Chapter 8. Intravenous Therapy
8.5 IV Administration Equipment
Intravenous fluids are administered through thin, flexible plastic tubing called an infusion set or primary infusion tubing/administration set (Perry et al., 2018). The infusion tubing/administration set connects to the bag of IV solution. IVs are then run either by gravity or by an intravenous infusion pump, sometimes referred to as electronic infusion device (EID).
Primary IV tubing is used to infuse continuous or intermittent fluids or medication. It consists of the following parts (see Figure 8.15):
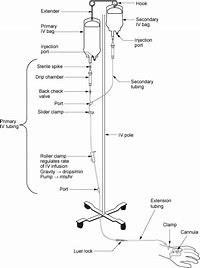
- Sterile spike: Connects the tubing into the IV bag.
- Drip chamber: Used to observe flow of IV fluids and / or to calculate drops per minute.
- Backcheck valve: Prevents fluid or medication from travelling up the IV.
- Access ports: Used to infuse secondary medications and give IV push medications.
- Roller clamp: Used to regulate the speed of, or to stop or start, a gravity infusion.
- Extension set: 10 to 20 cm IV tubing attached to IV cannula. Helps to reduce micro-movements at IV insertion sites and protects from BBF exposure during IV tubing changes.
- Slide clamps: Used to stop the infusion. Are needed to open and close IV infusion pump (a.k.a. EID).
The following table is intended to familiarize you with common IV equipment.
Table 8.7 Common IV Equipment |
|
| Primary IV tubing | IV tubing/administration set that connects to the bag of IV solution. May or may not contain injection ports. See Table 8.9 Frequency of IV Tubing Changes.
Primary IV tubing is either a macro-drip solution administration set that delivers 10, 15, or 20 gtts/ml, or a micro-drip set that delivers 60 drops/ml. Macro-drip sets are used for routine primary infusions. Micro-drip IV tubing is used mostly in pediatric or neonatal care, when small amounts of fluids are to be administered over a long period of time (Perry et al., 2014). The drop factor can be located on the packaging of the IV tubing.  Note: Suppliers provide different kinds of tubing for different purposes. Read the packaging and choose tubing that is appropriate. |
| Secondary tubing | It is shorter in length than primary tubing, with no access ports or backcheck valve. It is connected to a primary line via an access port near the top of the set. It is used to infuse intermittent medications or fluids. Secondary tubing should be changed every 24 hours. |
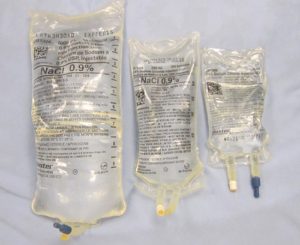
| IV bags | IV solutions come in a variety of solutions, concentrations, and volumes. They are considered medication, and as such the 7 Rights apply. The prescriber will order the IV solution and rate. The nurse monitors for signs of complications related to the solution and IV equipment. IV bags and tubing should have a sticker or label with the date, time, and initials of the healthcare provider marked on them to be valid. IV bags and/or IV tubing should be changed if:
 |
| Extension sets | A.k.a. “add on device,” 10 to 20 cm of IV tubing attached to IV cannula. Helps to reduce micro-movements at IV insertion sites, and protects from BBF exposure during IV tubing changes. Should be added to all PVAD-short and CVADs that do not have permanent extension tubing as part of their structure if tubing changes are expected.
If added at the time of insertion—does not require routine changing—it is considered part of the IV cannula. If added after the insertion, change when integrity is compromised and at each tubing change.  |
| Needleless cap | Added to the distal end of all extension sets of all lumens of VADs to prevent backflow of blood and BBF exposure to healthcare provider. Also added to indwelling subcutaneous devices (butterflies) to allow needleless access when administering medications.
Can be bonded to the extension set or can be separate (add on) to the extension set. Must be sanitized according to agency policy prior to accessing. Can be neutral, negative displacement, and positive pressure. Negative caps are not recommended. Changed q 7 days; each time a site is changed; following blood draws; following blood product administration; when all residual blood cannot be cleared for the device; when contamination is suspected or confirmed. Check agency protocols for correct flushing technique.  |
| IV tubing with filter | Used to filter bacterial particulate and candida. Available with different filter capabilities. Used when administering packed red blood cells, PN, and some medications. Refer to your agency’s parenteral practices guidelines. When administering packed red blood cells, always use a special blood administration set with a filter. |
| Vented set | IV administration set used for solutions that are supplied in glass containers. |
| Volume control set | A type of reservoir that holds a controlled volume of fluid from the IV bag. Limits volume of IV fluids or medications able to infuse into the patient. Often used in pediatrics. IV fluids are attached above the buretrol and refilled manually as the volume decreases.
 |
| Data sources: Interior Health, 2012; Perry et al., 2018; Vancouver Coastal Health, 2008 | |
Frequency of IV Tubing Changes
Primary and secondary administration sets should be changed regularly to minimize risk and prevent infection (CDC, 2017; Fraser Health Authority, 2014). Change IV tubing according to agency policy. Table 8.8 lists the frequency of IV tubing change.
Assessing an IV System
All patients with IV therapy (PVAD-short, midline catheters, and CVADs) are at risk for developing IV-related complications. The assessment of an IV system (including the IV site, tubing, rate, and solution) should take into account the IV administration system AND the patient. Checklist 65 provides general guidelines for assessing an IV system.
Critical Thinking Exercises
- What is the purpose of the back-check valve on primary IV tubing?
- When is it important for the nurse to know the drop factor of IV tubing?
- What is the purpose of extension tubing?
- The nurse has found the patient to have an IV administration set hung 96 hours prior. Explain the necessary next steps.
Attribution
Figure 8.15 IV Primary and Secondary Tubing Setup by BCIT is used under a CC BY-SA 4.0 international license.
Figure 8.16. drop factor by author is used under a CC BY-SA 4.0 international license.
Figure 8.17 Different volumes of IV bags by author is used under a CC BY-SA 4.0 international license.
Figure 8.18 Needleless caps by author is used under a CC BY-SA 4.0 international license.
Figure 8.19 Volume Control Set by author is used under a CC BY-SA 4.0 international license.