1. Basic Concepts and Definitions
1.2 System and surroundings
In thermodynamics, a system refers to a selected quantity of matter in the case of closed systems or a selected region in space in the case of open systems, see Figure 1.2.1. The rest of the universe outside the system is called surroundings, and the surface that separates the system and its surroundings is called boundary. A boundary may be fixed or movable, real or imaginary, rigid or flexible.

A system interacts with its surroundings through two mechanisms:
- Mass transfer
- Energy transfer (i.e., in the form of heat and work)
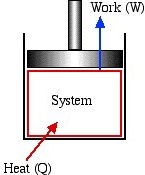
A system of a fixed mass is a closed system, which can only interact with its surroundings through energy transfer. Mass cannot cross the boundary of a closed system. For example, a sealed bottle of soft drink, Figure 1.2.2, can be modelled as a closed system because there is a fixed amount of liquid in the bottle. When you take the bottle out of your cooler, the liquid will warm up slowly due to the temperature difference between the bottle and the ambient air (surroundings). In other words, the system (the liquid in the bottle) interacts with its surroundings (the ambient air) through energy transfer (in the form of heat transfer). Figure 1.2.3 illustrates a piston-cylinder device, which can also be modelled as a closed system. The amount of the fluid in the cylinder (the system) remains constant as the piston moves. Only the transfer of energy, in the form of heat and work, may happen across the system boundary consisting of the cylinder walls and the lower surface of the piston.

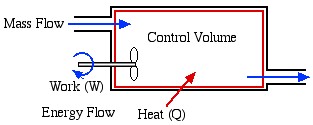
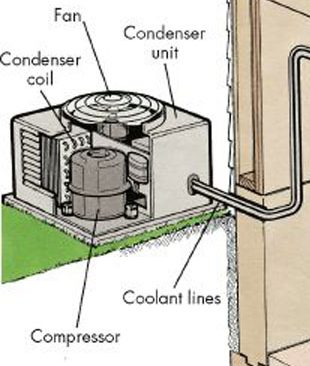
An open system, also called control volume, is a selected region in space. An open system always exchanges mass with its surroundings. It may exchange energy with its surroundings in the form of heat and work, but energy transfer is not a necessary condition for a system to be an open system. In other words, an open system doesn’t have to exchange heat or work with its surroundings at all. Figure 1.2.4 illustrates an open system, which typically encloses a device that involves mass flow through its inlet and outlet. Figure 1.2.5 illustrates the outdoor condensing unit of an air conditioner. It may be treated as an open system because the coolant can enter and leave the condensing unit (the system) via its connecting coolant lines.
If a system doesn’t allow the exchange of mass and energy with its surroundings, it is called an isolated system. An isolated system is an idealized, hypothetical system. In reality, no device is absolutely isolated.
Practice Problems
Media Attributions
- System and Surroundings © Krauss is licensed under a CC BY-SA (Attribution ShareAlike) license
- Piston Cylinder © Israel Urieli adapted by DIANA BAIRAKTAROVA is licensed under a CC BY-SA (Attribution ShareAlike) license
- Open system © Israel Urieli adapted by DIANA BAIRAKTAROVA is licensed under a CC BY-SA (Attribution ShareAlike) license
- Outdoor condensing unit of an air conditioner © Jackie Bese is licensed under a CC BY-SA (Attribution ShareAlike) license
In thermodynamic analysis, the universe is divided into two parts: a system and its surroundings. A system refers to a selected quantity of matter or a region in space.
In thermodynamic analysis, the universe is divided into two parts: a system and its surroundings. Surroundings refer to the rest of the universe outside of the system.
Boundary or system boundary refers to the surface that separates the system and its surroundings.
A closed system is a system of a fixed mass. Mass transfer cannot happen between a closed system and its surroundings.
Open system is also called control volume. It is a selected region in space, which allows mass and energy to transfer across the boundary between the system and its surroundings.
Control volume is also called open system. It is a selected region in space, which allows mass and energy to transfer across the boundary between the system and its surroundings.
An isolated system cannot exchange mass or energy with its surroundings.

