describe fossils fully using proper geological terms in Lab 5. In this lab, use everyday language to describe what you see.
- Special features or unusual minerals. The presence of some minerals in a rock can give important clues as to its origins, and hence may be noted at the end. The presence of rusty-coloured iron oxide minerals is an important clue that the sediment deposited in the presence of oxygen and thus on land. The presence of pyrite may indicate that the sediment was deposited in low oxygen conditions in water. We will be using this in Lab 2.
For example:
A tan-coloured rock reacts vigorously to HCl. It is seen to have a fine matrix (cement), with no visible shiny crystal faces. There are many smooth round spheres contained within the matrix that are 1-2 mm in diameter, and there are occasional shell fragments of things that look a bit like a modern day clam.
Descriptive Name:
Tan oomicrite with ooids 1-2 mm in diameter and sparse skeletal or fossil fragments (clam like)
