6. Entropy and the Second Law of Thermodynamics
6.0 Chapter introduction and learning objectives
Heat and work are two forms of energy. From the first law of thermodynamics, we may say “1 kJ of heat = 1 kJ of work”, but how do we interpret the “=” sign here? Does it mean that heat and work are equivalent?
Let us consider a daily example. In winter, you might need an electric resistance heater to warm up your room. From Figure 6.0.1 (a), the electric charges do work on the resistor as they flow in the direction of lower electric potential. In this process, the electric energy from the circuit is converted to heat, which is then dissipated in your room, or the surroundings. As a result, the room temperature will tend to rise. If the heater consumes 1 kW of electric power, it will generate and dissipate approximately 1 kW of heat to the surroundings. In terms of the amount of energy conversion, we can safely say that 1 kW of electric energy is no different from 1 kW of heat. However, is this energy conversion process reversible? If we simply cool the surroundings and provide 1 kW of heat to the resistor, is it possible to generate 1 kW of electric power from this 1 kW of heat? Unfortunately, the answers to both questions are no, see Figure 6.0.1 (b).
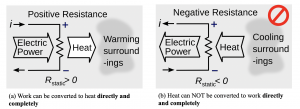
This example illustrates an important concept of heat-work conversion. Although heat and work can be converted between each other, they are not equivalent. Work can always be converted to heat completely and directly; but heat cannot be converted to work completely and directly even under an ideal condition, because heat is a form of energy of “low” quality (a more “random”, “disordered” system) and work is a form of energy of “high” quality (a highly “ordered” system). A natural, spontaneous process has a tendency to move toward a greater degree of randomness or disorder in a system. Therefore, an energy conversion process in nature tends to occur in the direction of converting energy from a “high” quality form to a “low” quality form. The first law of thermodynamics addresses the heat and work conversion quantitatively, but has no indication on whether or under what condition such conversion can actually happen. Satisfying the first law of thermodynamics does not ensure a process can take place in reality. Whether a process can happen or not is determined by the second law of thermodynamics.
This chapter introduces the concepts of reversible and irreversible processes, Carnot cycles, entropy generation and the second law of thermodynamics, and the applications of the second law to both closed and open systems.
Learning Objectives
After completing the chapter, you should be able to
- Demonstrate an understanding of reversible and irreversible processes and Carnot cycles
- Calculate the thermal efficiency of heat engines and the coefficient of performance (COP) of refrigerators and heat pumps
- Calculate the change in entropy in real substances, ideal gases, solids, and liquids
- Explain entropy generation, entropy transfer mechanisms, and the second law of thermodynamics
- Apply the second law of thermodynamics to closed systems
- Apply the second law of thermodynamics to various steady flow devices
Media Attributions
- Electric resistance heater © Chetvorno is licensed under a CC0 (Creative Commons Zero) license

