1. Basic Concepts and Definitions
1.1 What is thermodynamics about?
You probably have this experience; when you rub your hands quickly for a few minutes, your hands will start to feel warmer. How is this common phenomenon related to thermodynamics? Well, when you rub your hands quickly, your muscles do work. This work is then converted to heat; therefore, you feel warmer. Heat and work are two forms of energy. Work can be converted to heat, as seen in this daily example. However, can heat be converted to work? Can we use heat to produce work?
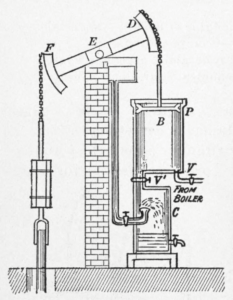
Heat engine is a device that produces work continuously by absorbing heat from a high-temperature heat source and rejecting the waste heat to a low-temperature heat sink. Since the 17th century, various heat engines were invented in an attempt to harness work from heat. Figure 1.1.1 illustrates Watt’s engine invented by Scottish engineer James Watt in the late 18th century. Watt’s engine is one of the most successful early heat engines. Its main components are a boiler (not shown in the figure) and a condenser, each connecting to a piston-cylinder device. The two valves, V and V’, control the flow of steam into and out of the cylinder. When valve V opens, valve V’ remains closed. Steam from the boiler enters the cylinder, pushing the piston up until it reaches the top of the cylinder. Then valve V’ opens, and valve V closes. The steam in the cylinder escapes to the condenser and is condensed, creating a vacuum in the cylinder. Consequently, the piston moves downward under atmospheric pressure. The reciprocating motion of the piston drives the pivoting beam DEF, which then powers the pump chained to the beam. Watt’s engine demonstrates how heat is converted to work. This conversion relies on the phase change of a working fluid, e.g., water, in the Watt’s engine. The boiler in the Watt’s engine is the heat source, where the hot steam is generated; and the condenser is the heat sink, where the hot steam is cooled and condensed to liquid water. All heat engines need a working fluid circulating in a specially-arranged set of equipment, which operates between a high-temperature heat source and a low-temperature heat sink. Figure 1.1.2 is a schematic drawing of a heat engine. The yellow circle represents the heat engine consisting of a set of equipment.
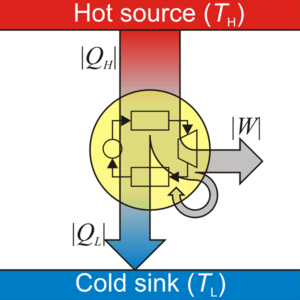
By examining Figure 1.1.2, you might notice that a certain amount of heat is not converted to work. It is true that not all of the heat from a heat source can be converted to useful work! Heat engines and their underlying principles are governed by the two fundamental laws of thermodynamics, the first and second laws of thermodynamics. We will briefly introduce the two laws here and will provide detailed explanations in Chapters 4-6.
- The first law of thermodynamics is about energy conservation. Energy can neither be created nor destroyed. It can only be converted between different forms.
- The second law of thermodynamics explains why all real processes are irreversible, and how the irreversibility of a process is quantified with the concept of entropy generation. In reality, all processes always occur in the direction of producing positive entropy generation due to the existence of irreversibilities. From the second law of thermodynamics, we can estimate the theoretical limit of efficiency that a real thermodynamic process or system can possibly achieve.
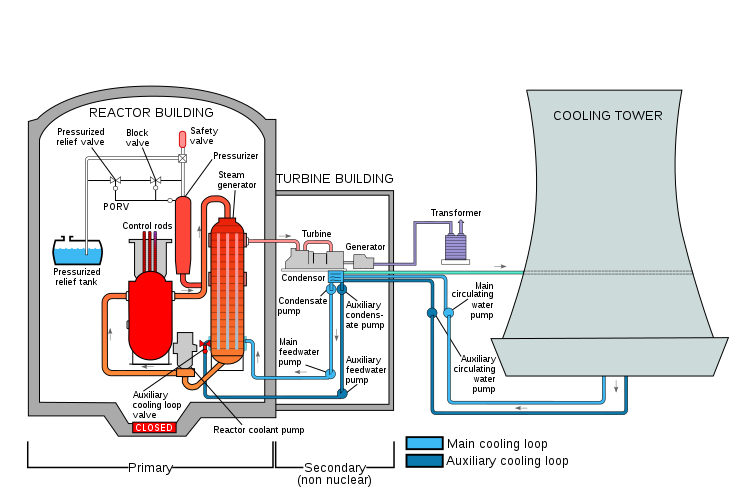
Thermodynamics emerged in the early 19th century with the inventions of heat engines. It originally focused on the scientific theories of heat-work conversion, and the operations and efficiency improvement of heat engines. Nowadays, the applications of thermodynamics have extended to all fields related to energy conversion and conservation. In engineering fields, the principles of thermodynamics are widely used in the design of thermal systems, such as power plants using different energy sources (e.g., steam, gas, nuclear, hydro, wind, and solar), air conditioning and refrigeration systems, jet engines, biomedical devices, and chemical processes, to name but a few. Figure 1.1.3 is a schematic drawing of a nuclear power plant, whose performance and efficiency are governed by the fundamental principles of thermodynamics.
Practice Problems
Media Attributions
- Watt’s Heat Engine © Caltrop is licensed under a Public Domain license
- Schematic drawing of a heat engine © Brundl15 is licensed under a CC BY-SA (Attribution ShareAlike) license
- Nuclear Power Plant © Anynobody is licensed under a CC BY-SA (Attribution ShareAlike) license
Work is a form of energy that is transferred to or from a body by applying a force on that body along a displacement.
Heat is a form of energy. Heat transfer will take place between two objects if they are at different temperatures.
Heat engine is a device that produces work by absorbing heat from a high-temperature body (heat source) and rejecting the waste heat to a low-temperature body (heat sink).
Thermodynamics is a branch of science. It originally focused on the scientific theories of heat-work conversion, and the operations and efficiency improvement of heat engines. Nowadays, the applications of thermodynamics have extended to all fields related to energy conversion and conservation.

