36 Reactive Energy Balances
Learning Objectives
By the end of this section, you should be able to:
Explain heats of reaction as well as endothermic and exothermic reactions
Determine the standard heat of reaction given other heats of reaction or heats of formation (Hess's Law)
Analyze energy balances involving reactive systems
What happens in reactions in terms of chemical bonds?
Bonds can be formed and broken. Breaking bonds takes energy and forming bonds releases energy.
- If more energy is released in forming bonds than absorbed in breaking bonds, then the reaction is exothermic.
- If more energy is absorbed in breaking bonds than released in forming bonds, then the reaction is endothermic.
Example: Chemical Bonds
Consider the simple reaction of the formation of water from hydrogen and oxygen:
[latex]2H_{2} + O_{2} → 2H_{2}O[/latex]
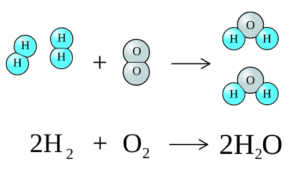
Image from すじにくシチュー / CC0
The formation of bonds takes energy while the breakage of bonds releases energy
In this reaction:
-
2 H-H bonds are broken
-
1 O-O bonds are broken
-
4 H-O bonds are formed
Heat of Reaction [latex]\Delta H_{r}(T,P)[/latex]
Heat of Reaction [latex]\Delta H_{r}(T,P)[/latex]: the stoichiometric enthalpy difference when reactants react completely to form products at a specified constant temperature and pressure.
[latex]\Delta H_{r}(T,P) = H_{products} - H_{reactants}[/latex]
In exothermic reactions, [latex]\Delta H_{r}(T,P)<0[/latex]
- [latex]H_{products} < H_{reactants}[/latex]
In endothermic reactions, [latex]\Delta H_{r}(T,P)>0[/latex]
- [latex]H_{products} > H_{reactants}[/latex]
Heats of reaction are directly proportional to the amount of reactants or products in a reaction.
Example: Formation of Nitrogen
Consider the formation of nitrogen dioxide
[latex]\frac{1}{2} N_{2} (g) + O_{2}(g) → NO_{2} (g)[/latex]
[latex]\Delta H_{r} = 33.2 kJ[/latex]
The formation of 1 mole of [latex]NO_{2}[/latex] yields an enthalpy change of 33.2 kJ
[latex]N_{2} (g) + 2O_{2}(g) → 2NO_{2} (g)[/latex]
[latex]\Delta H_{r} = 66.4 kJ[/latex]
The formation of 2 moles of [latex]NO_{2}[/latex] yields an enthalpy change of 66.2 kJ
Heat of reactions for a reaction in the forward direction is *equal to the negative heat of reaction* for the backward reaction.
Consider the formation of hydrogen chloride (or the corresponding decomposition):
[latex]H_{2} (g) + Cl_{2} (g) → 2 HCl (g)[/latex]
[latex]\Delta H_{r} = -184.6 kJ[/latex]
[latex]2HCl(g) → H_{2} (g) + Cl_{2} (g)[/latex]
[latex]\Delta H_{r} = +184.6 kJ[/latex]
Exercise: Heat of Reaction
Consider the oxidation of ammonia taking place in an isothermal and constant pressure reactor:
[latex]4 NH_{3}(g) + 5 O_{2}(g) → 4 NO(g) + 6 H_{2}O(v)[/latex]
where the heat of reaction is [latex]\Delta\hat{H}^{\circ}_{r} = -904.7 kJ[/latex]
The ammonia is fed into the reactor at [latex]100 mol/s[/latex] and the oxygen enters at [latex]200 mol/s[/latex]. Assuming the limiting reactant is completely consumed, what is the enthalpy change for this reaction?
Solution
Step 1: Find the limiting reactant by finding the smallest reactant extent if all of a reactant is consumed.
[latex]\text{if } N\!H_{3} \text{ is limiting: } \xi=\frac{100mol/s}{4}=25\frac{mol}{s}[/latex]
[latex]\text{if } O_{2} \text{ is limiting: } \xi=\frac{200mol/s}{5}=40\frac{mol}{s}[/latex]
[latex]N\!H_{3}[/latex] being the limiting reagent yields the smaller reaction extent, therefore [latex]N\!H_{3}[/latex] is the limiting reagent. The reaction extent is [latex]25\frac{mol}{s}[/latex].
Step 2: Multiply the reaction extent by the [latex]\Delta\hat{H}^{\circ}_{r} = -904.7 \frac{kJ}{molNH_{3}}[/latex] to obtain [latex]\Delta \dot{H}_{r}[/latex]
[latex]\Delta \dot{H}_{r}=\xi*\Delta\hat{H}^{\circ}_{r}[/latex]
[latex]\Delta \dot{H}_{r}=25\frac{mol}{s}*-904.7\frac{kJ}{mol}[/latex]
[latex]\Delta \dot{H}_{r}=-22620 \frac{kJ}{s}[/latex]
Hess's Law
Some reactions may be difficult to reproduce in a laboratory setting. Instead, for analyzing a certain overall reaction, we may break the reaction down in multiple steps:
For example, say we want to find the standard heat of reaction for the following hypothetical reaction:
[latex]A + 0.5 B → C[/latex]
If this reaction is difficult to carry out in a laboratory, we might break it up into 2 reactions, that are easier to carry out and where we can determine specified standard heats of reaction:
Reaction 1: [latex]A + B → D[/latex] with [latex]\Delta H^{\circ}_{rxn1}[/latex]
Reaction 2: [latex]C + 0.5 B → D[/latex] with [latex]\Delta H^{\circ}_{rxn2}[/latex]
We can combine reactions 1 and 2 to obtain our desired reaction by subtracting reaction 2 from reaction 1:
[latex]Reaction 1 - Reaction 2[/latex]
[latex](A + B) - (C + 0.5 B) → D - D[/latex]
[latex]A + 0.5 B - C →[/latex]
We obtain our desired reaction:
[latex]A + 0.5 B → C[/latex]
Therefore, the heat of reaction of the desired reaction will be:
[latex]\Delta H^{\circ} = \Delta H^{\circ}_{rxn1} - \Delta H^{\circ}_{rxn2}[/latex]
Exercise: Hess's Law
Consider the formation of chlorine trifluoride from chlorine monofluoride and fluorine:
[latex]ClF(g) + F_{2}(g) → ClF_{3}(g)[/latex]
[latex]\Delta H^{\circ} = ?[/latex]
Data on the following reactions are available:
| Number | Reaction | [latex]\Delta H^{\circ}[/latex] (kJ) |
|---|---|---|
| 1 | [latex]2OF_{2}(g) → O_{2}(g) + 2F_{2}(g)[/latex] | -49.4 |
| 2 | [latex]2ClF(g) + O_{2}(g) → Cl_{2}O(g) + OF_{2}(g)[/latex] | 214.0 |
| 3 | [latex]ClF_{3}(g) + O_{2}(g) → \frac{1}{2} Cl_{2}O(g) + \frac{3}{2}OF_{2}(g)[/latex] | 236.2 |
What is the [latex]\Delta H^{\circ}[/latex] for the desired reaction?
Solution
Step 1: Determine what reaction will give us [latex]ClF[/latex] in the reactants.
Since [latex]ClF(g)[/latex] is needed as a reactant, we can multiply reaction 2 by [latex]\frac{1}{2}[/latex] to obtain:
[latex]ClF(g) + \frac{1}{2}O_{2}(g) → \frac{1}{2}Cl_{2}O(g) + \frac{1}{2}OF_{2}(g)[/latex]
[latex]\Delta H^{\circ} = \frac{1}{2}(214.0 kJ) = 107.0 kJ[/latex]
Step 2: Determine what reaction will give us [latex]F_{2}[/latex] in the reactants.
Since [latex]F_{2}[/latex] is needed as a reactant, we can multiply reaction 1 by [latex]-\frac{1}{2}[/latex] to obtain:
[latex]F_{2}(g) + \frac{1}{2}O_{2}(g) → OF_{2}(g)[/latex]
[latex]\Delta H^{\circ} = -\frac{1}{2}(-49.4 kJ) = 24.7 kJ[/latex]
Step 3: Determine what reaction will give us [latex]ClF_{3}[/latex] as a product.
Since [latex]ClF_{3}[/latex] is needed as a product, we can multiply reaction 3 by -1 to obtain:
[latex]\frac{1}{2} Cl_{2}O(g) + \frac{3}{2}OF_{2}(g) → ClF_{3}(g) + O_{2}(g)[/latex]
[latex]\Delta H^{\circ} = -(236.2kJ) = -236.2 kJ[/latex]
Step 4: Add the reactions up to ensure that the desired reaction is obtained:
[latex]ClF(g) + \frac{1}{2}O_{2}(g) → \frac{1}{2}Cl_{2}O(g) + \frac{1}{2}OF_{2}(g)[/latex]
[latex]F_{2}(g) + \frac{1}{2}O_{2}(g) → OF_{2}(g)[/latex]
[latex]\frac{1}{2} Cl_{2}O(g) + \frac{3}{2}OF_{2}(g) → ClF_{3}(g) + O_{2}(g)[/latex]
[latex]↓(+)[/latex]
[latex]ClF(g) + F_{2}(g) → ClF_{3}(g)[/latex]
Step 5: Add up all the manipulated heat of reactions to obtain the desired [latex]\Delta H^{\circ}[/latex]
[latex]\Delta H^{\circ} = (107.0 + 24.7 + -236.2)kJ[/latex]
[latex]\Delta H^{\circ} = -104.5 kJ[/latex]
Heat of Reaction Method for Analyzing Energy Use in a Process
Now let's try applying the heat of reaction to determine energy use in a process, where reactants and products are coming in and exiting at a given temperature. The following process path is taken for the heat of reaction method, where the reference state is at [latex]25^{\circ}C[/latex]:
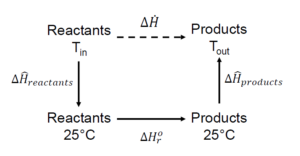
Generally, [latex]\Delta H ^{\circ}_{r}[/latex] refers to standard state conditions at [latex]25^{\circ}C[/latex] and 1 atm, but always double-check whether a different standard state condition is used.
In this process path, the enthalpy calculations are done in three steps:
- Temperature change: calculate the [latex]\Delta\hat{H}_{reactants}[/latex] using the heat capacities. The temperature change will be the difference between the inlet temperature of the reactants and the standard or reference temperature ([latex]25^{\circ}C[/latex] in this case).
- Reaction enthalpy: calculate the enthalpy of the reaction [latex]\Delta H^{\circ}_{r}[/latex]. This can be done using Hess's Law or the Heat of Formation method (discussed below).
- Temperature change: calculate the [latex]\Delta\hat{H}_{products}[/latex] using the heat capacities. The temperature change will be the difference between the outlet temperature of the products and the standard or reference temperature ([latex]25^{\circ}C[/latex] in this case).
The calculated enthalpy changes for each step are then added to obtain [latex]\Delta\dot{H}[/latex].
Example: Heat of Reaction Method for Energy Use
Consider a propane combustion chamber, where [latex]100 mol/s[/latex] of propane are feed to the chamber at [latex]25^{\circ}C[/latex] and air is fed at [latex]300^{\circ}C[/latex] ([latex]600 mol/s O_{2}(g)[/latex] and [latex]2256 mol/s N_{2}(g)[/latex]). The products stream exits at [latex]1000^{\circ}C[/latex] and consists of [latex]100 mol/s O_{2}(g)[/latex], [latex]2256 mol/s N_{2}(g)[/latex], [latex]300 mol/s CO_{2}(g)[/latex], and [latex]400 mol/s H_{2}O(v)[/latex]. How much heatis released by this combustion chamber? Assumming atmoshpere pressure, so water boils at 100°C.
[latex]C_{3}H_{8} (g) + 5 O_{2} → 3 CO_{2} (g) + 4 H_{2}O (l)[/latex]
[latex]\Delta H^{\circ}_{r} = -2220 kJ[/latex]
The following information is provided:
The values listed under each compound are specific enthalpies in kJ/mol
| Temperature ([latex]^{\circ}C[/latex]) | [latex]O_{2}[/latex] | [latex]N_{2}[/latex] | [latex]CO_{2}[/latex] | [latex]H_{2}O[/latex] |
| 25 | 0 | 0 | 0 | 0 |
| 100 | 2.24 | 2.19 | 2.90 | 2.54 |
| 300 | 8.47 | 8.12 | 11.58 | 9.57 |
| 1000 | 32.47 | 30.56 | 48.60 | 37.69 |
- [latex]C_{P,l(H_{2}O)} = 75.4*10^{-3} \frac{kJ}{molK}[/latex]
- [latex]C_{P,v(H_{2}O)} = 33.46*10^{-3}+0.688*10^{-5}*T + 0.7604*10^{-8}*T^{2} - 3.593*10^{-12}*T^{3} (J/molK)[/latex]
- [latex]\Delta\hat{H}_{vap}(100^{\circ}C) = 40.66 kJ/mol[/latex]
How can we solve the system's energy balance?
1 - Solve material balances as much as possible
This example already has the material balances solved
2 - Choose reference states for energy calculations. Like we say in the previous figure we want to calculate the changes in energy associated with bringing our reactants to our reference state, calculating the energy of the reaction at the reference state, and then the energy associated with bringing the products to their final state. We will choose reference states based on the information we have to try to make these calculations as easy as possible.
For reacting species:
Since [latex]\Delta H^{\circ}_{r}[/latex] is given, we will assume this is at [latex]25^{\circ}C[/latex] and 1 atm (as there is nothing indicating otherwise), and we will use this as our reference state.For non-reacting species:
We can use any convenient temperature and pressure as a reference state (inlet temperature, outlet temperature, temperature in enthalpy table). With this example, the enthalpy table given uses a reference state of [latex]25^{\circ}C[/latex], and we will assume everything is at 1 atm (as no other values for pressure are given).
3 - Calculate the extent of reaction for all reactions (in this case we just have the one reaction)
[latex]\xi = \frac{(\dot{n}_{i})_{out}-(\dot{n}_{i})_{in}}{\dot{\nu}_{i}}[/latex]
[latex]\xi = \frac{(\dot{n}_{CO_{2}})_{out}-(\dot{n}_{CO_{2}})_{in}}{\dot{\nu}_{CO_{2}}}[/latex]
[latex]\xi = \frac{300\frac{mol}{s} - 0 \frac{mol}{s}}{3} = 100 mol/s[/latex]
4 - Prepare an inlet-out enthalpy table (this will show what enthalpies we need to calculate associated with energy changes in the reactants or products)
| Substance | [latex]\dot{n}_{in}[/latex](mol/s) | [latex]\hat{H}_{in}[/latex] (kJ/mol) | [latex]\dot{n}_{out}[/latex] (mol/s) | [latex]\hat{H}_{out}[/latex] (kJ/mol) |
| [latex]C_{3}H_{8}[/latex] | 100 | [latex]\hat{H}_{1}[/latex] | - | - |
| [latex]O_{2}[/latex] | 600 | [latex]\hat{H}_{2}[/latex] | 100 | [latex]\hat{H}_{4}[/latex] |
| [latex]N_{2}[/latex] | 2256 | [latex]\hat{H}_{3}[/latex] | 2256 | [latex]\hat{H}_{5}[/latex] |
| [latex]CO_{2}[/latex] | - | - | 300 | [latex]\hat{H}_{6}[/latex] |
| [latex]H_{2}O[/latex] | - | - | 400 | [latex]\hat{H}_{7}[/latex] |
5 - Calculate all the enthalpies. For [latex]\hat{H_{1}}[/latex] to [latex]\hat{H_{6}}[/latex], the specific enthalpies at different temperatures are given as the difference in enthalpy from the reference state (25°C, 1atm, which is the same as what we choose as reference state):
[latex]\hat{H}_{1}=\Delta\hat{H}_{C_{3}H_{8}} (25^{\circ}C → 25^{\circ}C) = 0 kJ/mol[/latex]
[latex]\hat{H}_{2}=\Delta\hat{H}_{O_{2}} (25^{\circ}C → 300^{\circ}C) = (8.47 - 0) kJ/mol[/latex]
[latex]\hat{H}_{3}=\Delta\hat{H}_{N_{2}} (25^{\circ}C → 300^{\circ}C) = (8.12 - 0) kJ/mol[/latex]
[latex]\hat{H}_{4}=\Delta\hat{H}_{O_{2}} (25^{\circ}C → 1000^{\circ}C) = (32.47 - 0) kJ/mol[/latex]
[latex]\hat{H}_{5}=\Delta\hat{H}_{N_{2}} (25^{\circ}C → 1000^{\circ}C) = (30.56 - 0) kJ/mol[/latex]
[latex]\hat{H}_{6}=\Delta\hat{H}_{CO_{2}} (25^{\circ}C → 1000^{\circ}C) = (48.60 - 0) kJ/mol[/latex]
[latex]\hat{H}_{7}=\Delta\hat{H}_{H_{2}O} (l,25^{\circ}C → H_{2}O (v,1000^{\circ}C))[/latex]
6 - For [latex]\hat{H_{7}}[/latex], the specific enthalpy for water is calculated. From the reference state of 25°C to the stream temperature of 1000°C, water is first heated to 100°C, which is its natural boiling point at 1atm, then vaporized, then heated in vapor phase to 1000°C. The enthalpy change of each process is calculated separately and added together to get [latex]\hat{H_{7}}[/latex].
Note: The [latex]T[/latex] in the formulas to calculate [latex]C_{P}[/latex] is given in kelvin. This doesn't make a difference when [latex]C_{P}[/latex] is given as a number, as the scales for one degree of Celcius and kelvin are the same, but the temperature must be converted to kelvin when [latex]T^2[/latex] is higher power of [latex]T[/latex] is used in calculation.
[latex]100°C=373K, 1000°C=1273K[/latex]
\begin{align*}
\hat{H}_{7}& = \int^{100^{\circ}C}_{25^{\circ}C} C_{P,l}dT + \Delta \hat{H}_{vap}(100^{\circ}C) + \int^{1000^{\circ}C}_{100^{\circ}C} C_{P,v}dT\\& = \int^{100^{\circ}C}_{25^{\circ}C} 75.4*10^{-3}dT + \Delta \hat{H}_{vap}(100^{\circ}C) \\& \;\;\;\;+\int^{1273K}_{373K} (33.46*10^{-3}+0.688*10^{-5}*T + 0.7604*10^{-8}*T^{2} - 3.593*10^{-12}*T^{3})dT\\& = 75.4*10^{-3}*(100-25)+ 40.66 \\& \;\;\;\; +(33.46*10^{-3}*T+\frac{1}{2}*0.688*10^{-5}*T^2 + \frac{1}{3}*0.7604*10^{-8}*T^{3} - \frac{1}{4}*3.593*10^{-12}*T^{4})\bigg\vert^{1273K}_{373K}\\& = (5.65 + 40.66 + 35.1)kJ/mol\\& = 81.46 kJ/mol
\end{align*}[latex]\hat{H}_{7}= (5.65 + 40.66 + 35.1)kJ/mol = 81.46\frac{kJ}{mol}[/latex]
Therefore, we have calculated all the specific enthalpy for the reactants and products:
| Substance | [latex]\dot{n}_{in}[/latex](mol/s) | [latex]\hat{H}_{in}[/latex] (kJ/mol) | [latex]\dot{n}_{out}[/latex] (mol/s) | [latex]\hat{H}_{out}[/latex] (kJ/mol) |
| [latex]C_{3}H_{8}[/latex] | 100 | 0 | - | - |
| [latex]O_{2}[/latex] | 600 | 8.47 | 100 | 32.47 |
| [latex]N_{2}[/latex] | 2256 | 8.12 | 2256 | 30.56 |
| [latex]CO_{2}[/latex] | - | - | 300 | 48.60 |
| [latex]H_{2}O[/latex] | - | - | 400 | 81.46 |
8 - Finally, solve the energy balance
[latex]\Delta\dot{H} + \Delta\dot{E}_{k} +\Delta\dot{E}_{p} = \dot{Q} + \dot{W}_{s}[/latex]
[latex]\Delta\dot{E}_{k}[/latex], [latex]\Delta\dot{E}_{p}[/latex], and [latex]\dot{W}_{s}[/latex] are assumed negligible for this system (as no information is provided on these)
[latex]\dot{Q} = \Delta\dot{H}[/latex]
[latex]\dot{Q} = -126 MW[/latex]
The combustion chamber releases [latex]126 MW[/latex] of heat to the environment
Heat of Formation [latex]\Delta H^{\circ}_{f}[/latex]
Formation Reaction: a reaction in which the compound is formed from its elemental constituents as they would normally occur in nature (eg. [latex]O_{2}[/latex] rather than [latex]O[/latex]).
For elemental consistituents, the energy of formation is [latex]\Delta H_{f}^{\circ}[/latex] = 0, since they would be forming themselves: [latex]O_{2}→O_{2}[/latex]
Standard specific heat of formation [latex]\Delta H^{\circ}_{f}[/latex]: the enthalpy change associated with forming 1 mole of the compound of interest at standard temperature ([latex]25^{\circ}C[/latex]) and pressure (1 atm)
| Compound | Reaction | [latex]\Delta\hat{H}^{\circ}_{f,i}[/latex] (kJ/mol) |
|---|---|---|
| Water | [latex]H_{2}(g) + \frac{1}{2}O_{2}(g) → H_{2}O(l)[/latex] | -285.83 |
| Methane | [latex]C(s) + 2H_{2}(g) → CH_{4}(g)[/latex] | -74.8936 |
| Ethane | [latex]2C(s) + 3H_{2}(g) → C_{2}H_{6}(g)[/latex] | -83.82 |
| Propane | [latex]3C(s) + 4H_{2}(g) → C_{3}H_{8}(g)[/latex] | -104.68 |
| Benzene | [latex]6C(s) + 3H_{2}(g) → C_{6}H_{6}(l)[/latex] | 82.88 |
| Toluene | [latex]7C(s) + 4H_{2}(g) → C_{7}H_{8}(l)[/latex] | 50.17 |
| Oxygen | [latex]O_{2}(g) → O_{2}(g)[/latex] | 0 |
| Nitrogen | [latex]N_{2}(g) → N_{2}(g)[/latex] | 0 |
We can use these heats of formation and Hess's law to find the heat of reaction for a given reaction:
| [latex]\Delta H^{\circ}_{r} = \Sigma_{i}\nu_{i}\Delta\hat{H}^{\circ}_{f,i} = \Sigma_{products}|\nu_{i}|\Delta\hat{H}^{\circ}_{f,i}-\Sigma_{reactants}|\nu_{i}|\Delta\hat{H}^{\circ}_{f,i}[/latex] |
Example: Heat of Formation
Consider the combustion of ethane:
[latex]C_{2}H_{6} (g) + \frac{7}{2} O_{2}(g) → 2 CO_{2} (g) + 3 H_{2}O (l)[/latex]
where [latex]\Delta H^{\circ}_{r} = -1560 kJ/mol[/latex]
To calculate the enthalpy using the heat of formation method, the following steps are taken:
[latex]\Delta H^{\circ}_{r} = (3*\Delta\hat{H}^{\circ}_{f,H_{2}O} + 2*\Delta\hat{H}^{\circ}_{f,CO_{2}}) - (1*\Delta\hat{H}^{\circ}_{f,C_{2}H_{6}}+\frac{7}{2}*\Delta\hat{H}^{\circ}_{f,O_{2}})[/latex]
[latex]\Delta H^{\circ}_{r} = (3*-285.83 + 2*-393.51)kJ/mol - (1*-83.82+\frac{7}{2}*0)kJ/mol[/latex]
[latex]\Delta H^{\circ}_{r} = -1561 kJ/mol[/latex]
The enthalpy of reaction using the heat of formation method is very close to the enthalpy of reaction (combustion).
Where to find Heat of Formation Data
One source of heat of formation data is the National Institute of Standards and Technology (NIST) Webbook NIST
To find the heat of formation data at this site:
1 - Look up the compound of interest
2 - Heat of formation data will be under "condensed phase thermochemistry data" (for liquids or solids) or "gas phase thermochemistry data" (for gases).
Heats of formation can also be found in Appendix E of "Introductory Chemical Engineering Thermodynamics" by J. Richard Elliot and Carl T. Lira.
Heat of Formation Method for Analyzing Energy Use in a Process
- Solve the material balance as much as possible: this can include solving for mass or molar flows using stoichiometry or mass balances
- Choose reference states for energy calculations: reference states provide a basis for enthalpy calculations. Choose reference states that make your calculations convenient or reference states that match the available data. Most data is provided at [latex]25^{\circ}C[/latex] and 1 atm, therefore this is a common reference state.
- Prepare and inlet-outlet enthalpy table: this table will include all the compounds involved in the system, the inlet molar flow, the inlet enthalpy values, the outlet molar flow, and the outlet enthalpy values. Fill out the table with all the known values and number the enthalpy values accordingly.
- Calculate all the enthalpies: use process paths to calculate the enthalpies listed in the table in step 3. The enthalpy change of each compound is calculated by summing the heat of formation, enthalpy change due to temperature change in the same state (which can be calculated using the [latex]C_{P}[/latex] values), and enthalpy change due to phase change when phase change is involved.
- Calculate the [latex]\Delta\dot{H}[/latex] for the system: this is done by multiplying each molar flow by the corresponding enthalpy and using the following expression. Remember that the stoichiometric coefficients are negative for reactants and positive for products: [latex]\Delta\dot{H} = \Sigma\dot{n}_{out}*\hat{H}_{out} - \Sigma\dot{n}_{in}*\hat{H}_{in}[/latex]
- Solve the energy balance: determine which energy terms are present in the system and solve accordingly.
Example: Heat of Formation Method for Energy Use
Let's consider the propane combustion chamber problem we analyzed before with the heat of reaction method, and let's see if we can get a similar answer using the heat of formation method.
1 - Solve the material balance as much as possible
This example already has the material balances solved
2 - Choose reference states for energy calculations
For reacting species: elemental species that make up reacting species at standard conditions; we will choose [latex]25^{\circ}C[/latex] at 1 atm with [latex]C(s)[/latex], [latex]H_{2}(g)[/latex], and [latex]O_{2}(g)[/latex].
For non-reacting species (same as [latex]\Delta H_{r}[/latex]): Use any convenient temperature (inlet temperature, outlet temperature, temperature in enthalpy table) Here, [latex]25^{\circ}C[/latex] and 1 atm works because of our enthalpy table values
3 - Prepare an inlet-outlet enthalpy table
| Substance | [latex]\dot{n}_{in}[/latex](mol/s) | [latex]\hat{H}_{in}[/latex] (kJ/mol) | [latex]\dot{n}_{out}[/latex] (mol/s) | [latex]\hat{H}_{out}[/latex] (kJ/mol) |
| [latex]C_{3}H_{8}[/latex] | 100 | [latex]\hat{H}_{1}[/latex] | - | - |
| [latex]O_{2}[/latex] | 600 | [latex]\hat{H}_{2}[/latex] | 100 | [latex]\hat{H}_{4}[/latex] |
| [latex]N_{2}[/latex] | 2256 | [latex]\hat{H}_{3}[/latex] | 2256 | [latex]\hat{H}_{5}[/latex] |
| [latex]CO_{2}[/latex] | - | - | 300 | [latex]\hat{H}_{6}[/latex] |
| [latex]H_{2}O[/latex] | - | - | 400 | [latex]\hat{H}_{7}[/latex] |
4 - Calculate all the enthalpies
| Substance | [latex]\Delta H^{\circ}_{f}[/latex] (kJ/mol) |
|---|---|
| [latex]C_{3}H_{8}[/latex] | -103.8 |
| [latex]O_{2}[/latex] | 0 |
| [latex]N_{2}[/latex] | 0 |
| [latex]CO_{2}[/latex] | -393.51 |
| [latex]H_{2}O (v)[/latex] | -241.835 |
[latex]\hat{H}_{1} = \Delta\hat{H}^{\circ}_{f,C_{3}H_{8}(g)} = -103.8 kJ/mol[/latex]
[latex]\hat{H}_{2}=\Delta\hat{H} (O_{2} (25^{\circ}C → 300^{\circ}C) = (8.47 - 0) kJ/mol[/latex]
[latex]\hat{H}_{3}=\Delta\hat{H} (N_{2} (25^{\circ}C → 300^{\circ}C) = (8.12 - 0) kJ/mol[/latex]
[latex]\hat{H}_{4}=\Delta\hat{H} (O_{2} (25^{\circ}C → 1000^{\circ}C) = (32.47 - 0) kJ/mol[/latex]
[latex]\hat{H}_{5}=\Delta\hat{H} (N_{2} (25^{\circ}C → 1000^{\circ}C) = (30.56 - 0) kJ/mol[/latex]
[latex]\hat{H}_{6}=\Delta\hat{H}^{\circ}_{f,CO_{2}(g)} + \int^{1000^{\circ}C}_{25^{\circ}C} C_{p,CO_{2}(g)}dT = -344.9 kJ/mol[/latex]
[latex]\hat{H}_{7}=\Delta\hat{H}^{\circ}_{f,H_{2}O(v)} + \int^{1000^{\circ}C}_{25^{\circ}C} C_{p,H_{2}O(v)}dT = -204.1 kJ/mol[/latex]
Here, you assume that water forms as vapor directly in the reaction. Therefore, there is no need to account for heat of vaporization.
| Substance | [latex]\dot{n}_{in}[/latex](mol/s) | [latex]\hat{H}_{in}[/latex] (kJ/mol) | [latex]\dot{n}_{out}[/latex] (mol/s) | [latex]\hat{H}_{out}[/latex] (kJ/mol) |
| [latex]C_{3}H_{8}[/latex] | 100 | -103.8 | - | - |
| [latex]O_{2}[/latex] | 600 | 8.47 | 100 | 32.47 |
| [latex]N_{2}[/latex] | 2256 | 8.12 | 2256 | 30.56 |
| [latex]CO_{2}[/latex] | - | - | 300 | -344.9 |
| [latex]H_{2}O[/latex] | - | - | 400 | -204.1 |
5 - Calculate $\Delta\dot{H}$ for the reactor
[latex]\Delta\dot{H} = \Sigma\dot{n}_{out}*\hat{H}_{out} - \Sigma\dot{n}_{in}*\hat{H}_{in}=1.26x10^{5} kJ/s[/latex]
6 - Finally, solve the energy balance
[latex]\dot{Q} = \Delta\dot{H}[/latex]
[latex]\dot{Q} = -126 MW[/latex]
Exercise: Heat of Formation
Consider a reactor where the following reaction reaction takes place :
[latex]3 NO_{2}(g) + H_{2}O(l) → 2HNO_{3}(aq) + NO(g)[/latex]
[latex]\Delta H^{\circ} = ?[/latex]
The reactor uses an 0.8 kW impeller (mixer for liquids) that mixes the reactor contents. The reaction occurs at standard temperature and pressure. A feed consisting of 100 mol/s and 300 mol/s of water and [latex]NO_{2}[/latex], respectively, enters the reactor. The reaction goes to completion and the products stream consists of 200 mol/s of [latex]HNO_{3}[/latex] and 100 mol/s of [latex]NO[/latex].
The following heats of formation are available:
| Number | Reaction | [latex]\Delta H^{\circ}_{f,i}[/latex] (kJ/mol) |
|---|---|---|
| 1 | [latex]\frac{1}{2}N_{2}(g) + O_{2}(g) → NO_{2}(g)[/latex] | 33.2 |
| 2 | [latex]H_{2}(g) + \frac{1}{2}O_{2}(g) → H_{2}O(l)[/latex] | -285.8 |
| 3 | [latex]\frac{1}{2}H_{2}(g) + \frac{1}{2}N_{2}(g) + \frac{3}{2}O_{2}(g) → HNO_{3}O(aq)[/latex] | -207.4 |
| 4 | [latex]\frac{1}{2}N_{2}(g) + \frac{1}{2}O_{2}(g) → NO(g)[/latex] | 90.2 |
How much heat must be removed from the reactor in one day for it to remain at standard temperature?
Solution
Step 1: Determine what combination of the given reactions will give the desired reaction (Hess's Law)
For this desired reaction:
\begin{align*}
& -3*Reaction1\\
& -1*Reaction2\\
& +2*Reaction3\\
& +1*Reaction4
\end{align*}
Summing all the reactions above yields:
[latex]3 NO_{2}(g) + H_{2}O(l) → 2HNO_{3}(aq) + NO(g)[/latex]
Step 2: Add all the individual reaction enthalpies:
[latex]\Delta H^{\circ}_{r} = -3*\Delta H^{\circ}_{1} + -1*\Delta H^{\circ}_{2} + 2*\Delta H^{\circ}_{3} + \Delta H^{\circ}_{4}[/latex]
[latex]\Delta H^{\circ}_{r} = -3*33.2 kJ/mol + -1*-285.8 kJ/mol + 2*-207.4 kJ/mol + 90.2 kJ/mol[/latex]
[latex]\Delta H^{\circ}_{r}= (-99.6+285.8+-414.8+90.2)kJ/mol = -138.4 kJ/mol[/latex]
Step 3: Calculate the overall change in enthalpy using the extent of the reaction:
\begin{align*}
\xi &= \frac{|(\dot{n}_{i})_{out}-(\dot{n}_{i})_{in}|}{|(\dot{\nu}_{i})|}hat\\& = \frac{|(\dot{n}_{H_{2}O})_{out}-(\dot{n}_{H_{2}O})_{in}|}{|(\dot{\nu}_{H_{2}O})|} \\& =\frac{|0\frac{mol}{s} - 100 \frac{mol}{s}|}{|-1|}\\ &= 100 mol/s
\end{align*}
[latex]\Delta\dot{H} = \xi*\Delta H^{\circ}_{r}[/latex]
[latex]\Delta\dot{H} = 100\frac{mol}{s}*-138.4\frac{kJ}{mol} = -138400 \frac{kJ}{s}[/latex]
Step 4: Calculate how much work is done on the reactor by the impeller in one day:
[latex]W_{s} = 0.8\frac{kJ}{s}*3600\frac{s}{hr}*24hr[/latex]
[latex]W_{s} = 69120 kJ[/latex]
Step 5: Perform an energy balance on the reactor
[latex]\dot{Q}=\Delta H^{\circ}-W_{s}[/latex]
[latex]\dot{Q} = (-138400 - 69120 ) kJ = -207520 kJ[/latex]
Summary of Heat of Reaction and Formation Methods
Heat of Reaction Method
[latex]\Delta\dot{H} = \xi\Delta\dot{H}^{\circ}_{r} + \Sigma\dot{n}_{out}*\hat{H}_{out} - \Sigma\dot{n}_{in}*\hat{H}_{in}[/latex]
where [latex]\hat{H}_{i} = \int^{T}_{T_{ref}}C_{P}dT[/latex] assuming there are no phase changes
If a phase change occurs, an additional heat term (i.e. heat of vapourization) will be added to the [latex]\hat{H}_{i}[/latex] term in both methods
| [latex]\Delta\dot{H} = \xi\Delta\dot{H}_{r} + \Sigma\dot{n}_{out}*\int^{T_{out}}_{T_{ref}}C_{P}dT - \Sigma\dot{n}_{in}*\int^{T_{in}}_{T_{ref}}C_{P}dT[/latex] |
Heat of Formation Method
[latex]\Delta\dot{H} = \Sigma\dot{n}_{out}*\hat{H}_{out} - \Sigma\dot{n}_{in}*\hat{H}_{in}[/latex]
where [latex]\hat{H}_{i} = \hat{H}^{\circ}_{f,i} + \int^{T}_{T_{ref}}C_{P}dT[/latex] assuming there are no phase changes
[latex]\Delta\dot{H} = \Sigma\dot{n}_{out}*(\hat{H}^{\circ}_{f,i} + \int^{T_{out}}_{T_{ref}}C_{P}dT) - \Sigma\dot{n}_{in}*(\hat{H}^{\circ}_{f,i} + \int^{T_{in}}_{T_{ref}}C_{P}dT)[/latex]
For the same system, equating [latex]\Delta\dot{H}[/latex] for both methods, we can see that the [latex]C_{P}[/latex] terms cancel. We obtain the following:
| [latex]\xi\Delta\dot{H}^{\circ}_{r} = \Sigma\dot{n}_{out}*\hat{H}^{\circ}_{f,i} - \Sigma\dot{n}_{in}*\hat{H}^{\circ}_{f,i}[/latex] |
Overall, the final [latex]\Delta\dot{H}[/latex] for both methods will be the same. This means we can choose either method based on which is easiest to use with the information we have available.
Exercise: Comparing Both Methods
Ethane and oxygen are fed into a furnace at 100 kmol/hr and 500 kmol/hr, respectively. Ethane goes through complete combustion in the furnace. Oxygen, carbon dioxide, and water exit the furnace at 150 kmol/hr, 200 kmol/hr, and 300 kmol/hr, respectively.
[latex]C_{2}H_{6}(g) + \frac{7}{2}O_{2}(g) → 2CO_{2}(g) + 2H_{2}O(l)[/latex]
where the heat of reaction is: [latex]\Delta H^{\circ}_{r} = -1560.7 kJ/mol[/latex]
The following data on heats of formations is available:
| Compound | [latex]\Delta H^{\circ}_{f,i}[/latex] (kJ/mol) |
| Ethane | -83.82 |
| Oxygen | 0 |
| Carbon Dioxide | -393.51 |
| Water (liquid) | -285.83 |
Assume the reaction takes place in the furnace at standard temperature and pressure. What is the [latex]\Delta H^{\circ}[/latex] for the desired reaction? Calculate the change in enthalpy using both the heat of reaction and heat of formation method
Solution
Heat of Reaction Method
Step 1: Determine the extent of the reaction:
\begin{align*}
\xi &= \frac{|(\dot{n}_{i})_{out}-(\dot{n}_{i})_{in}|}{|(\dot{\nu}_{i})|}hat\\& = \frac{|(\dot{n}_{C_{2}H_{6}})_{out}-(\dot{n}_{C_{2}H_{6}})_{in}|}{|(\dot{\nu}_{C_{2}H_{6}})|} \\& =\frac{|0\frac{kmol}{hr} - 100 \frac{kmol}{hr}|}{|-1|}\\ &= 100 kmol/hr
\end{align*}
Step 2: Multiply the extent of the reaction by the heat of reaction for combusion
[latex]\Delta \dot{H} = \xi*\Delta H^{\circ}_{r}[/latex]
[latex]\Delta \dot{H} = 100\frac{kmol}{hr}*-1560.7 \frac{kJ}{mol} * 1000 \frac{mol}{kmol}[/latex]
[latex]\Delta \dot{H} = -156 MJ/hr[/latex]
Heat of Formation Method
Step 1: Set up the heat of formation formula
[latex]\Delta\dot{H} = \Sigma\dot{n}_{out}*\hat{H}_{out} - \Sigma\dot{n}_{in}*\hat{H}_{in}[/latex]
[latex]\Delta\dot{H} = (\dot{n}_{out,ethane}*\hat{H}_{ethane})+(\dot{n}_{out,oxygen}*\hat{H}_{oxygen}) + (\dot{n}_{out,water}*\hat{H}_{water}) + (\dot{n}_{out,carbon dioxide}*\hat{H}_{carbon dioxide}) - (\dot{n}_{in,ethane}*\hat{H}_{ethane})+(\dot{n}_{in,oxygen}*\hat{H}_{oxygen}) + (\dot{n}_{in,water}*\hat{H}_{water}) + (\dot{n}_{in,carbon dioxide}*\hat{H}_{carbon dioxide})[/latex]
Step 2: Plug in the enthalpies of formation and calculate the change in enthalpy
| Compound | [latex]n_{in}[/latex] (kmol/hr) | [latex]n_{out}[/latex] (kmol/hr) |
|---|---|---|
| Ethane | 100 | 0 |
| Oxygen | 500 | 150 |
| Carbon Dioxide | 0 | 200 |
| Water | 0 | 300 |
[latex]\Delta\dot{H} = ((0\frac{kmol}{hr}*-83.82\frac{kJ}{mol})+(150\frac{kmol}{hr}*0\frac{kJ}{mol}) + (200\frac{kmol}{hr}*-393.51\frac{kJ}{mol}) + (300\frac{kmol}{hr}*-285.83\frac{kJ}{mol}) - (100\frac{kmol}{hr}*-83.82\frac{kJ}{mol}) - (500\frac{kmol}{hr}*0\frac{kJ}{mol}) - (0\frac{kmol}{hr}*-393.51\frac{kJ}{mol}) - (0\frac{kmol}{hr}*-285.83\frac{kJ}{mol}))*1000\frac{mol}{kmol}[/latex]
[latex]\Delta\dot{H} = -150 MJ[/latex]
Notice that the methods aren't exactly equal. This is due to slight differences in available enthalpy data and assumptions made regarding temperature


Feedback/Errata