Neuromuscular Control of Motion
The Neuromuscular Junction and Nervous Control of Skeletal Muscle
J. Gordon Betts; James A. Wise; Kelly A. Young; Eddie Johnson; Brandon Poe; Dean H. Kruse; Oksana Korol; Jody E. Johnson; Mark Womble; and Peter DeSaix
Learning Objectives
By the end of this section, you will be able to:
- Identify the components of the neuromuscular junction.
- Describe excitation-contraction coupling and formation of a power stroke.
- Explain how muscles contract and relax.
- Describe the mechanisms of ATP regeneration for use in muscle contraction.
The Neuromuscular Junction
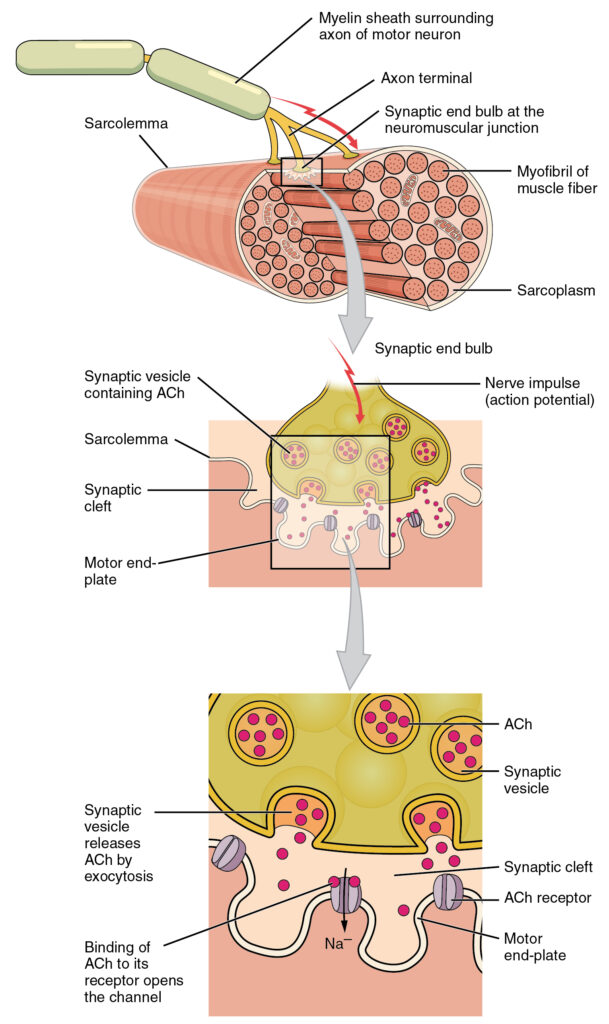
A specialization of the skeletal muscle is the site where a motor neuron’s terminal meets the muscle fiber—called the neuromuscular junction (NMJ). This is where the muscle fiber first responds to electrical signaling by the motor neuron. Every skeletal muscle fiber in every skeletal muscle is innervated by a motor neuron at the NMJ. Excitation signals from the neuron are the only way to functionally activate the fiber to contract.
The sequence of events that result in the contraction of an individual muscle fiber begins with a signal—the neurotransmitter, acetylcholine (ACh)—from the motor neuron innervating that muscle fiber. The sarcolemma, the membrane surrounding the fibers, will depolarize as positively charged sodium ions (Na+) enter. This is where the Excitation-Contraction Coupling begins.
Excitation-Contraction Coupling
All living cells have membrane potentials, or electrical gradients across their membranes. The inside of the membrane is usually around -60 to -90 mV, relative to the outside. This is referred to as a cell’s membrane potential. Neurons and muscle cells can use their membrane potentials to generate electrical signals. They do this by controlling the movement of charged particles, called ions, across their membranes to create electrical currents. This is achieved by opening and closing specialized proteins in the membrane called ion channels. Although the currents generated by ions moving through these channel proteins are very small, they form the basis of both neural signaling and muscle contraction.
Both neurons and skeletal muscle cells are electrically excitable, meaning that they are able to generate action potentials. An action potential is a special type of electrical signal that can travel along a cell membrane as a wave. This allows a signal to be transmitted quickly and faithfully over long distances.
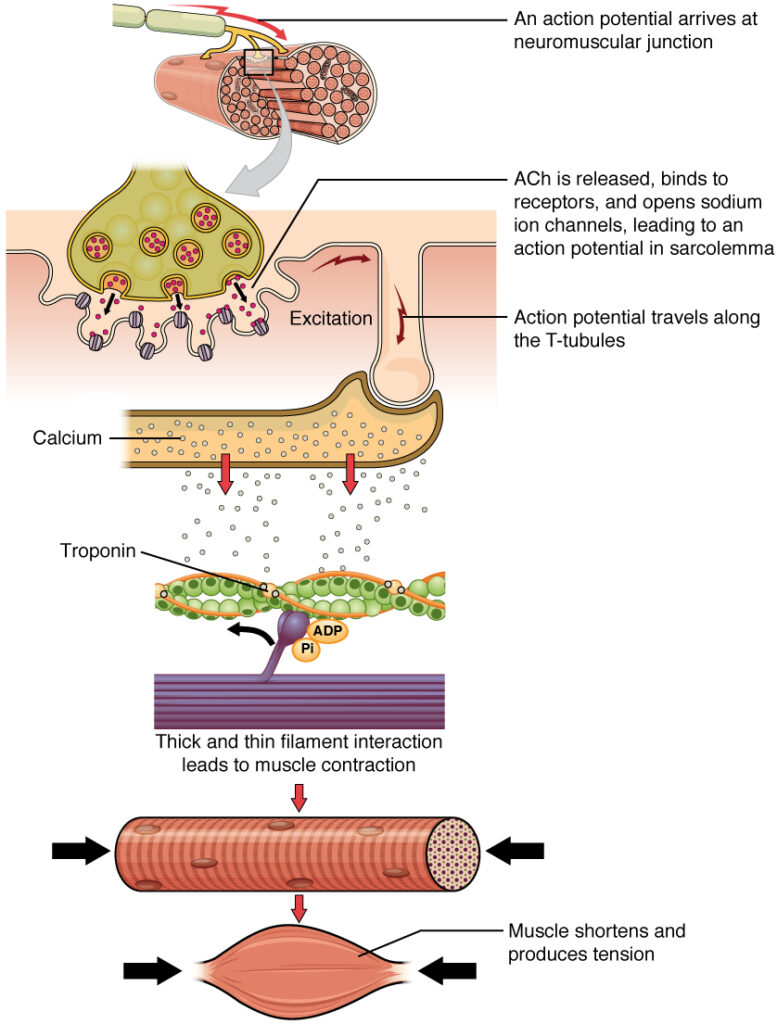
For a skeletal muscle fiber to contract, its membrane must first be “excited”—in other words, it must be stimulated to fire an action potential. The muscle fiber action potential, which sweeps along the sarcolemma as a wave, is “coupled” to the actual contraction through the release of calcium ions (Ca++) from the SR. Once released, the Ca++ interacts with the shielding proteins, forcing them to move aside so that the actin-binding sites are available for attachment by myosin heads. The myosin then pulls the actin filaments toward the center, shortening the muscle fiber.
Signaling begins when a neuronal action potential travels along the axon of a motor neuron, and then along the individual branches to terminate at the NMJ. At the NMJ, the axon terminal releases a chemical messenger, or neurotransmitter, called acetylcholine (ACh). The ACh molecules diffuse across a minute space called the synaptic cleft and bind to ACh receptors located within the motor end-plate of the sarcolemma on the other side of the synapse. Once ACh binds, a channel in the ACh receptor opens and positively charged ions can pass through into the muscle fiber, causing it to depolarize, meaning that the membrane potential of the muscle fiber becomes less negative (closer to zero.)
As the membrane depolarizes, another set of ion channels called voltage-gated sodium channels are triggered to open. Sodium ions enter the muscle fiber, and an action potential rapidly spreads (or “fires”) along the entire membrane to initiate excitation-contraction coupling.
Propagation of an action potential along the sarcolemma is the excitation portion of excitation-contraction coupling. Recall that this excitation actually triggers the release of calcium ions (Ca++) from its storage in the cell’s sarcoplasmic reticulum (SR). For the action potential to reach the membrane of the SR, there are periodic invaginations in the sarcolemma, called T-tubules (“T” stands for “transverse”). You will recall that the diameter of a muscle fiber can be up to 100 μm, so these T-tubules ensure that the membrane can get close to the SR in the sarcoplasm. The arrangement of a T-tubule with the membranes of SR on either side is called a triad (The T-tubule). The triad surrounds the cylindrical structure called a myofibril, which contains actin and myosin.
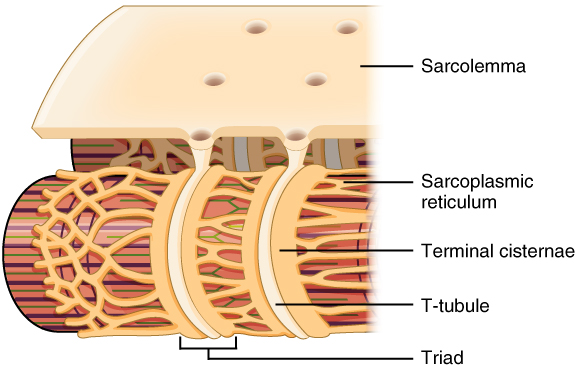
The T-tubules carry the action potential into the interior of the cell, which triggers the opening of calcium channels in the membrane of the adjacent SR, causing Ca++ to diffuse out of the SR and into the sarcoplasm. It is the arrival of Ca++ in the sarcoplasm that initiates contraction of the muscle fiber by its contractile units, or sarcomeres. This sets the stage for the power stroke of the sliding filament theory.
The Sliding Filament Model of Contraction
The sliding filament model of contraction can be used to demonstrate the process of muscle contraction. When signaled by a motor neuron, a skeletal muscle fiber contracts as actin is pulled and then slides past the myosin within the fiber’s sarcomeres. The sliding can only occur when myosin-binding sites on the actin filaments are exposed by a series of steps as explained above.
When a sarcomere contracts, the Z lines, or the junction of actin filaments marking the boundaries of each sarcomere, move closer together. The I bands, or areas of myosin only, becomes smaller. The A band, or areas of both actin and myosin, stays the same width. At full contraction, the actin and myosin filaments overlap completely.
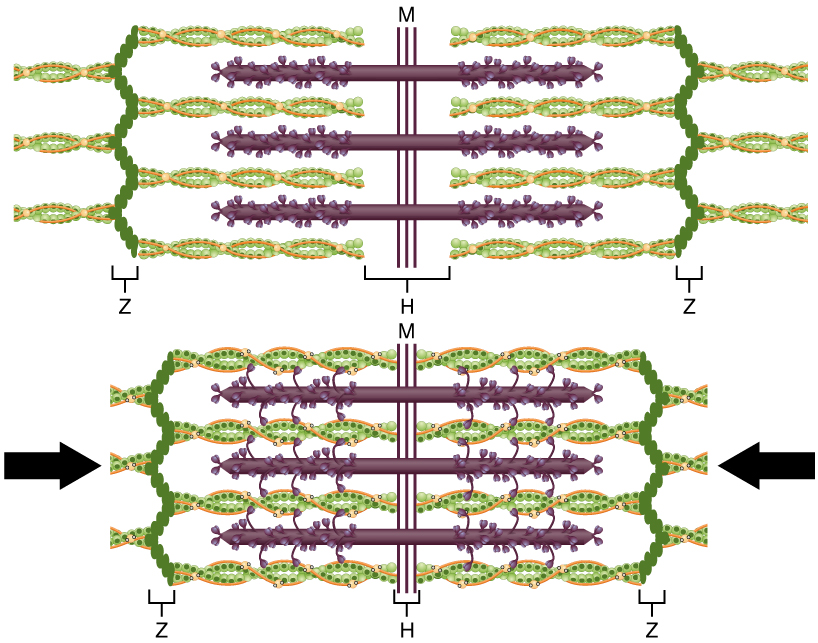
Tropomyosin (a protein that winds around actin and covers actin’s myosin-binding sites) binds to troponin to form a complex. This troponin-tropomyosin complex prevents the myosin “heads” from binding to the active sites on the actin microfilaments. To initiate muscle contraction, tropomyosin has to expose the myosin-binding site on an actin filament. The first step in the process of contraction is for Ca2+ to bind to Ca2+ binding sites on troponin so that tropomyosin can slide away from the binding sites on the actin strands. This allows the myosin heads to bind to these exposed binding sites and form cross-bridges. The actin filaments are then pulled by the myosin heads to slide past the myosin filaments toward the center of the sarcomere. But each head can only pull a very short distance before it has reached its limit and must be “re-cocked” (or reset) before it can pull again, a step that requires energy provided by ATP.
For actin filaments to continue to slide past myosin filaments during muscle contraction, myosin heads must:
- Pull the actin at the binding sites
- Detach
- Re-cock (reset)
- Repeat (attach to more binding sites)
This repeated movement is known as the cross-bridge cycle. This motion of the myosin heads is similar to the oars when an individual rows a boat: The paddle of the oars (the myosin heads) pull, are lifted from the water (detach), repositioned (re-cocked) and then immersed again to pull, as shown in the figure below.
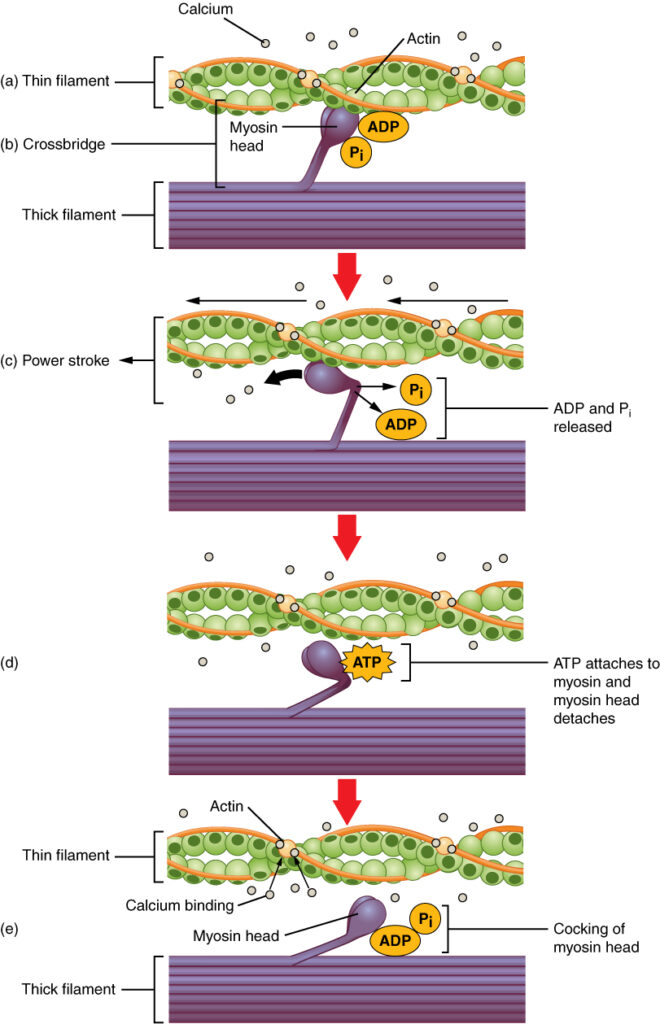
Cross-bridge formation occurs when the myosin head attaches to the actin while adenosine diphosphate (ADP) and inorganic phosphate (Pi) are still bound to myosin (Skeletal Muscle Contraction – a, b). Pi is then released, causing myosin to form a stronger attachment to the actin, after which the myosin head moves toward the M-line, pulling the actin along with it. The filaments move approximately 10 nm toward the M-line. This movement is called the power stroke (Skeletal Muscle Contraction – c). In the absence of ATP, the myosin head will not detach from actin.
One part of the myosin head attaches to the binding site on the actin, but has another binding site for ATP that allows the myosin head to detach from the actin (Skeletal Muscle Contraction – d). After this occurs, ATP is converted to ADP and Pi by the intrinsic ATPase activity of myosin. The energy released during ATP hydrolysis changes the angle of the myosin head into a cocked position (Skeletal Muscle Contraction – e). The myosin head is now in position for further movement.
When the myosin head is cocked, myosin is in a high-energy configuration. This energy is expended as the myosin head moves through the power stroke. At the end of the power stroke, the myosin head is in a low-energy position. After the power stroke, ADP is released; however, the formed cross-bridge is still in place, and actin and myosin are bound together. As long as ATP is available, it readily attaches to myosin, the cross-bridge cycle can recur, and muscle contraction can continue.
Note that each thick filament of roughly 300 myosin molecules has multiple myosin heads, and many cross-bridges form and break continuously during muscle contraction. Multiply this by all of the sarcomeres in one myofibril, all the myofibrils in one muscle fiber, and all of the muscle fibers in one skeletal muscle, and you can understand why so much ATP is needed to keep skeletal muscles working. In fact, it is the loss of ATP that results in the rigor mortis observed soon after someone dies. With no further ATP production possible, there is no ATP available for myosin heads to detach from the myosin-binding sites on actin, so the cross-bridges stay in place, causing the rigidity in the skeletal muscles.
Relaxation of a Skeletal Muscle
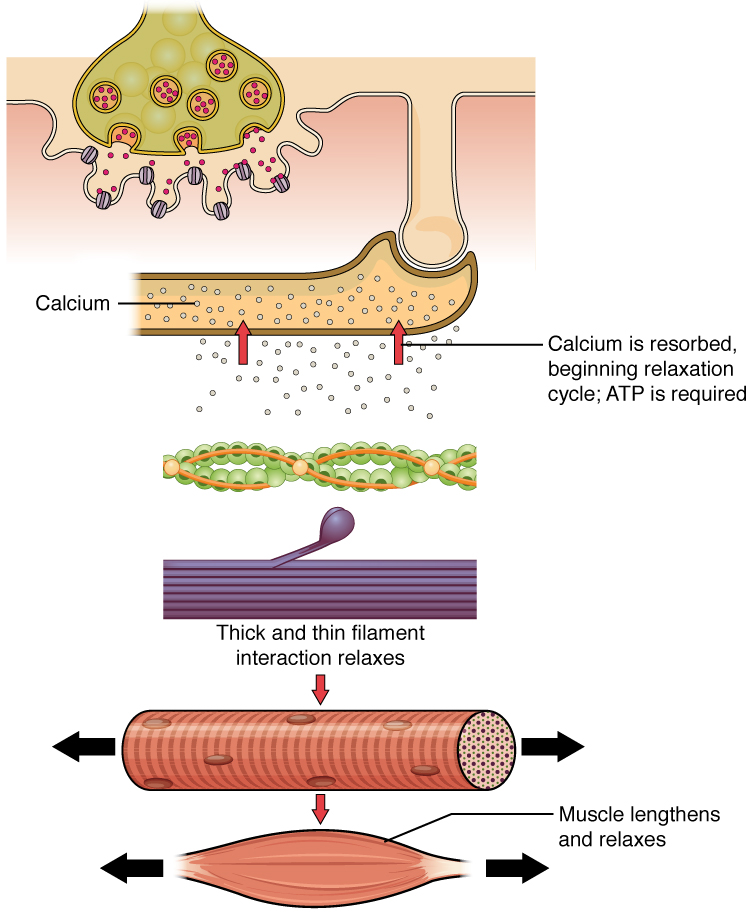
Things happen very quickly in the world of excitable membranes (just think about how quickly you can snap your fingers as soon as you decide to do it). Immediately following depolarization of the membrane, it repolarizes, re-establishing the negative membrane potential. Meanwhile, the ACh in the synaptic cleft is degraded by the enzyme acetylcholinesterase (AChE) so that the ACh cannot rebind to a receptor and reopen its channel, which would cause unwanted extended muscle excitation and contraction.
Muscle contraction usually stops when signaling from the motor neuron ends. This repolarizes the sarcolemma and T-tubules, and closes the voltage-gated Ca2+ channels in the SR. Ca2+ ions are then pumped back into the SR, which causes the tropomyosin to reshield the binding sites on the actin strands. A muscle also can stop contracting when it runs out of ATP and becomes fatigued (Muscle Relaxation).
Thus, this combination of repolarized membranes (in both the motor neuron and sarcolemma) and the lack of Ca+2 available for troponin causes the skeletal muscle to relax and prepare for the next contraction.
Muscle Strength
The number of skeletal muscle fibers in a given muscle is genetically determined and does not change. Muscle strength is directly related to the amount of myofibrils and sarcomeres within each fiber. Factors, such as hormones and stress (and artificial anabolic steroids), acting on the muscle can increase the production of sarcomeres and myofibrils within the muscle fibers. This change is called hypertrophy, which results in the increased mass and bulk in a skeletal muscle. Likewise, decreased use of a skeletal muscle – whether due to inactivity or lack of nervous stimulation – results in atrophy, where the number of sarcomeres and myofibrils disappear (but not the number of muscle fibers).
Disorders of the Muscular System
Duchenne muscular dystrophy (DMD), one of several diseases collectively referred to as “muscular dystrophy,” is a progressive weakening of the skeletal muscles. DMD is caused by a lack of the protein dystrophin, which helps actin bind to the sarcolemma. Without sufficient dystrophin, muscle contractions cause the sarcolemma to tear, causing an influx of Ca2+, leading to cellular damage and muscle fiber degradation. Over time, muscle mass is lost, and greater functional impairments develop.
DMD is an inherited disorder caused by a mutation in the gene that codes for dystrophin in an abnormal X chromosome. It primarily affects males, and it is usually diagnosed in early childhood. DMD usually first appears as difficulty with balance and motion, and then progresses to an inability to walk. It continues progressing upward in the body from the lower extremities to the upper body, where it affects the muscles responsible for breathing and circulation. It ultimately causes death due to respiratory failure, and those afflicted do not usually live past their 20s.
Section Review
Muscle contraction starts with ACh release which prompts the sarcolemma to depolarize (Na+ enters), creating an action potential. Ca2+ is then released from the SR which binds to troponin so that tropomyosin slides away to keep myosin-binding sites exposed. This allows myosin heads to bind to actin for contraction. In the sliding filament model of muscle contraction, actin is pulled and slides past myosin. In a power stroke, phosphate and ADP (hydrolyzed from the ATP used to release the myosin head from the previous contraction cycle) is released, causing the myosin head to have a stronger attachment to actin. The myosin head then moves actin towards the M line (sarcomere center). Myosin detaches from actin when ATP attaches to the myosin head and resets the head for more contraction cycles. Repolarization occurs when ACh has stopped being released and Ca2+ returns to the SR. In the absence of Ca2+, tropomyosin reshields the myosin-binding sites on actin which prevents myosin heads from binding.
Interactive Link Questions
The release of Ca2+ initiates muscle contractions. Watch this video to learn more about the role of Ca2+.
Review Questions
Adaption
This chapter was adapted by Valerie Swanston and Meihua Eva Su from the following texts:
Skeletal Muscle in Anatomy and Physiology by OSCRiceUniversity is licensed under a Creative Commons Attribution 4.0 International License
Unit 6: Muscular System (Module 18) in Anatomy & Physiology I & II by Open Learning Initiative is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License
Media Attributions
- 1009_Motor_End_Plate_and_Innervation © OSCRiceUniversity is licensed under a CC BY (Attribution) license
- 1023_T-tubule © OSCRiceUniversity is licensed under a CC BY (Attribution) license
- 1010a_Contraction © OSCRiceUniversity is licensed under a CC BY (Attribution) license
- 1006_Sliding_Filament_Model_of_Muscle_Contraction © OSCRiceUniversity is licensed under a CC BY (Attribution) license
- 1008_Skeletal_Muscle_Contraction © OSCRiceUniversity is licensed under a CC BY (Attribution) license
- 1010b_Relaxation © OSCRiceUniversity is licensed under a CC BY (Attribution) license

