11 Definitions of Reaction Rate and Extent of Reactions
Learning Objectives
By the end of this section, you should be able to:
Define Reaction rate and reaction extent
Calculate the reaction rate for the reaction, the rate of formation of compounds and the reaction extent
Reaction Rate
A reaction rate shows the rates of production of a chemical species. It can also show the rate of consumption of a species; for example, a reactant. In general though, we want an overall common reaction rate to describe changes in a chemical system.
Let's look at an example. Say we have reaction represented as: [latex]A + 2B → 3C + D[/latex]
For this system reaction rate can be expressed as follows:
[latex]r = \frac{d[D]}{dt}[/latex]
The reaction rate is represented by the letter "r" or the Greek letter upsilon "[latex]\upsilon[/latex]"
NOTE: I will stick with r as upsilon looks like "[latex]\nu[/latex]" which we will use to represent the stoichiometric coefficient
Above we have written the reaction rate as if a substance with a coefficient of 1 was reacting (or being produced). This is the typical form of an overall reaction rate describing a reaction.
Rearranging the above equation, we can find the rate of production/consumption for any species based on this overall reaction rate, note that stoichiometric coefficients are positive for products and negative for reactants:
[latex]r_{A}=\frac{d[A]}{dt}=-\nu_{A}×r=(-1)×r[/latex]
[latex]r_{B}=\frac{d[B]}{dt}=-\nu_{B}×r=(-2)×r[/latex]
[latex]r_{C}=\frac{d[C]}{dt}=\nu_{C}×r=3×r[/latex]
[latex]r_{D}=\frac{d[D]}{dt}=\nu_{D}×r=1×r[/latex]
The general equation for calculating the reaction rate:
General notation: J is used to denote any compound involved in the reaction.
| [latex]r = \frac{1}{\nu}\frac{d[J]}{dt}[/latex] |
Reaction rate at a given time can also be found from the graph of concentration of components in a system vs. time:
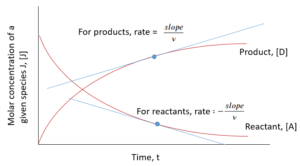
Exercise: Calculating the Reaction Rate
If we have the reaction
[latex]2 NOBr_{(g)} ⇌ 2 NO_{(g)} + Br_{2(g)}[/latex]
and we measure that the rate of formation of NO is 1.6 mmol/(L·s), what are the overall reaction rate, and the rate of formation of [latex]Br_{2}[/latex] and [latex]N\!O\!Br[/latex]?
Solution
Step 1: Determine the overall reaction rate from the rate of formation for NO.
\begin{align*}
r & = \frac{1}{\nu_{j}} \frac{d[NO]}{dt} \\
& = \frac{1}{2} \left( 1.6 \frac{mmol}{L·s}\right)\\
& = 0.8\frac{mmol}{L·s}
\end{align*}
Step 2: Use the reaction rate to determine rate of formation for the other compounds
NOTE: rate of formation is positive for products and negative for reactants
\begin{align*}
\frac{d[Br_{2}]}{dt}& = r \\
& = 0.8\frac{mmol}{L·s}
\end{align*}
\begin{align*}
\frac{d[NOBr]}{dt}& = -2r \\
& = -1.6\frac{mmol}{L·s}
\end{align*}
Reaction rates can be given in a variety of units over time. In this class we will just explore molarity and partial pressure, although other forms exist.
Molarity - molar concentration - expressed in units of [latex]\frac{mol}{volume * time}[/latex] (eg. [latex]\frac{mol}{L*s}[/latex] )
Partial pressure - the pressure produced by one gaseous component if it occupies the whole system volume at the same temperature, commonly used for gasses - units of [latex]\frac{pressure}{time}[/latex]( eg. [latex]\frac{Pa}{s}[/latex] )
Extent of Reaction
We use the extent of reaction ([latex]\xi[/latex]) to describe the change in an amount of a reacting speicies J.
| [latex]d n_{j} = \nu_{j} d\xi[/latex] |
where:
- [latex]dn_{j}[/latex] = change in the number of moles of a certain substance
- [latex]\nu_{j}[/latex] = the stoichiometric coefficient
- [latex]d\xi[/latex] = the extent of reaction
We can get a relationship between the reaction extent and the rate of reaction when the system volume is constant:
| [latex]r = \frac{1}{V} \frac{d\xi}{dt} = \frac{1}{\nu_{j}} \frac{1}{V} \frac{dn_{j}}{dt}[/latex] |
where:
[latex]V[/latex]= volume


Feedback/Errata