6. Entropy and the Second Law of Thermodynamics
6.3 The second law of thermodynamics: Kelvin-Planck and Clausius statements
The first law of thermodynamics focuses on energy conservation. It does not describe any restrictions or possibilities for a process to take place. A process satisfying the first law of thermodynamics may or may not be achievable in reality. In fact, whether a process is possible is governed by both the first and the second laws of thermodynamics. There are two classical statements of the second law of thermodynamics; one imposes the limits on the operation of heat engines, and the other on the operation of refrigerators and heat pumps.
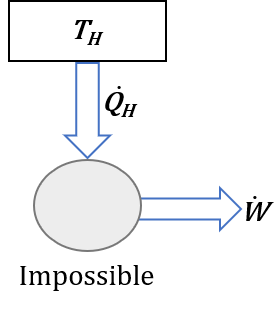
Kelvin-Planck Statement: it is impossible for any device that operates on a cycle to receive heat from a single reservoir and produce only a net amount of work. Figure 6.3.1 illustrates the Kelvin-Planck statement. Any device violating the Kelvin-Planck statement would produce a net work output equivalent to the amount of the heat received by the device, or [latex]\dot{W}=\dot{Q}_H[/latex], thus resulting in a thermal efficiency [latex]\eta_{th}=\dot{W}/\dot{Q}_H=100 \%[/latex].
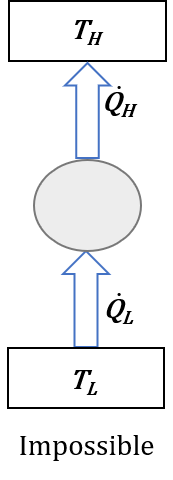
Clausius statement: it is impossible to construct a device that operates in a cycle and produces no effect other than the transfer of heat from a lower-temperature body to a higher-temperature body. Figure 6.3.2 illustrates the Clausius statement. Any device violating the Clausius statement would have zero work input and nonzero heat transfer received and delivered by the device, or [latex]\dot{W}=0[/latex], [latex]\dot{Q}_H=\dot{Q}_L \neq 0[/latex], and thus a [latex]COP=\dot{Q}_L / \dot{W}=\infty[/latex].
Although the Kelvin–Planck and Clausius statements refer to different cycles, they are equivalent in their consequences. Any device that violates the Kelvin–Planck statement must violate the Clausius statement, and vice versa.
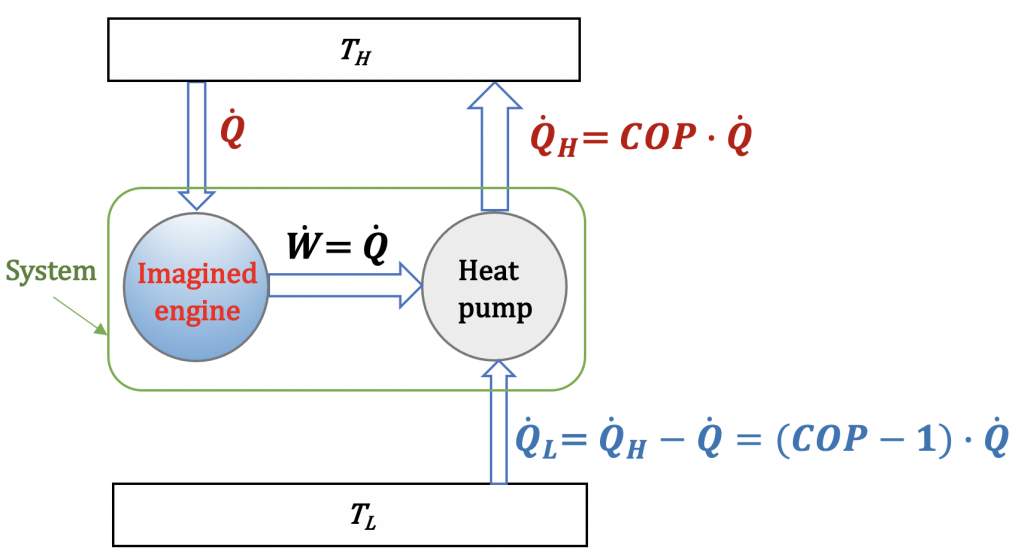
Let us consider a combined system, which consists of a heat pump and an imagined engine operating between a heat source at a higher temperature [latex]T_H[/latex] and a heat sink at a lower temperature [latex]T_L[/latex], as shown in Figure 6.3.3.
- Let us assume the imagined engine violates the Kelvin-Planck statement; therefore, it can convert all of the heat absorbed from the heat source to useful power, or [latex]\dot{W}=\dot{Q}[/latex]. The power generated from the imagined engine is then used to drive the heat pump.
- For the heat pump, [latex]COP=\dot{Q}_H / \dot{W}[/latex]. Therefore, the heat released by the heat pump to the heat source is [latex]\dot{Q}_H=COP\cdot \dot{Q}[/latex].
- Applying the first law to the heat pump, the heat absorbed by the heat pump from the heat sink must be [latex]\dot{Q}_L=\dot{Q}_H-\dot{Q}=(COP-1) \dot{Q}[/latex].
- Now let us compare the net heat absorbed by the combined system from the heat sink, [latex]\dot{Q}_L=(COP-1) \dot{Q}[/latex], and the net heat rejected by the combined system to the heat source, [latex]\dot{Q}_H-\dot{Q}=(COP-1) \dot{Q}[/latex]. They are exactly the same! What the combined system does is to transfer heat from the heat sink (lower-temperature body) to the heat source (higher-temperature body) without consuming any power at all!
It is apparent that the combined system violates the Clausius statement because the imagined engine violates the Kelvin-Planck statement. Since the Kelvin-Planck and Clausius statements are equivalent, both of them may be used to describe the second law of thermodynamics.
A real process or device must satisfy both the first and the second laws of thermodynamics: the first law sets a constraint on the amount of energy that must be conserved in a process or a device; the second law indicates whether a process is possible in reality. Any device that violates either the first or the second law is called a perpetual-motion machine, which attempts to produce work from nothing or convert heat completely to useful work. In fact, no perpetual-motion machine can actually work.
Example 1
Consider the following scenarios proposed for a heat engine or a refrigerator. For each of the scenarios, determine if the proposed heat engine or refrigerator satisfies the first and second laws of thermodynamics.
- Proposed heat engine: [latex]\dot{Q}_H=10\ {\rm{kW}}, \dot{Q}_L=3\ {\rm{kW}}, \dot{W}_{net, out}=5\ {\rm{kW}}[/latex]
- Proposed refrigerator: [latex]\dot{Q}_H=9\ {\rm{kW}}, \dot{Q}_L=4\ {\rm{kW}}, \dot{W}_{in}=0\ {\rm{kW}}[/latex]
Solution:
(1) Proposed heat engine: [latex]\dot{Q}_H = 10 \ {\rm{kW}}, \dot{Q}_L = 3 \ {\rm{kW}}, \dot{W}_{net,out} = 5 \ {\rm{kW}}[/latex]
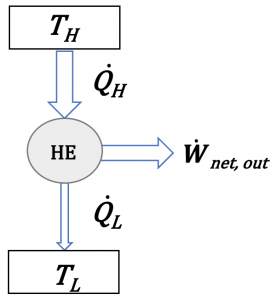
The first law requires [latex]\dot{Q}_{H} = \dot{Q}_{L} + \dot{W}_{net,out}[/latex], and the second law requires [latex]\dot{Q}_L > 0[/latex]. Since [latex]10 \neq 3 +5[/latex] and [latex]\dot{Q}_L = 3 \ {\rm{kW}}>0[/latex], the proposed heat engine doesn't satisfy the first law of thermodynamics; but it satisfies the second law of thermodynamics or the Kelvin-Planck statement.
(2) Proposed refrigerator: [latex]\dot{Q}_{H} = 9 \ {\rm{kW}}, \dot{Q}_{L} = 4 \ {\rm{kW}}, \dot{W}_{in} = 0 \ {\rm{kW}}[/latex]
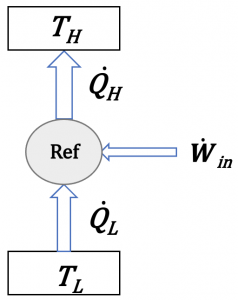
The first law requires [latex]\dot{Q}_{H} = \dot{Q}_{L} + \dot{W}_{in}[/latex], and the second law requires [latex]\dot{W}_{in} > 0[/latex]. Since [latex]9 \neq 4 + 0[/latex] and [latex]\dot{W}_{in} = 0[/latex], the proposed refrigerator satisfies neither the first nor the second laws of thermodynamics. It violates the Clausius statement.
Practice Problems
It is impossible for any device that operates on a cycle to receive heat from a single reservoir and produce a net amount of work.
It is impossible to construct a device that operates in a cycle and produces no effect other than the transfer of heat from a lower-temperature body (heat sink) to a higher-temperature body (heat source).

