Chapter 7. Parenteral Medication Administration
7.7 Administering IV Medication via Mini-Bag (Secondary Line) or Continuous Infusion
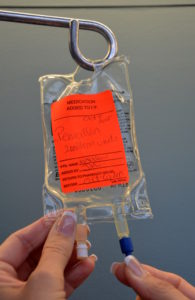
Intravenous intermittent infusion is an infusion of a volume of fluid / medication over a set period of time at prescribed intervals and then stopped until the next dose is required. An intermittent IV medication is also called a piggyback medication, a secondary medication, or a mini-bag medication (see Figure 7.33). Intravenous medications may be given in small volumes of sterile IV solution (25 to 250 ml) and infused over a desired amount of time (given for 30 minutes every four hours) or as a single dose. Many medications must be given slowly to prevent harm to the patient, and this method of administration reduces the risk of rapid infusion. Always check the Parenteral Drug Therapy Manual (PDTM) to ensure the correct guidelines are followed for each specific IV medication. The PDTM provides guidelines on how to mix the IV medication, the amount and type of solution, and the rate of infusion (Perry et al., 2018).
An intermittent medication may be administered by gravity or on an electronic infusion device (EID), also known as an infusion (IV) pump. Many piggyback IV medications must be delivered using an IV pump programmed specifically to that medication. The IV infusion pumps provide hard- and soft-dose limits and safety practice guidelines to aid in safe medication administration (Lynn, 2011). IV medications may also be given by gravity infusion, in which case the health care provider must calculate the infusion rate for drops per minute.
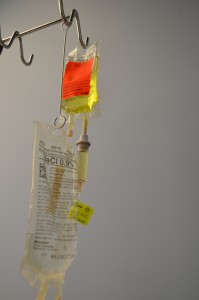
At times, a volume-controlled (intermittent infusion) set may be used to deliver medication for children, older adults, or critically ill patients where fluid volume is a concern. A volume-controlled intermittent set is a small device attached below the primary infusion to regulate the mini-bag. The medication can be added to a small amount of IV solution in the device and administered (Lynn, 2011).
Intravenous medications are always prepared using the SEVEN rights and THREE checks as per agency policy. IV medications have a high-risk of adverse events associated with them. As such nurses must follow specific guidelines when administering IV medications. A PDTM or drug monograph provides additional information including: the generic & brand names, classification of the drug, what routes the medication can be administered, indications for use, contraindications, dosage (age dependent), administration/dilution guidelines, adverse effects, administration guidelines (e.g., specialized monitoring required, must be on an IV pump / controller, given over a specific time frame), compatibility and incompatibility (Alberta Health Services, 2009). Some medications may only be given via a piggyback method; others through large-volume IV solutions; other IV medications require dilution and administration over a specific time frame.
The Institute for Safe Medication Practices (2014) has created a list of high-alert medications that bear the heightened risk of significant harm when they are used in error. Specific safeguards for these medications can be found in the PDTM. It is vital to understand which medications are considered high risk prior to administration. A link to the list of high-alert medications can be found under Suggested Online Resources at the end of this chapter. In addition to the SEVEN rights × 3 for medication preparation, readers are referred back to Table 7.8
Checklist 63 lists the steps to administering an intermittent IV medication by gravity or an IV infusion pump.
Checklist 63: Administering an Intermittent IV Medication by a Mini-Bag (Initial Dose)Disclaimer: Always review and follow your agency policy regarding this specific skill. |
|||
Safety considerations:
|
|||
Steps |
Additional Information |
||
| 1. Prepare one medication for one patient at the correct time as per agency policy.
Always check the physician orders, PDTM, and MAR. Mathematical calculations may be required to determine the correct dose to prepare. |
Always apply the SEVEN rights of medication administration. Review the agency policy and the PDTM. If a medication is a stat, first-time, loading, or one-time dose, be extra diligent in reviewing the PDTM. Memory slips are a common source of error with medication administration.
Complete all assessments (vital signs), and check laboratory values that may influence the medication administration. Premixed bags may require refrigeration. Premixed bags still require the nurse who is administering to add a label with two patient identifiers, nurse initials, and date / time of administration. Some health agencies require a second independent check with high-alert medications. Always follow agency policy. Ensure the mini-bag medication & solution are compatible with the primary solution and any additives. |
||
| 2. Perform hand hygiene and bring medication and MAR to bedside. Create privacy if possible. | Additional equipment required includes secondary tubing, a metal or plastic extension hanger, an alcohol swab, and a timer with a second hand (if running the mini-bag via gravity).
Creating privacy provides comfort to the patient. |
||
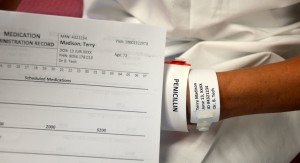
| 3. Compare the MAR with the patient’s wristband, and use two patient identifiers (name and birth date), according to agency policy, to confirm patient ID. | This ensures you have the correct patient and complies with agency standard for patient identification.
 |
||
| 4. Ask about allergies. | This ensures allergy status is correct on the MAR and the patient’s allergy band. | ||
| 5. Discuss purpose, action, and possible side effects of the medication. Provide patient an opportunity to ask questions. Encourage patient to report discomfort at the IV site (pain, swelling, or burning). | Keeping the patient informed of what is being administered helps decrease anxiety. | ||
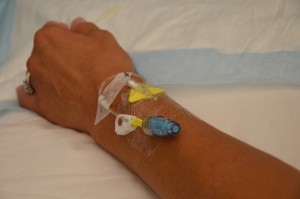
| 6. Assess IV site for patency and other complications (see checklist 65).
Assess for medication specific medication criteria. |
Is the site patent? Ensure IV site is free from redness, swelling, and pain prior to administering the medication.
 IV medications may require assessment of vital signs and lab values prior to administration. |
||
| 7. If required, flush the VAD according to agency protocol. | Flushing protocols contribute to the safety of VADs in particular VADs which remain insitu for an extended period.
Flushing clears the lumen of the VAD to promote patency of the line by reducing residue build up in the lumen |
||
| 7. Select the access port on the primary IV tubing closest to the primary bag. Clean the upper access port with an alcohol swab and friction for 15 seconds. Allow the alcohol to dry. | Secondary tubing is generally short. Accessing a port above the IV pump allows you to use the safety features of the pump in relation to IV medication administration.
 |
||
| 8. Prime secondary tubing.
Luer lock the secondary tubing into the primary tubing. |
Remove secondary tubing from packaging and close the clamp. Hang the medication IV bag on the IV pole.
Remove the sterile blue cap from the IV bag Remove the protective cover of the spike on the IV tubing. Spike the minibag. Fill the drip chamber 1/2 to 1/3 full. Open the roller clamp slowly and prime the tubing Remove the cap on the distal end of the secondary tubing and Luer lock it to the upper access port of the primary tubing. Label the secondary tubing with date and time. Another way to prime the secondary tubing is to spike the mini-bag. Luer lock the secondary tubing into the primary tubing. Lower the mini-bag below the primary bag, open the roller clamp until the drip chamber is 1/2 to 1/3 full. |
||
| 9. Lower the primary IV solution bag, and hang it on the extension hook. | Ensure the secondary bag (mini-bag) is hung above the primary IV solution bag. The position of the IV solutions influence the flow of the IV fluid into the patient. The setup is the same if the medication is given by gravity or through an IV infusion pump. Always follow manufacturer’s directions for infusion pumps.
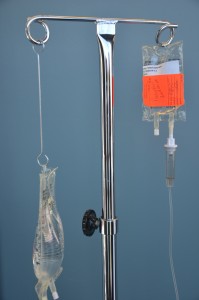 |
||
| 10. Ensure clamp on secondary tubing is open. | This prevents the patient from missing a dose of medication.
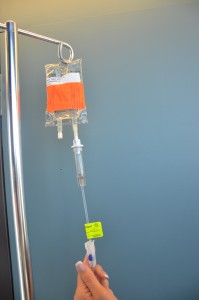 |
||
| Set the infusion rate according to PDTM.
11a. If using gravity infusion, use the roller clamp on the primary set to regulate the rate. The rate will need to be calculated for gtts/mins.
11b. If using an IV infusion pump, program the rate and volume of the secondary infusion. Most infusion pumps automatically restart the primary infusion at the previously established rate. |
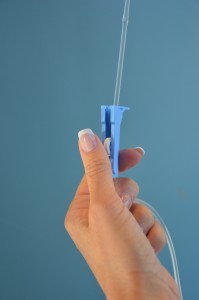 If the medication is administered via gravity, remember to return to the patient when the mini-bag is infused and readjust the rate for the primary IV infusion. The primary IV solution will resume infusing at the rate of the secondary infusion, which could lead to rapid infusion of the primary solution. If medication is being given for the first time, stay with the patient for the first five minutes to monitor for any potential adverse effects. Encourage the patient to notify the health care provider if IV site becomes red, painful, or swollen, or if patient notices any adverse effects from the medication. |
||
| 12. Leave secondary IV mini-bag and tubing in place for future drug administration. Check agency policy to verify if this practice is acceptable. | Repeated changes in IV tubing increase risk for infection transmission. Secondary IV tubing should be changed as per agency policy (usually every 24 hours). | ||
| 13. Perform hand hygiene. | Hand hygiene reduces the transmission of microorganisms. | ||
| 14. Document administration of the IV mini-bag on MAR. If the patient requires monitoring of fluid balance, record on the fluid balance documents (as per agency policy). | Document time, therapeutic effect (if appropriate), and any adverse reactions. Prompt documentation avoids the possibility of accidentally repeating the administration of the drug. If the drug was omitted or refused, record this appropriately and notify the primary health care provider. | ||
| Data source: Berman & Snyder, 2016; Goossens, 2015; Lynn, 2011; Perry et al., 2018; WHO, 2012 | |||
Checklist 63 lists the steps to administer an intermittent IV medication using an existing secondary line, by gravity or an IV infusion pump.
Checklist 63: Administering an Intermittent IV Medication by a Mini-Bag
|
|||
Safety considerations:
|
|||
Steps |
Additional Information |
||
| 1. Prepare one medication for one patient at the correct time as per agency policy.
Always check the physician’s order, PDTM, and MAR. Mathematical calculations may be required to determine the correct dose to prepare. |
Always apply the SEVEN rights and THREE checks of medication administration. Review the agency policy and the PDTM. If a medication is a stat, first-time, loading, or one-time dose, be extra diligent in reviewing the PDTM. Memory slips are a common source of error with medication administration.
Complete all assessments and laboratory values that may influence the medication administration. If piggyback (secondary) medication is made up by the pharmacist, ensure the medication label on the mini-bag includes the medication, date and time the medication was mixed, dose and concentration, and expiry time. Premixed bags may require refrigeration. Premixed bags still require the nurse who is administering to add a label with two patient identifiers, nurse initials, and date / time of administration. Some health agencies require a second independent check with high-alert medications. Always follow agency policy. |
||
| 2. Bring medication and MAR to bedside.
Create privacy if possible. |
Additional equipment required includes secondary tubing, a metal or plastic extension hanger, an alcohol swab, and a timer with a second hand (if running the mini-bag via gravity).
Creating privacy provides comfort to the patient. |
||
| 3. Compare the MAR with the patient’s wristband, and use two patient identifiers (name and birth date), according to agency policy, to confirm patient ID. | This ensures you have the correct patient and complies with agency standard for patient identification.
 |
||
| 4. Ask about allergies. | This ensures allergy status is correct on the MAR and the patient’s allergy band. | ||
| 5. Discuss purpose, action, and possible side effects of the medication. Provide patient an opportunity to ask questions. Encourage patient to report discomfort at the IV site (pain, swelling, or burning). | Keeping patient informed of what is being administered helps decrease anxiety. | ||
| 6. Perform hand hygiene. | Hand hygiene prevents the transmission of microorganisms. | ||
| 7. Assess IV site for patency and other complications (see checklist 65). Assess for medication specific criteria.
Complete necessary assessments as required. Assess IV site for patency. If required flush the VAD according to agency protocol. |
IV medications may require assessment of vital signs and lab values prior to administration. IV site must be patent prior to use.
IV access devices such as PICCS and central lines which remain in situ for long periods have the potential for medication residue to build up in the lumen of the catheter posing a risk for occlusion and reaction with non compatible medications. Turbulent flushing clears the lumen of these. |
||
8. Besides knowing compatibility of the primary solution with the mini-bag medication, ensure the new medication to be hung is compatible with the medication in the previous mini-bag.
Close the clamp on the secondary IV line. Empty the drip chamber into the mini-bag. Remove the old mini-bag from the secondary IV tubing. Hang the new mini-bag onto the IV pole. Remove sterile blue cover on new medication bag, and insert the spike of the secondary IV tubing into it.
|
Check expiration date on secondary IV tubing.
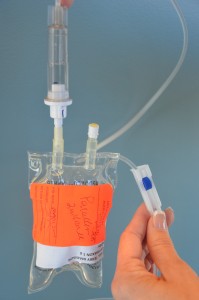 Follow principles of asepsis. Do not contaminate any connections. 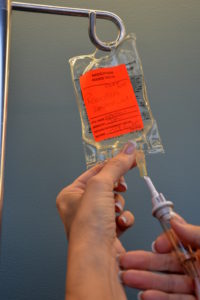  |
||
| 9. Ensure piggyback mini-bag is hung above the primary IV solution bag. | Position of the IV solutions influences the flow of the IV fluid into the patient. The setup is the same if the medication is given by gravity or through an IV infusion pump. Always follow manufacturer’s directions for infusion pumps. | ||
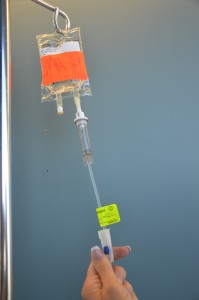
| 10. Ensure clamp on secondary tubing is open. | This prevents the patient from missing a dose of medication.
 |
||
| Set the infusion rate according to PDTM.
11a. If using gravity infusion, use the roller clamp on the primary set to regulate the rate. The rate will need to be calculated for gtts/mins.
11b. If using an IV infusion pump, program the rate and volume of the secondary infusion. Most infusion pumps automatically restart the primary infusion at the previously established rate. |
 If the medication is administered via gravity, remember to return to the patient when the minibag is infused and readjust the rate for the primary IV infusion. The primary IV solution will resume infusing at the rate of the secondary infusion, which could lead to rapid infusion of the primary solution. If medication is being given for the first time, stay with the patient for the first five minutes to monitor for any potential adverse effects. Encourage patient to notify the health care provider if IV site becomes red, painful, or swollen, or if patient notices any adverse effects from the medication. |
||
| 12. Leave IV mini-bag and tubing in place for future drug administration. Check agency policy to verify if this practice is acceptable. | Repeated changes to IV tubing increase risk for infection transmission. Secondary IV tubing should be changed as per agency policy (usually every 24 hours). | ||
| 13. Perform hand hygiene. | Hand hygiene reduces the transmission of microorganisms. | ||
| 14. Document administration of the IV piggyback on MAR, and as per agency policy. | Document time, therapeutic effect (if appropriate), and any adverse reactions. Prompt documentation avoids the possibility of accidentally repeating the administration of the drug. If the drug was omitted or refused, record this appropriately and notify the primary health care provider. | ||
| Data source: Clayton et al., 2010; Lynn, 2011; Perry et al., 2018; WHO, 2012 | |||
Continuous Intravenous (Medication) Infusion
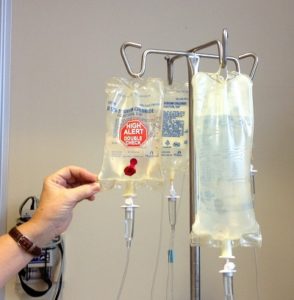
A continuous intravenous medication infusion is the infusion of a parenteral drug over several hours to several days. It involves adding medication to sterile IV solution (100 to 1,000 ml bag), and hanging the IV solution as a primary infusion. A continuous infusion must be ordered by the prescriber and listed in the PDTM as a medication to be given by IV continuous infusion. Examples of continuous IV infusion medications include heparin, KCL, and pantaprazole. Continuous intravenous infusions often come pre-mixed from the pharmacy. They might be labelled with the patient name. They will be labelled with IV solution; volume, amount, and concentration of medication; initials of person who prepared it; and date and time prepared (Alberta Health Services, 2009) or they might be ward stock direct from the manufacturer. Always refer to the PDTM for guidelines on how to administer, regulate, and titrate continuous infusions.
An electronic infusion device (EID) should be used to infuse continuous IV medications and MUST be used for specific medications like IV insulin and high dose KCL. Assessments and lab values must be monitored following the PDTM guidelines. A health care provider must assess the continuous medication infusion for the dose, rate, and patency of the IV site, and assess the patient for therapeutic and adverse reactions to the medication. The Institute for Safe Medication Practices (ISMP) (2013) recommends that all high-alert medications be independently double-checked to detect potential harmful errors before they reach the patient. Independent double checks have been shown to detect up to 95% of errors (ISMP Canada, 2014a).
Critical Thinking Exercises
- Explain your thinking about whether or not the same secondary IV tubing be used more than once and for more than one kind of medication.
- What is the purpose of hanging the secondary (piggyback) IV medication higher than the primary IV solution?
- Your patient has an IV antibiotic infusing via a mini-bag and requests analgesic. The analgesic is IV route and not compatible with the current antibiotic. Explain your next action(s).
Attributions
Figure 7.33 Secondary Medication by BCIT is used under a CC BY 4.0 International license.
Figure 7.34. Untitled from pxhere has been designated to the public domain (CC0).