19 Kinetic & Thermodynamic Control
Learning Objectives
By the end of this section, you should be able to:
Understand kinetic and thermodynamic control
Reactants can sometimes give rise to a variety of products.
Consider the nitration of nitrobenzene:
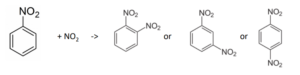
The relative portion of the products before reaching equilibrium is given by the ratio of the rates of production.
[latex]A+B→P_{1}[/latex] where [latex]r_{P1}=k_{r1}[A][B][/latex]
[latex]A+B→P_{2}[/latex] where [latex]r_{P2}=k_{r2}[A][B][/latex]
Here, before equilibrium:
| [latex]\frac{[P_{2}]}{[P_{1}]}=\frac{k_{r,2}}{k_{r,1}}[/latex] |
This is called kinetic control, and it is dictated by reaction rates.
As opposed to thermodynamic control, which is dictated by reaction equilibrium (after a long time):
Say we have the system:
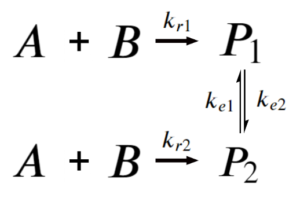
If [latex]k_{e1},k_{e2}\text{<<}k_{r1},k_{r2}[/latex]
Then at any time before the equilibrium reaction start severely affecting product concentration, the reaction simplifies to:
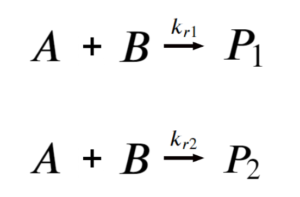
| [latex]\frac{[P_{1}]}{[P_{2}]}=\frac{k_{r1}}{k_{r2}}[/latex] |
The reaction is kinetically controlled: the amount of products depends on the rates of reaction.
Proof to show: [latex]\frac{[P_{1}]}{[P_{2}]}=\frac{k_{r1}}{k_{r2}}[/latex]
\begin{align*}
r_{P1}=k_{r1}[A][B]&=\frac{d[P1]}{dt}\\
r_{P2}=k_{r2}[A][B]&=\frac{d[P2]}{dt}
\end{align*}
Say that both [latex]P_{1}[/latex], [latex]P_{2}[/latex] start at a concentration of 0. We can express the change in concentration for [latex]P_{1}[/latex] and [latex]P_{2}[/latex] at any time before [A][B] reaches 0. Note that once [A][B] reaches 0, the equilibrium reaction starts to dominate as we no longer have forward reactions that consume A and B to produce P1 and P2.
[latex]\frac{[P1]}{[P2]}=\frac{\frac{d[P_{1}]}{dt}}{\frac{d[P_{2}]}{dt}}=\frac{k_{r1}[A][B]}{k_{r2}[A][B]}=\frac{k_{r1}}{k_{r2}}[/latex]
If [latex]k_{e1},k_{e2}>>k_{r1},k_{r2}[/latex]
Then at any given time, this reaction simplifies to:
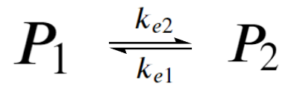
| [latex]\frac{[P_{1}]}{[P_{2}]}=\frac{k_{e1}}{k_{e2}}[/latex] |
The reaction is thermodynamically controlled : the amount of products depends on the equilibrium state.
Proof to show: [latex]\frac{[P_{1}]}{[P_{2}]}=\frac{k_{e1}}{k_{e2}}[/latex]:
At equilibrium:
Forward reaction rate: [latex]\frac{d[P2]}{dt}=k_{e2}[P1][/latex]
Reverse reaction rate: [latex]\frac{d[P1]}{dt}=k_{e1}[P2][/latex]
At equilibrium, the forward and reverse reaction rates are equal:
\begin{align*}
k_{e2}[P1] & = k_{e1}[P2]\\
\frac{[P_{1}]}{[P_{2}]} & = \frac{k_{e1}}{k_{e2}}
\end{align*}


Feedback/Errata