6. Entropy and the Second Law of Thermodynamics
6.1 Heat engine
A heat engine is a continuously operating device that produces work by transferring heat from a heat source (high-temperature body) to a heat sink (low-temperature body) using a working fluid. In a heat engine cycle, a working fluid may remain as a single-phase fluid or experience phase changes.
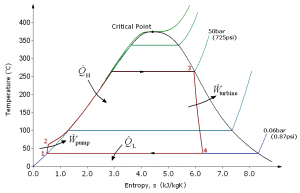
A steam engine is a type of heat engine commonly used in steam power generating plants. It operates on Rankine cycles and uses water as the working fluid. We will use a steam engine to illustrate how heat is converted to work in heat engines. A typical steam engine consists of four main equipment: boiler, turbine, condenser, and pump, as shown in Figure 6.1.1. The T-s diagram in Figure 6.1.2 illustrates the four processes in a Rankine cycle:
- Water at a low pressure and a low temperature (state 1) is pumped to a boiler. The pump consumes power, [latex]\dot{W}_{pump}[/latex], in order to maintain a continuous supply of water to the boiler while increasing the pressure of the water entering the boiler (state 2). Process 1-2 may be assumed adiabatic.
- In the boiler, the liquid water absorbs heat, [latex]\dot{Q}_{H}[/latex], from an external heat source and changes into high-temperature, high-pressure steam (state 3). The pressure drop in the boiler is usually negligible; therefore, process 2-3 may be assumed isobaric.
- The high-temperature, high-pressure steam then expands in the turbine, making the turbine rotate continuously, and thus generating mechanical power, [latex]\dot{W}_{turbine}[/latex]. During the expansion process, the temperature and pressure of the steam decrease. Consequently, the steam leaving the turbine (state 4) becomes a low-temperature, low-pressure, two-phase mixture. Process 3-4 may be assumed adiabatic.
- The steam leaving the turbine then enters a condenser and is condensed to a saturated or compressed liquid (state 1). During this process, heat, [latex]\dot{Q}_{L}[/latex], is removed from the steam. The pressure drop in the condenser is usually negligible; therefore, process 4-1 may be assumed isobaric.
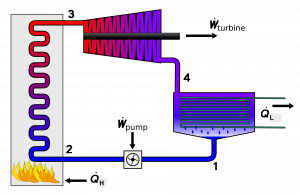
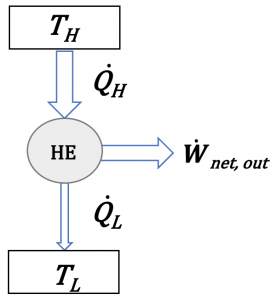
Figure 6.1.3 is a simplified schematic for analyzing the energy conservation in heat engines. Applying the first law of thermodynamics to the cycle, we can write
[latex]\dot{Q}_{H} - \dot{Q}_{L} = \dot{W}_{turbine} - \dot{W}_{pump} = \dot{W}_{net, out} [/latex]
Clearly, the heat removed by the condenser, [latex]\dot{Q}_L[/latex], cannot be converted to useful work. It is wasted in order to complete the cycle. In other words, a heat engine cannot convert all the heat supplied by the heat source (e.g., boiler) to useful work, even under ideal conditions. Thermal efficiency is a dimensionless parameter used to measure the performance of a heat engine.
[latex]\eta_{th}=\displaystyle\frac{desired\ output}{required\ input}=\frac{{\dot{W}}_{net,\ out}}{{\dot{Q}}_H}=1-\frac{{\dot{Q}}_L}{{\dot{Q}}_H}[/latex]
where
[latex]\dot{Q}_H[/latex]: heat absorbed from the heat source, in kW
[latex]\dot{Q}_L[/latex]: heat rejected to the heat sink, in kW
[latex]\dot{W}_{net,\ out}[/latex]: net work output from the heat engine, in kW
[latex]\eta_{th}[/latex]: thermal efficiency of the heat engine, dimensionless
Example 1
Figures 6.1.e1 and 6.1.e2 illustrate a Rankine cycle consisting of a two-stage steam engine and a feedwater heater. The steam engine is enclosed in the red outlines in Figure 6.1.e1. The two stages of the turbine are labelled as HE1 and HE2, respectively. In stage 1, the steam absorbs heat, [latex]\dot{Q}_H[/latex], from the boiler and generates a power, [latex]\dot{W}_{1}[/latex]. A portion of the exhaust steam from stage 1 then enters stage 2, further generating a power, [latex]\dot{W}_{2}[/latex]. The remaining exhaust steam from stage 1 is used to preheat the feed water. If the thermal efficiencies of the two turbine stages are [latex]\eta_{th,1}[/latex] and [latex]\eta_{th,2}[/latex], what is the overall thermal efficiency of the cycle as a function of [latex]\eta_{th,1}[/latex] and [latex]\eta_{th,2}[/latex]? Assume 90% of the exhaust steam exiting from stage 1 enters stage 2 and generates the power, [latex]\dot{W}_{2}[/latex].
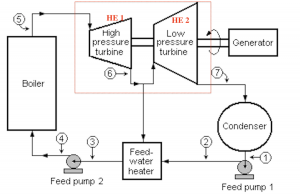
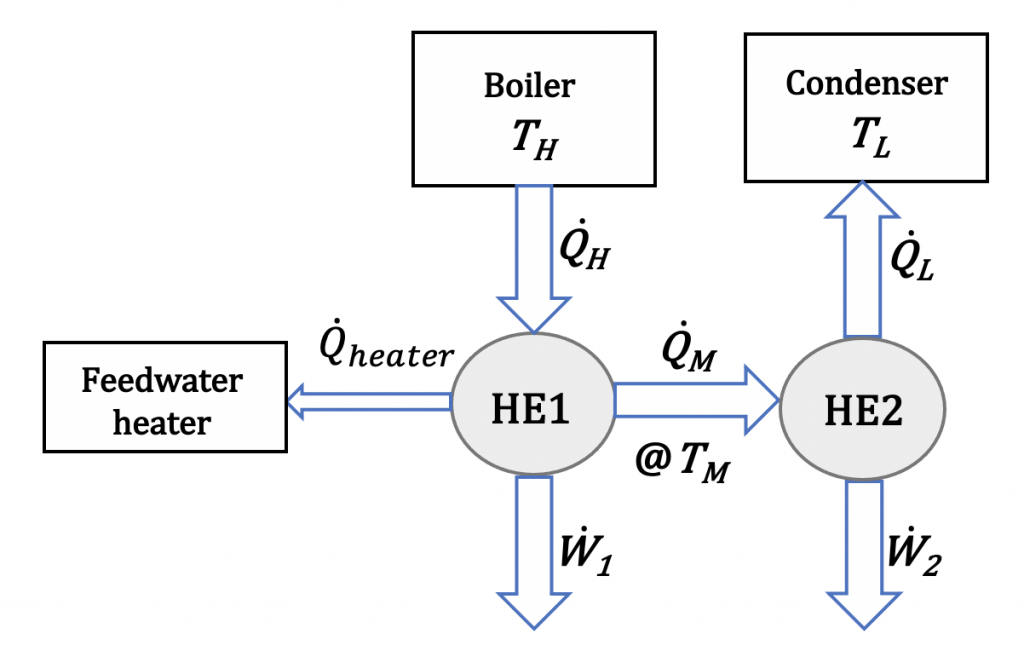
Solution:
The thermal efficiency of the first and second stages of the steam turbine can be written as
[latex]\eta_{th,1} = \dfrac{\dot{W}_{1}}{\dot{Q}_{H}}[/latex] [latex]\eta_{th,2} = \dfrac{\dot{W}_{2}}{\dot{Q}_{M}}[/latex]
The desired output of the cycle is the total power generated by the turbine and the required energy input comes from the boiler; therefore,
[latex]\eta_{th} = \dfrac{\dot{W}_{tot}}{\dot{Q}_{H}}=\dfrac{\dot{W}_{1} + \dot{W}_{2}}{\dot{Q}_{H}}[/latex]
Apply the first law of thermodynamics to the first stage, HE1. Note that 90% of the exhaust steam from stage 1 enters stage 2; therefore,
[latex]\dot{Q}_{M} + \dot{Q}_{Heater} = \dot{Q}_{H} - \dot{W}_{1}[/latex]
and
[latex]\dot{Q}_{M} = 0.9 (\dot{Q}_{H} - \dot{W}_{1})[/latex]
Combine the above equations and rearrange,
[latex]\begin{align*} \because\eta_{th} &= \dfrac{\dot{W}_{1}}{\dot{Q}_{H}} + \dfrac{\dot{W}_{2}}{\dot{Q}_{H}} \\&=\eta_{th,1} + \dfrac{\eta_{th,2}\dot{Q}_{M}}{\dot{Q}_{H}} \\&=\eta_{th,1} + \dfrac{\eta_{th,2} \times 0.9(\dot{Q}_{H}-\dot{W}_{1})}{\dot{Q}_{H}} \end{align*}[/latex]
[latex]\therefore \eta_{th} = \eta_{th,1} + 0.9 \eta_{th,2}(1-\eta_{th,1})[/latex]
Practice Problems
Media Attributions
- Rankine cycle © Andrew Ainsworth is licensed under a CC BY-SA (Attribution ShareAlike) license
- Rankine cycle, T-s diagram © Andrew Ainsworth is licensed under a CC BY-SA (Attribution ShareAlike) license
- Two-stage steam turbine with a feedwater heater © Milton Beychok is licensed under a CC BY-SA (Attribution ShareAlike) license
A heat engine, refrigerator or heat pump must operate between a high-temperature body and a low temperature body. The high-temperature body is called heat source.
A heat engine, refrigerator or heat pump must operate between a high-temperature body and a low-temperature body. The low-temperature body is called heat sink.