6. Entropy and the Second Law of Thermodynamics
6.7 Specific entropy of a state
6.7.1 Determining the specific entropy of pure substances by using thermodynamic tables
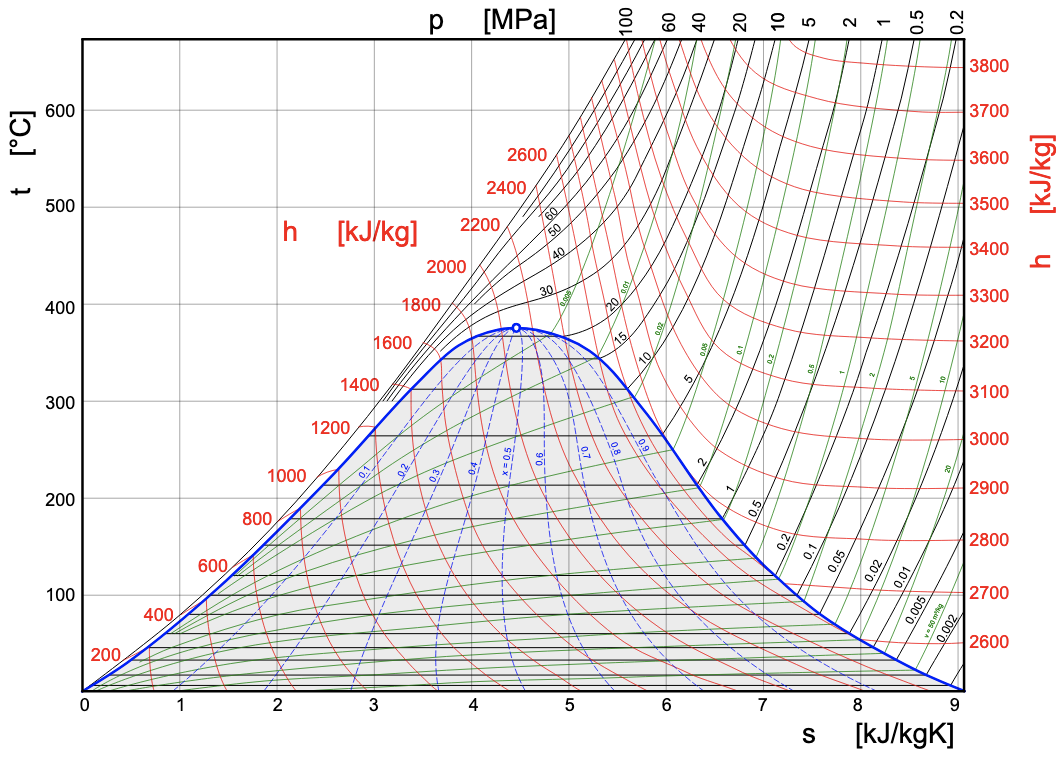
The specific entropy of a pure substance can be found from thermodynamic tables if the tables are available. The procedures are explained in Section 2.4. In addition to the [latex]P-v[/latex] and [latex]T-v[/latex] diagrams, the [latex]T-s[/latex] diagram is commonly used to illustrate the relation between temperature and specific entropy of a pure substance. Figure 6.7.1 shows the [latex]T-s[/latex] diagram for water.
Example 1
Fill in the table.
| Substance | T, oC | P, kPa | v, m3/kg | Quality x | s, kJ/kg-K | Phase |
| Water | 250 | 0.02 | ||||
| R134a | -2 | 100 |
Solution:
Water: T = 250 oC, [latex]v[/latex] = 0.2 m3/kg
From Table A1: T = 250 oC, [latex]v_f[/latex] = 0.001252 m3/kg, [latex]v_g[/latex] = 0.050083 m3/kg
Since [latex]v_f < v < v_g[/latex], water at the given state is a two phase mixture; the saturation pressure is Psat = 3976.17 kPa, and [latex]s_f[/latex] = 2.7935 kJ/kgK, [latex]s_g[/latex] = 6.0721 kJ/kgK
The quality is
[latex]x = \dfrac{v - v_f}{v_{g} - v_{f}} = \dfrac{0.02 - 0.001252}{0.050083 - 0.001252} = 0.383936[/latex]
The specific entropy is
[latex]\begin{align*} s &= s_f + x(s_g - s_f) \\&= 2.7935 + 0.383936 \times (6.0721 -2.7935) = 4.0523 \ \rm{kJ/kgK} \end{align*}[/latex]
R134a: T = -2 oC, P = 100 kPa
From Table C1: by examining the saturation pressures at 0 oC and - 5 oC, we can estimate that the saturation pressure for T = -2 oC is about 270 kPa; therefore, R134a at the given state is a superheated vapour.
From Table C2,
P = 100 kPa, T = -10 oC, [latex]v[/latex]= 0.207433 m3/ kg, [latex]s[/latex] = 1.7986 kJ/kgK
P = 100 kPa, T = 0 oC, [latex]v[/latex]= 0.216303 m3/ kg, [latex]s[/latex] = 1.8288 kJ/kgK
Use linear interpolation to find [latex]v[/latex] and [latex]s[/latex] at T = -2 oC.
[latex]\because \dfrac{v - 0.207433}{0.2160303 - 0.207433} = \dfrac{s - 1.7986}{1.8288 - 1.7986} = \dfrac{-2 - (-10)}{0 - (-10)}[/latex]
[latex]\therefore v = 0.214529 \ \rm{m^3/kg}[/latex] and [latex]s = 1.8228 \ \rm{kJ/kgK}[/latex]
In summary,
| Substance | T
oC |
P
kPa |
v
m3/kg |
Quality x | s
kJ/kg-K |
Phase |
| Water | 250 | 3976.17 | 0.02 | 0.383936 | 4.0523 | two-phase |
| R134a | -2 | 100 | 0.214529 | n.a. | 1.8228 | superheated vapour |
Example 2
A rigid tank contains 3 kg of R134a initially at 0oC, 200 kPa. R134a is now cooled until its temperature drops to -20oC. Determine the change in entropy, [latex]\Delta S[/latex], of R134a during this process. Is [latex]\Delta S=S_{gen}[/latex]?
Solution:
The initial state is at T1 = 0oC and P1 =200 kPa. From Table C2 in Appendix C,
[latex]s_1[/latex] = 1.7654 kJ/kgK, [latex]v_1[/latex] = 0.104811 m3/kg
The tank is rigid; therefore, [latex]v_2[/latex] = [latex]v_1[/latex] = 0.104811 m3/kg.
From Table C1, at T2 = -20oC:
[latex]v_f[/latex] = 0.000736 m3/kg, [latex]v_g[/latex] = 0.147395 m3/kg
[latex]s_f[/latex] = 0.9002 kJ/kgK, [latex]s_g[/latex] = 1.7413 kJ/kgK
Because [latex] v_f < v_2 < v_g[/latex], the final state is a two-phase mixture.
[latex]x_2 = \dfrac{v_2 - v_f}{v_g-v_f} = \dfrac{0.104811 - 0.000736}{0.147395-0.000736}=0.70964[/latex]
[latex]\begin{align*} s_2 &= s_f + x_2 (s_g-s_f) \\&= 0.9002 + 0.70964 \times (1.7413-0.9002)=1.4971 \ \rm{kJ/kgK} \end{align*}[/latex]
The total entropy change is
[latex]\Delta S = m (s_2 - s_1) = 3 \times (1.4971 - 1.7654) = - 0.8049 \ \rm{kJ/K}[/latex]
It is important to note that [latex]\Delta S \neq S_{gen}[/latex] in general. The total entropy of R134a decreases in this cooling process, but the entropy generation is always greater than zero in a real process.
6.7.2 Determining the specific entropy of solids and liquids
The specific entropy of a solid or a liquid depends mainly on the temperature. The change of specific entropy in a process from states 1 to 2 can be calculated as,
[latex]s_2-s_1=C_pln\dfrac{T_2}{T_1}[/latex]
where
[latex]s[/latex]: specific entropy, in kJ/kgK
[latex]C_p[/latex]: specific heat, in kJ/kgK. Note that for solids and liquids, [latex]C_p=C_v[/latex]. Table G2 and Table G3 list the specific heats of selected solids and liquids, respectively.
[latex]T[/latex]: absolute temperature, in Kelvin
6.7.3 Determining the specific entropy of ideal gases
The specific entropy of an ideal gas is a function of both temperature and pressure. Here we will introduce a simplified method for calculating the change of the specific entropy of an ideal gas in a process by assuming constant specific heats. This method is reasonably accurate for a process undergoing a small temperature change.
[latex]s_2-s_1=C_pln\displaystyle\frac{T_2}{T_1}-Rln\frac{P_2}{P_1}[/latex]
[latex]s_2-s_1=C_vln\displaystyle\frac{T_2}{T_1}+Rln\frac{v_2}{v_1}[/latex]
where
[latex]C_p[/latex], [latex]C_v[/latex] and [latex]R[/latex] are the constant-pressure specific heat, constant-volume specific heat, and gas constant, respectively, in kJ/kgK. Table G1 lists these properties of selected ideal gases.
[latex]T[/latex]: absolute temperature, in Kelvin
[latex]P[/latex]: pressure, in kPa
[latex]s[/latex]: specific entropy, in kJ/kgK
[latex]v[/latex]: specific volume, in m3/kg
Example 3
Air is compressed from an initial state of 100 kPa, 27oC to a final state of 600 kPa, 67oC. Treat air as an ideal gas. Calculate the change of specific entropy, [latex]\Delta s[/latex], in this process. Is [latex]\Delta s = s_{gen}[/latex]?
Solution:
From Table G1: Cp = 1.005 kJ/kgK, R = 0.287 kJ/kgK
[latex]\begin{align*} \Delta s &= s_2 - s_1 = C_pln\displaystyle\frac{T_2}{T_1}-Rln\frac{P_2}{P_1} \\&= 1.005 ln\dfrac{273.15 + 67}{273.15 + 27} - 0.287ln\dfrac{600}{100} = -0.3885 \ \rm{kJ/kgK} \end{align*}[/latex]
It is important to note that [latex]\Delta s \neq s_{gen}[/latex] in general. The specific entropy decreases in this process, but the rate of entropy generation is always greater than zero in a real process.
6.7.4 Isentropic relations for an ideal gas
If a process is reversible and adiabatic, it is called an isentropic process and its entropy remains constant. An isentropic process is an idealized process. It is commonly used as a basis for evaluating real processes. The concept of isentropic applies to all substances including ideal gases. The following isentropic relations, however, are ONLY valid for ideal gases.
[latex]Pv^k= \rm{constant}[/latex] and [latex]\displaystyle\frac{P_2}{P_1}=\displaystyle\left(\displaystyle\frac{v_1}{v_2}\right)^k=\left(\frac{T_2}{T_1}\right)^{k/(k-1)}[/latex]
where
[latex]k=\dfrac{C_p}{C_v}[/latex]: specific heat ratio. The [latex]k[/latex] values of selected ideal gases can be found in Table G1.
[latex]T[/latex]: absolute temperature, in Kelvin
[latex]P[/latex]: pressure, in kPa
[latex]v[/latex]: specific volume, in m3/kg
It is noted that the isentropic relation [latex]Pv^k = \rm{constant}[/latex] for ideal gases is actually a special case of the polytropic relation [latex]Pv^n = \rm{constant}[/latex] with [latex]n = k = \dfrac{C_p}{C_v}[/latex].
Example 4
Derive the isentropic relation [latex]Pv^k= \rm{constant}[/latex]
Solution:
For an ideal gas undergoing an isentropic process,
[latex]\Delta s = s_2 - s_1 =C_pln\displaystyle\frac{T_2}{T_1}-Rln\frac{P_2}{P_1}=0[/latex]
Substitute [latex]C_p = \dfrac{kR}{k-1}[/latex] in the above equation and rearrange,
[latex]\because \dfrac{k}{k-1}ln\dfrac{T_2}{T_1} = ln\dfrac{P_2}{P_1}[/latex]
[latex]\therefore ln\left(\dfrac{T_2}{T_1}\right)^{\dfrac{k}{k-1}} = ln\dfrac{P_2}{P_1}[/latex]
[latex]\therefore \dfrac{P_2}{P_1} = \left(\dfrac{T_2}{T_1}\right)^{\dfrac{k}{k-1}}[/latex]
Combine with the ideal gas law, [latex]Pv = RT[/latex],
[latex]\therefore \dfrac{P_2}{P_1} = \left(\dfrac{T_2}{T_1}\right)^{\dfrac{k}{k-1}} = \left(\dfrac{P_{2}v_{2}}{P_{1}v_{1}}\right)^{\dfrac{k}{k-1}}[/latex]
[latex]\therefore \dfrac{P_2}{P_1} = \left(\dfrac{v_1}{v_2}\right)^k[/latex]
[latex]\therefore Pv^k = \rm{constant}[/latex] and [latex]\dfrac{P_2}{P_1} = \left(\dfrac{v_1}{v_2}\right)^k = \left(\dfrac{T_2}{T_1}\right)^{\dfrac{k}{k-1}}[/latex]
Practice Problems
Media Attributions
- T-s diagram for water © Kaboldy is licensed under a CC BY-SA (Attribution ShareAlike) license
An isentropic process refers to a process that is reversible and adiabatic. The entropy remains constant in an isentropic process.

