Abdomen
Clinical Conditions
CLINICAL CONDITIONS OF THE URINARY SYSTEM
Urinary Tract Infection
Definition:
Most commonly, urinary tract infections (UTIs) occur in sexually active young women, but anyone can get them. There are two types: cystitis and urethritis. Cystitis is an infection of the bladder and urethritis is an infection of the urethra.
Symptoms:
- Burning sensation during micturition
- Increased frequency and urgency of micturition
- Fever
- Cloudy or blood-tinged urine.
- Severe back pain (This is because the infection can travel up to the kidneys)
- Headaches
Causes:
Bacteria from the external genitals can be driven up into the urethra during intercourse. This causes inflammation of the urinary tract.
Treatment & Surgical Intervention:
Typically, UTIs can be treated simply with antibiotics. Most symptoms should go away within a day or two after taking antibiotics.
Renal Calculi
Definition:
More common in men than in women, calicum, magnesium, or uric acid salts can crystallize and precipitate in the calices or renal pelvis of the kidney. This crystallization forms what is known as renal calculi or kidney stones. Most of these kidney stones only form about 5mm in diameter and can pass through the urinary tract without causing serious problems. However, when they get larger, they can cause pain by obstructing ureters or when the contracting ureters contact the sharp edges of the stones during peristalsis. There are 3 regions where the stones frequently get lodged: at the level of L2 (where the renal pelvis thins out into the ureter), at the sacroiliac joint (where the ureter enters the true pelvis), and where the ureters enter the bladder. These regions are very clinically important when searching for kidney stones on X-rays, CT, and ultrasound images.
Symptoms:
- Pain in the lateral and anterior abdominal wall
- Pain in the groin
- Blood in urine or discolored urine
- Fever and chills
- Frequent need to urinate and only urinating in small amounts of urine
- Nausea and vomiting
Causes:
Some causes of renal calculi is dehydration or drinking less than one liter of water a day. Also people aged 20 – 50 are at a higher risk to get them. Some more risk factors are obesity, ethnicity, diet, and family history.
Prevention:
Drinking a lot of water is the main preventer of renal calculi. Eating low amounts of oxalate-rich foods will help lower the risk.
Treatments & Surgical Intervention:
For treatment of kidney stones, the typical go-to method has been to surgically remove them, however a new treatment is taking over, called extracorporeal shock wave therapy, to break up the renal calculi non-invasively. Also drinking more water will increase urine flow which will help get rid of the crystal.
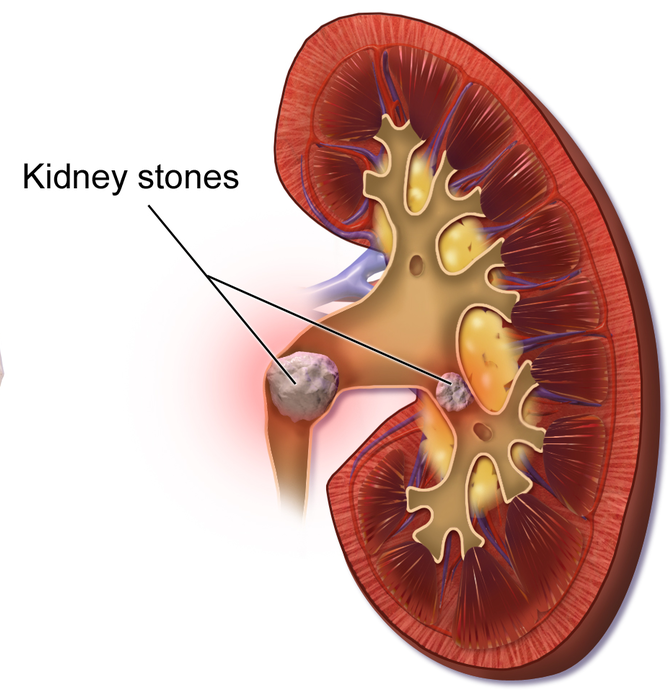
Figure 1. An anterior frontal section of the kidney depicting the renal calculi (kidney stones) that have formed in the renal pelvis, obstructing urine from leaving the kidney and being transported to the urinary bladder.
Cancer of the Bladder
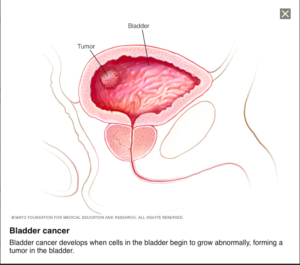
Figure 2. A visualization of a tumor growth in the bladder. (Image retrieved from: https://www.mayoclinic.org/diseases-conditions/bladder-cancer/symptoms-causes/syc-20356104).
Definition:
Bladder cancer involves cells of the epithelial lining. Often it will start in the cells of the urothelium which lines the bladder, ureter, urethra, and renal pelvis. This is call urothelial carcinoma which makes up more than 90% of all bladder cancers.
Symptoms:
- Blood in the urine (Bladder Cancer)
- Pain during urination
- Being unable to urinate
- Loss of appetite and weight loss
- Feeling tired or weak
- Bone pain
Causes:
Bladder cancer has been linked to environmental carcinogens that become deposited in the urine. Some examples of these carcinogens are tars from tobacco, industrial chemicals, and even some artificial sweetener.
Treatment & Surgical Intervention:
Most often, surgical removal of the bladder and chemotherapy are the treatments to control and prevent metastasizing of the cancer cells.
Cancer of the Kidney
Definition:
Kidney cancer begins from the epithelial cells of either the renal tubules or the renal pelvis and calices. The cancer often starts in the tubules. These tubules collect the waste materials and chemicals. This type of cancer is called renal cell carcinoma. It is the more common type of kidney cancer. By the time kidney cancer is detected it has typically already metastasized and survival time is limited to about 12-18 months in most cases
Symptoms:
- Early stages do not have any symptoms
- Late stages
- Blood in urine
- Pain in the back and lateral aspect of the abdomen
- Night sweats
- Weight loss and loss of appetite
Causes:
Some of the factors increasing risk for kidney cancer are obesity, high blood pressure, and possibly a high protein diet
Treatment & Surgical Intervention:
As renal cancers are resistant to traditional chemotherapy and radiation treatment, surgical removal of the entire kidney, its lymph nodes and superior adrenal gland are required.
CLINICAL CONDITIONS OF THE SPLEEN
Spleen Ruptures
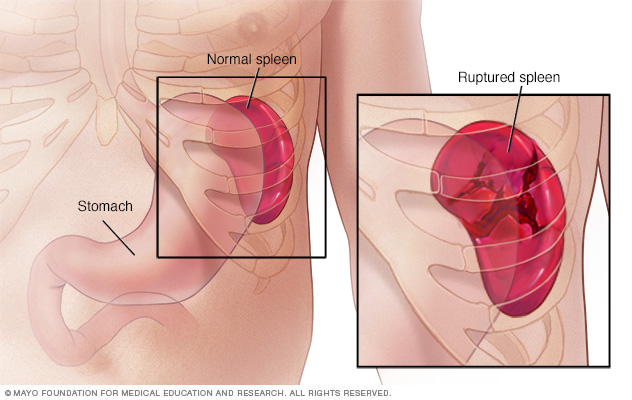
Definition:
A ruptured spleen occurs when there is a break in the spleen. The spleen is located under the rib cage on the left side and its purpose is to aid with infection. Ruptured spleens are common and can be life-threatening as it may internally bleed into the abdominal cavity.
Symptoms:
- Pain and tenderness in upper left abdomen
- Confusion, lightheadedness or dizziness
- Left shoulder pain
Causes:
Ruptured spleens are most often caused by broken ribs often from a sporting mishap, fistfight or car crash. Another cause for a ruptured spleen is when the spleen becomes enlarged. An enlarged spleen occurs when blood gathers in the spleen because of blood cancers, mononucleosis, liver disease, and infections
Treatment & Surgical Intervention:
Treatments for this include removing a portion of the spleen, the whole spleen itself or surgically repairing the spleen.
CLINICAL CONDITIONS OF THE DIGESTIVE SYSTEM AND ACCESSORY ORGANS
Gallstones
Definition:
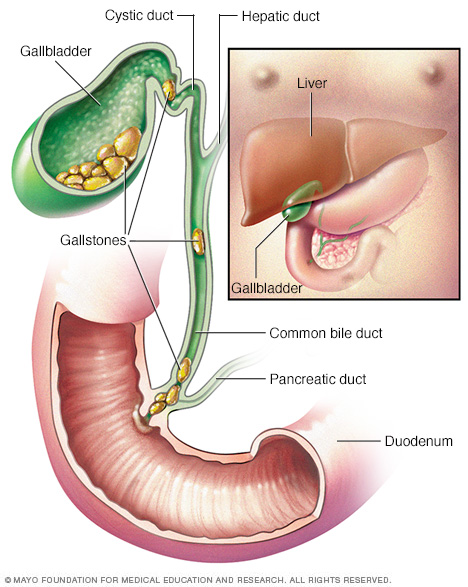
A clinical case involving the gallbladder is a condition called gallstones, gallstones are hard deposits that form when too much cholesterol, bilirubin or bile salts accumulate in the bile. There are two different kinds of gallstones: cholesterol and pigment.
Symptoms:
- Back pain
- Shoulder pain
- Upper right quadrant of the abdomen pain
- Nausea or vomiting
- Yellowing of skin and sclera (whites of eyes)
- Chills and high fever
Causes:
The cholesterol gallstones are mainly made up of cholesterol and small amounts of other substances such as bile salts, these gallstones are usually yellow. Pigment gallstones are composed of bilirubin, this is produced when hemoglobin is broken down inside old red blood cells, these gallstones are brown or black. Gallstones can come just singularly or in multiples and can be the size of a grain of sand to the size of a golf ball.
Treatment & Surgical Intervention:
Small ones that don’t produce symptoms can usually go untreated, while bigger ones usually require surgical removal of the gallbladder. Bigger gallstones can be resolved in two different ways, one is cholecystectomy, (surgical removal of the gallbladder) or oral medications to break down gallstones.
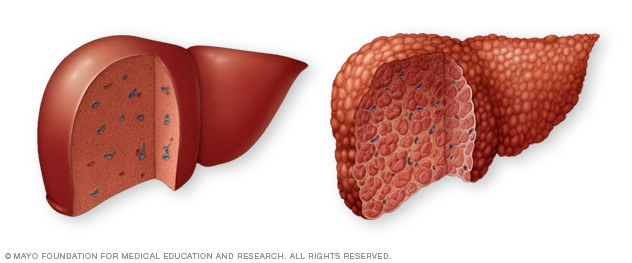
Cirrhosis
Definition:
Since the liver has so many functions it’s very clinically significant, this also means that multiple different problems can arise. Cirrhosis can become an issue after many of these diseases and infections have occurred. Cirrhosis is inflammation of the liver that progressively comes about from scarring caused by different diseases, infections, and conditions. Since the liver is a regenerating organ once these diseases have caused damage to the hepatocytes they try and replace themselves. The connective tissue in the liver replaces faster with scar tissue of the dead hepatocytes than the still living ones can replace themselves. As cirrhosis continues to make its way through the whole liver, killing cells and creating scar tissue it makes it harder for the liver to carry out its functions. Decompensated cirrhosis is when complications arise from the depleting function, this is life-threatening.
Symptoms:
- Fatigue
- Easy bleeding and bruising
- Jaundice (yellow skin and eyes)
- Ascites (fluid accumulation in the abdomen
- Redness in palms
- Testicular atrophy in men
Causes:
Cirrhosis is caused by different diseases, infections, and conditions. Most commonly caused by chronic alcoholism, accumulation on fat in the liver and hepatitis B and C.
Treatment & Surgical Intervention:
Depending on the stage of cirrhosis, treatments focus on preventing further damage to the liver. Treatments for prevention of further liver damage are cutting out alcohol, weight loss for fat accumulation, prescriptions for hepatitis and other underlying causes. Liver transplants in late stages of cirrhosis may be the only option, but patients may only be candidates depending on their current health.
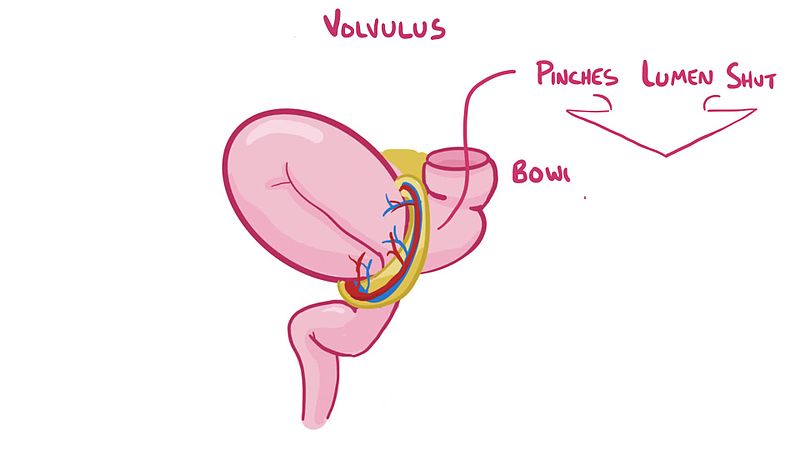
Volvulus
Definition:
Though the mesentery only really plays the role of housing blood vessels, nerves lymph and being a suspending structure, it does have a potentially fatal clinical case connected to it. Volvulus is a condition when a portion of the intestine wraps itself and mesentery around itself.
Symptoms:
- Abdominal tenderness
- Nausea
- Vomiting bile looking material
- Bloody stool
- Trouble passing stool
Causes:
This is usually caused by birth defect where the bowels have irregular attachments to the abdominal wall, so it’s commonly found in children by the age of one. This causes the bowel movements to be blocked and can block blood supply to the intestine if it is twisted tight enough causing bowel infarction. Bowel infarction is an irreversible condition that can result in death.
Treatment & Surgical Intervention:
The only treatment is once volvulus is identified a laparotomy (incision involving the abdominal region) to avoid further complications needs to be done as soon as possible and the bowels are untwisted.
Omental Cysts
Definition:
Related conditions to the greater omentum are quite limited compared to the other accessory organs. Omental cysts are one of the more commonly reported diseases, these cysts are filled with fluid.
Symptoms:
- Infection
- Bleeding
- Volvulus
- Ascites (Fluid accumulation in the abdominal region)
- Rupture of the cyst
Causes:
Omental cysts occurs when the lymphatic channels experience obstruction hindering their ability to drain.
Treatment & Surgical Intervention:
The only treatment of these cysts is surgical removal.
Laryngopharngeal Reflux
Description:
Laryngopharngeal reflux (LPR), also commonly referred to as extraesophageal reflux disease (EERD) or silent reflux, is a form of gastroesophageal reflux disease (GERD) that is characterized by the backflow of gastric contents (refluxate) up the esophagus into the larynx, orthopharynx, and nasopharnyx as a result of a loss in function in the upper and lower sphincters located at the proximal and distal aspects of the esophagus respectively. Exposure to this acidic refluxate can cause chemical damage to tissue, mediate an inflammatory response, and stimulate the development of a number of diseases associated with the upper aerodigestive tract.
Symptoms:
- Hoarseness
- Post-nasal drip (drainage of mucus down the back of the throat)
- Globus sensation (feeling of having a “lumpy structure” embedded in the throat)
- Persistent cough and frequent need to clear throat
- Irritation and a burning sensation in the throat
- Dysphasia (difficulty swallowing)
Mechanism & Diagnosis:
Upon the ingestion of food, a number of anatomical structures systematically work in conjunction with one another to promote the proper travel of the food to the stomach for chemical and mechanical breakdown, and to prevent any potential obstructions in respiratory function. As mentioned earlier, the pharynx plays an important role in connecting the mouth and nasal cavity with the rest of the digestive and respiratory pathways. At the laryngopharynx, which is the most inferior region of the pharynx, the junction between the esophagus and larynx is formed. In this region, a leaf-shaped cartilaginous structure known as the epiglottis is responsible for preventing the entrance of food and fluids into the lower respiratory pathways. When food, liquid, pharyngeal secretion, or other large molecules breach the laryngeal inlet, it is referred to as pulmonary aspiration. A pharyngeal (“gag”) reflex will be initiated to help clear any foreign substances that breach the pharyngeal inlet. Upon swallowing, the larynx is pulled superiorly through the elevation of the hyoid bone, and the epiglottis will slant inferiorly to protect the laryngeal inlet. Food that meets with the esophagus, which is a muscular tube lined with a layer stratified squamous epithelium mucosa, will travel through superior and inferior esophageal sphincters before collecting in the stomach for chemical and mechanical breakdown via churning and the release of hydrochloric acid (HCL) and pepsin. Functionally, the rhythmical contraction and relaxation of these sphincters will permit or prevent the entrance of food or liquid into the esophagus and stomach.
The larynx, also commonly referred to as the voice box, is an upper respiratory region that is composed of a complex cartilaginous foundation held together by a number of membranes and ligaments. Functionally, the larynx is responsible for: producing sound through coordinated vibrations initiated in the vocal cords, protecting of the lower respiratory pathways and promoting the proper distribution of food, liquid, and air into their intended pathways through the epiglottis acting as a regulation mechanism, and allowing the entrance and exit of gases in and out of the lower respiratory pathways for proper respiratory function.
Under certain circumstances, the inferior and superior esophageal sphincters can experience a loss in function in response to structural alterations and weakness. Causes of these alterations exhibited among the esophageal sphincters can include: esophageal motility disorders, such as achalasia, physiological and/or anatomical defects, such as possessing an underdeveloped lower esophageal sphincter, pregnancy, and obesity. In individuals with inactive or weak diaphragmatic muscle fibers, the esophageal hiatus that connects the esophagus to the thoracic and abdominal cavities can become enlarged. This can cause the stomach to translate superiorly, hindering the diaphragms ability to support the function of the cardiac sphincter. As a result, the cardiac sphincter can become structurally and functionally impaired. Once the structural and functional integrity of this sphincter is compromised, stomach acid then gains the ability to reflux up the esophagus and come into contact with many of the delicate structures encompassed in the upper aerodigestive tract, such as the nasal cavity, laryngopharnyx, and vocal cords. Variables that can increase the frequency and severity of this response include: a reduction in gastric emptying, a highly acidic diet, and the gravitation forces utilized via lying in a supine or prone position. Typically, the structural integrity of the esophagus is preserved through the production of a thick layer of mucous, prostaglandins, esophageal growth factors, and a bicarbonate coating provided by swallowed saliva. But when the esophageal lining is frequently exposed to stomach acid, a gradual breakdown in the epithelial lining can occur. This microscopic breakdown can eventually manifest into the development of ulcers in the lower esophageal lining, commonly referred to as hiatal hernias. This condition is referred to as Barrett’s esophagus, and can lead to the development of various esophageal cancers, such as adenocarcinoma. In addition to structural damage, the gastric contents that travel up the esophagus can also create a burning sensation in the thoracic region. This is commonly referred to as pyrosis, or heartburn. Many of the other structures that lie superiorly and posteriorly to the esophagus are lined with an even more sensitive and delicate epithelium compared to the esophagus, such as the larynx, as they aren’t anatomically designed to have prolonged contact with highly acidic compounds. If these delicate regions do happen to come into contact with acid compounds during gastric reflux, they can become subject to substantial chemical damage and irritation. An enzyme known as carbonic anhydrase isoenzyme (CA-III) is typically released when this region is exposed to acidic compounds. This enzyme facilitates the production of bicarbonate, which is effective at neutralizing the pH of acidic compounds in the body. When referring to pepsin, which is an endopeptidease commonly found in stomach acid, it stimulates a reduction in the release of CA-III and mucous. As a result of this, many of the delicate regions encompassed in the upper aerodigestive tract are prone to a higher degree of structural damage, irritation, and inflammation when exposed to the acidic properties of the pepsin and HCL. When referring to the larynx specifically, the onset of an inflammatory response is commonly referred to as laryngitis. Besides structural damage and irritation provided by acidic deterioration, the literature also proposes the theory of H.pylori, a type of bacterium commonly found in the stomach and intestinal tract, being responsible for infecting the tissue of the larynx.
When referring to the diagnostic process for LPR, many practitioners utilize a number of evidence-based testing modalities to check for inflammation in the upper aerodigestive tract. These modalities can include: the barium swallow test, a fibroptic laryngoscopy of the pharynx, larynx, and esophgus, and a double-probe or immunoassay pH measurement of the esophagus. The barium swallow test involves the ingestion of a chalky white substance known as barium. This substance is typically mixed with water, and when ingested, it will adhere to the walls of the esophagus, stomach, and initial portion of the small intestine. This coating has the ability to absorb x-rays, allowing doctors to monitor the movement of the upper GI tract during ingestion of food and liquid. Fiberoptic laryngoscopy involves the insertion of a small, flexible tube-like structure equipped with a camera and light into one of the nostrils to monitor the state of the larynx. The double-probe pH measurement test involves the insertion of two probes that possess pH measuring capabilities into the proximal and distal ends of the esophagus via manometeric or endoscopic assistance. The proximal probe is typically oriented 2cm below the upper esophageal sphincter, and the distal probe is typically oriented 5cm above the lower esophageal sphincter. Together, these probes work collectively to measure the pH along the length of the esophagus over a 24-hour interval. A visual representation of the double-probe pH measurement test is provided in Figure 7. This approach is considered to be the gold standard for detecting LPR in patients. Esophageal pH can also be measured through the detection of pepsin in respiratory sputum, which is a mixture of saliva and mucus, via the usage of an enzyme-linked immunosorbent assay (ELISA). According to the literature, the preferred approach for diagnosing LPR consists of utilizing a fibroptic laryngoscopy as a primary test coupled with a double-probe pH measurement as an adjunct test.
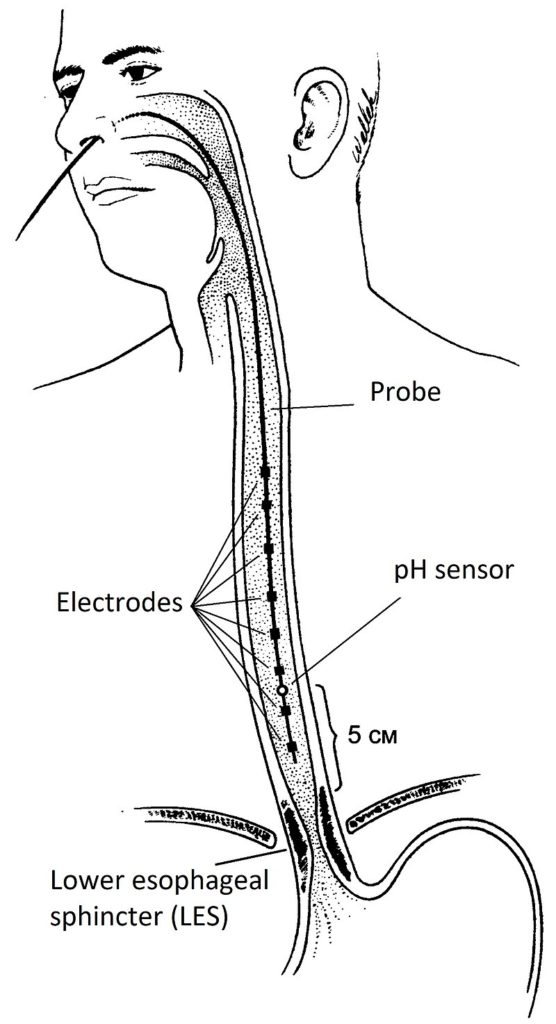
Figure 7. A visual representation of the various equipment and structures utilized during a double-probe pH measurement test. (Image retrieved from: https://en.wikipedia.org/wiki/Impedance%E2%80%93pH_monitoring).
Treatment & Surgical Intervention
With regards to treating LPR, the first steps that should be accounted for are changes in lifestyle and diet. Dietary changes can include: reducing one’s consumption of highly acidic foods such as lemons, oranges, sauerkraut, and tomatoes, avoiding overfilling of the stomach through reducing the size of meals, reducing carbohydrate intake, and refraining from the usage of certain drugs, such as cigarettes and alcohol. Practicing these small dietary changes can play an important role in reducing gastric pH and the volume of gastric acid present in the stomach during a given bout of gastric breakdown, and can reduce the concentration of subcutaneous and visceral fat in the abdominal region through stimulating a metabolic transition from carbohydrates to fats as a primary energy source. Changes in lifestyle can include: engaging in regular aerobic activity, refraining from wearing any clothes or equipment that would constrict the abdominal region, and elevating the neck during sleep. Together, these small changes in lifestyle can reduce additional constriction around the abdomen and stomach by relieving pressure exhibited by visceral fat, subcutaneous fat, and external forces. Elevating the neck during sleep will help reduce the impact that gravitational forces have on promoting the migration of stomach acid towards the lower esophageal sphincter while the body is in a prone or supine position.
Anti-acid medications, such as histamine H2 receptor antagonists and proton pump inhibitors (PPIs), are also commonly used when treating LPR. Some examples of histamine H2 receptor blockers include: nizatidine, famotidine, and ranitidine. Some examples of PPIs include: omeprazole, lansoprazole, and pantoprazole. Together, these medications will help improve symptoms associated with LPR and related conditions by reducing HCL production in the walls of the stomach. According to the literature, the preferred therapeutic algorithm exhibited among most otolaryngologists and gastroenterologists involves a primary line of treatment consisting of dietary alterations, PPIs, and gastroprokinetic, and a secondary line of H2 receptor blockers if the patient isn’t exhibiting any symptomatic improvement when exposed to the initial treatment over an extended period of time. Although the treatment modalities stated above can all play a role in alleviating the symptoms associated with LPR in some patients, they don’t actually restore the function of the lower esophageal valve. In this situation, fundoplication surgery is considered to be an effective option for restoring proper function to the lower esophageal sphincter. Fundoplication surgery involves otolaryngologists and/or gastroenterologists wrapping and securing the superior curve of the fundus around the distal end of the esophagus via surgical stitching and incisions made to the abdomen. As a result of this procedure, the lower esophageal sphincter will regain its intended function through contractile reinforcement provided by the stomach muscles, preventing the reflux of gastric contents into the esophagus. A visual representation of the fundoplication procedure is provided in Figure 8.
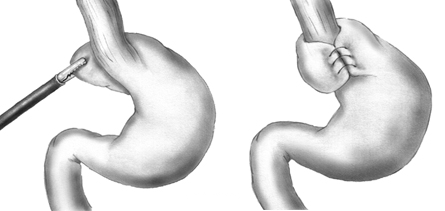
Figure 8. A visual representation of the fundoplication surgical procedure. (Image retrieved from: https://commons.wikimedia.org/w/index.php?curid=10492233).
Peritonitis
Description
Peritonitis is a particular clinical condition that is characterized by inflammation in the peritoneum of the abdominopelvic cavity as a result of being breached by a number of pathogenic invaders, such as bacteria and fungi.
Symptoms
- Referred pain and tenderness originating in the abdominopelvic region
- Distention of the abdomen (the sensation of feeling bloated or enlarged in the abdomen)
- Fever, nausea, and vomiting
- Anorexia (loss of appetite)
- Frequent bouts of diarrhea and constipation
- Minimal urinary output (increased neurological need to ingest fluid, but a reduction in urinary excretions)
- Fatigue and bodily weakness
- Increased tone of the anterolateral abdominal wall
Mechanism & Diagnosis
Similar to most bodily cavities, the abdominopelvic cavity is lined with delicate serous membrane. The outer component, known as the parietal peritoneum, is responsible for lining the entirety of the inner walls in the abdominopelvic cavity. The inner component, known as the visceral peritoneum, covers all the delicate organs that reside in the abdominoplevic region, such as the stomach, spleen, liver, small and large intestines, and the pancreas. Separating these two membranes is a potential space known as the peritoneal cavity. Similar to the pleura of the lungs, the peritoneum is responsible for secreting serous fluid that lubricates the parietal and visceral surfaces of the cavity, allowing the internal organs to slide past one another and the internal walls of the cavity with minimal friction during digestive movements. Some of the structures that are responsible for reinforcing the internal organs to the parietal peritoneum of the cavity include: the mensenteries, omentum, and the ligaments that have structural connections with many of the organs in the abdominopelvic region.
In certain situations, the peritoneal lining can become subject to infection. This is commonly referred to as peritonitis. According to the literature, the predominant etiology of peritonitis is bacterial agents. Some of the more common bacteria include: escherichia coli, klebsiella pneumoniae and pneumococci. When bacterial infection is determined to be the etiology (cause), the condition is deemed as spontaneous bacterial peritonitis (SBP). The onset of SBP is initiated by a number of different ways. The first method in which bacteria breach the peritoneal cavity is via entrance through perforation in the abdominal region as a result of trauma. Similarly, poorly administered surgeries that target the abdominal region can also provide bacteria with entrance into the peritoneal tissue. Another method of infection is through the formation of stomach ulcers in the gastric lining as a result of over-stimulated HCL production in the gastric glands of the stomach. This is typically caused by substantial bodily distress, such as extended exposure to high-stress environments, or input provided by H.pylori bacteria in the stomach, providing the H. pylori bacteria, which are a common bacterium that resides in gastric juices, with excess to the peritoneal cavity. In individuals that have compromised liver and kidney function, which is often exhibited in those with cirrhosis and renal disease, an excess production of peritoneal fluid can occur. This excess production can impair the body’s ability to regulate the fluid, creating ideal conditions for bacterial growth and infection. This over-accumulation of fluid is referred to as ascites. According to the literature, the most common modality of bacterial infection in the peritoneal tissue is through the act of the appendix rupturing and spilling its internal components into the peritoneal cavity as a result of intrinsic infection. Since the appendix typically holds an accumulation of bodily waste products that collect in the appendiceal lumen, which is the area inside of the appendix, these infectious components will then be spread (generalize) throughout the peritoneal cavity, providing bacteria with an opportunity to infect the peritoneum. Similar to appendicitis, diverticulitis, which involves the infection and eventual rupture of small pouches in the digestive tract, can also leech bacteria into the peritoneal cavity. A visual representation of the infection occurring in the intestinal pouches during diverticulitis is provided in Figure 9.
With regards to the diagnostic process, there are a numbers of methods for detecting peritonitis. Administering blood tests are among one of the more common diagnostic processes for peritonitis. The extraction and examination of blood culture allows practitioners to detect any abnormalities that are indicative of peritonitis, such as high levels or white blood cells and bacteria in the bloodstream. If gastrointestinal perforations, such as stomach ulcers, are the suspected cause of the condition, the usage of imaging instruments, such as CT scans, x-rays, and ultrasounds, will provide the practitioners with an accurate 3-D image of any abnormalities. If ascites is the suspected cause of the condition, the insertion of a thin needle into the peritoneal cavity and the extraction of peritoneal fluid through the process of paracentesis, allows the practitioners to provide a thorough analysis of the fluid. Similar to the blood samples, elevated levels of white blood cells and bacteria in the fluid would be indicative of infection. A visual representation of the paracentesis procedure is provided in Figure 10.
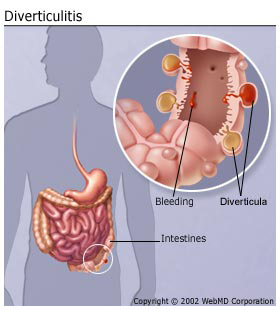
Figure 9. A visual representation of the infected intestinal pouches in an individual with diverticulitis. (Image retrieved from: https://www.webmd.com/digestive-disorders/understanding-diverticulitis-basics).
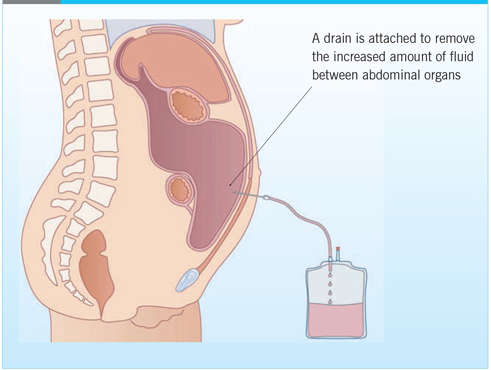
Figure 10. A visual representation of the paracentesis procedure in an individual with ascites. (Image retrieved from: https://journals.rcni.com/cancer-nursing-practice/developing-a-nurseled-daycase-abdominal-paracentesis-service-cnp2013.06.12.5.14.e942).
Treatment
Since peritonitis is almost exclusively caused by bacterial agents, the prescription of antibiotics coupled with medication to help alleviate the symptoms, such as non-steroidal anti-inflammatory drugs (NSAIDs) and corticosteroids, would act as an effective option for combating bacterial invaders. Some of the more commonly utilized antibiotics include: cephalosporins, amoxicillin, and quinolones. According to the research, cephalosporins are the more preferred medication among practitioners due to providing a higher degree of effectiveness and lower risk of nephrotoxicity (poisoning of kidneys) compared to other substances. According to a study conducted by Runyon, Akriviadis and Sattler (1991), cefotaxime, a form of cephalosporin, was shown to be capable of completely sterilizing infected peritoneal fluid in 94% of users following the initial dosage. The overall effectiveness of this drug is attributed to its ability to locate and penetrate ascetic fluid effectively.
Acute Appendicitis
Description:
Acute Appendicitis is a particular medical condition that is characterized by the onset of an acute inflammatory response in the vermiform appendix due to a blockage in the appendiceal lumen. As a result of this blockage, intraluminal pressure will begin to rise. If left untreated, this accumulation in pressure can cause the vermiform appendix to perforate, followed by an eventual rupture, causing its contents to spread and infect other regions associated with the peritoneal cavity.
Symptoms:
- An initial sharp, spontaneous painful sensation in the umbilical or epigastric region
- Eventual migration of the pain to the right lower quadrant of the abdomen, followed by an exacerbation in the perceived level of pain
- Additional pain produced during coughing, walking, and other movements
- Nausea, vomiting, and loss of appetite
- The onset of a low-grade fever as the condition progresses
- Varied feelings of abdominal distension, constipation, and diarrhea
Mechanism & Diagnosis:
Following the majority of the nutrient absorption that occurs in the small intestine, the remaining intestinal contents are passed over to the large intestine. This transition occurs between the terminal end of the ileum and cecum at the ileocecal junction. The cecum, which is a pouch-like structure that resides in the right iliac fossa, is separated from the terminal ileum via the ileocecal valve. Based on the amount of chyme present in the cecum, the sphincter will contract or relax to allow for the proper passageway of intestinal contents and prevent refluxate from re-entering the small intestine. Travelling superiorly, the cecum is continuous with the ascending colon.
Inferior to the ileocecal junction on the posteromedial wall of the cecum, a small, worm-like structure, known as the vermiform appendix exists. The vermiform appendix contains a vast collection of lymphoid follicles. Although there is no current research to support the function of this structure, many theories propose that one of its primary functions is to contribute to the immunological response produced by the body through the detection of antigens, neutralization of pathogenic invaders through the recruitment of white blood cells, and development of immunity through the synthesis of B and T lymphocytes. Another paradigm suggests that the appendix is an area of synthesis and storage for probiotics. In a situation where the bacterial composition of the intestinal tract is greatly altered (i.e., antibiotic usage), the appendix is theorized to release these stored probiotics into the intestinal tract to help re-establish proper gut flora populations.
In certain situations, the lumen that connects the internal environment of the appendix with the cecum can become blocked. Upon a blockage of the appendiceal lumen, an inflammatory response can be induced in the appendiceal wall. This is commonly referred to as acute appendicitis. A visual representation of the inflammatory response that occurs in the vermiform appendix during acute appendicitis is provided in Figure 11. The onset of inflammation will increase the intraluminal pressure, which will consequently lead to excess venous hypertension in the vessels associated with the structure. As lymphatic and venous drainage is compromised, thrombosis (clotting of blood) may occur in the appendicular artery, which would consequently cause the appendiceal tissue to become hypoxic (oxygen deficient). Without adequate blood flow to the region, ischemia (insufficient blood flow) can occur in the appendiceal mucosa. This will eventual manifest into necrosis and gangrene, or death of the tissue, which would deteriorate the structural integrity of the mucosa barrier protecting the appendix, making the structure prone to the infection and proliferation of intraluminal bacteria. As the appendiceal function is completely compromised, the mucus secretions and fecal matter that are usually cycled in and out of the appendix begin to accumulate. When the intraluminal pressure become too great, perforation will occur in the appendiceal wall, followed by a complete rupture in the appendiceal wall. Following a rupture of the appendix, the infectious contents that were accumulating within the appendix are then generalized throughout the peritoneal cavity, exponentially increasing one’s likelihood of developing peritonitis. Other outcomes associated with generalized appendiceal contents include: increased abdominopelvic pain, vomiting and nausea, and increased rigidity in the abdominal muscles.
There are a number of theorized etiologies (causes) for acute appendicitis. Some of these include: faecaliths, or stony masses that form in fecal matter, lymphoid hyperplasia, foreign bodies, such as parasites, and the presence of primary and metastatic tumours. According to the literature, lymphatic hyperplasia in response to exposure to a number of pathogenic agents, such as salmonella and shigella, is the most prevalent etiology for acute appendicitis in younger populations. In older populations, the development and blockage of faecaliths in the appendiceal lumen has shown to be the most prevalent etiology for acute appendicitis in older populations.
With regards to the diagnostic process, detecting appendicitis can be incredibly difficult as there are no current concrete tests in existence beyond the physical examination of the patient. In addition to this, the symptoms associated with acute appendicitis vary greatly among individuals. Although this is the case, there are a number of physical examination practices that can be utilized to help develop insight into whether or not someone has appendicitis. Simple practices such as: palpating the lower right quadrant of the abdominal region to see if the pain worsens upon compression and checking for rigidity in abdominal muscles, which could be indicative of muscular guarding that would occur in response to the pain experienced deep to the muscles, can be effective measures. The usage of blood and urine tests can also be used to provide insight into any potential abnormalities in the blood stream, such as a high white blood cell count, which would be indicative of infection. The usage of various imagining modalities, such as x-rays and CT scans can also be used, which would allow the practitioners to detect inflammation and other structural abnormalities associated with the appendix.
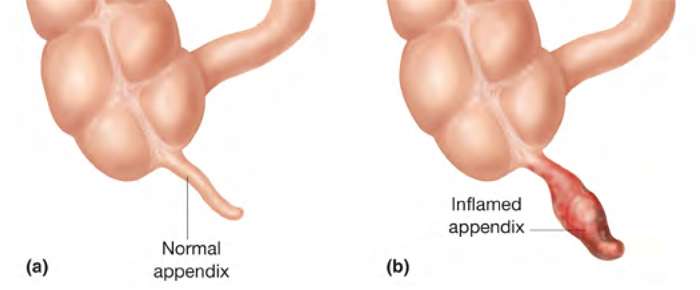
Figure 11. A visual representation of the structural changes that occur in the vermiform appendix during acute appendicitis. (Image retrieved from: https://www.pulse.ng/lifestyle/beauty-health/wellness-tips-5-symptoms-that-abdominal-pain-is-appendicitis-id5721321.html).
Treatment & Surgical Intervention
With regards to treating acute appendicitis, there are a couple of approaches that can be utilized. The first would be the non-surgical approach consisting of antibiotics coupled with percutaneous drainage of the appendix. Many antibiotics have shown to merit a certain degree of success towards improving the condition, such as penicillin, streptomycin, gentamicin, and cefotaximine. According to Eriksson and Granstrom (1995), the simultaneous prescription of cefotaximine and tinidazole has shown to yield a success rate of up to 95% in patients with uncomplicated acute appendicitis. If an abscess or accumulation of fluid is present in the appendix, periodically undergoing percutaneous abscess drainage in conjunction with antibiotics may be beneficial. This procedure involves the penetration of the appendix and associated structures superficial to it via a small needle under image screen guidance. If the condition is too severe or isn’t responding well to the non-surgical treatment, then surgical measures should be taken. The predominant surgical approach used to treat appendicitis in the past was an open appendectomy (OA). OA involves the practitioner performing a single muscle splitting gridiron incision over the right iliac fossa, followed by the removal of the infected appendix. Once the appendix is extracted, the remaining stump is surgically tucked into the cecum via a pouch or Lembert’s suture. If the appendix has already ruptured, an open appendectomy is typically required to disinfect the peritoneal cavity. If the appendix hasn’t ruptured, an alternative approach is a laparoscopic appendectomy (LA). This procedure involves the practitioners making one to four small incisions in the skin superficial to the right iliac fossa, followed by the insertion of a fiber-optic laparoscope to assist with the extraction of the appendix. According to the research, LA is the preferred procedure for most practitioners as it yields a number of advantages, such as: requiring less time to administer, allowing for a higher degree of precision during the extraction process and a lower degree of post-operative complications, less post-operative pain, and a shorter recovery period for the patient. A visual representation of the laparoscopic appendectomy procedure is provided in Figure 12.
![]()
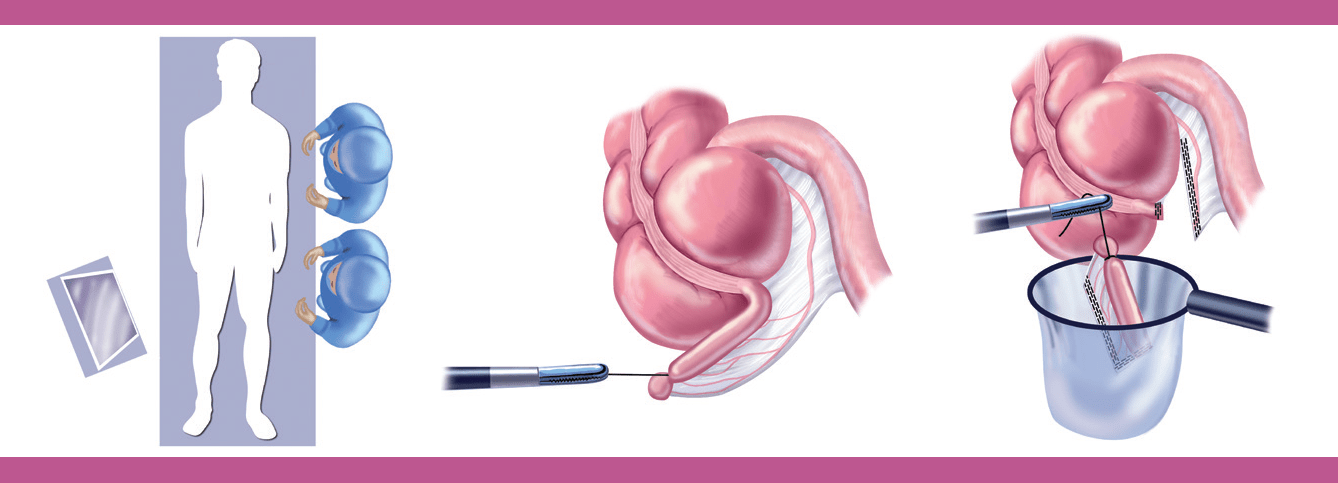
Figure 12. A visual representation of the laparoscopic appendectomy procedure. (Image retrieved from: https://www.sages.org/publications/patient-information/patient-information-for-laparoscopic-appendectomy-from-sages/).
References
Acute appendicitis Diagnosis and Treatment in 2009: Part I (2009). Retrieved from:http://www-lexisnexis-com.ezproxy.viu.ca/hottopics/lnacademic/?shr=t&csi=365633&sr=(%22Acute+Appendicitis+Diagnosis+Treatment+in+2009+Part+I%22)+and+date+is+2009
Ali, M. (2008). Laryngopharyngeal Reflux: Diagnosis and Treatment of a Controversial Disease. Current Opinion in Allergy and Clinical Immunology, 8(1), 28-33. doi: 10.1097/ACI.0b013e3282f3f44f
Appendicitis. (2018, July 6). Mayo Clinic. Retrieved from: https://www.mayoclinic.org/diseases-conditions/appendicitis/diagnosis-treatment/drc-20369549
Dever, J & Sheikh, M. (2015). Review Article: Spontaneous Bacterial Peritonitis – Bacteriology, Diagnosis, Treatment, Risk factors and Prevention. Alimentary Pharmacology & Therapeutics, 41(11), 1116-1131. doi:10.1111/apt.13172
Fundoplication Surgery for Gastroesophageal Reflux Disease (GERD). (2017, May 5). HealthLinkBC. Retrieved from: https://www.healthlinkbc.ca/health-topics/hw95701
Knight, J., Lively, M., Johnston, N., Dettmar, P & Koufman, J. (2005). Sensitive Pepsin Immunoassay for Detection of Laryngopharyngeal Reflux. The Laryngoscope, 115(8), 1473-1478. doi: https://doi.org/10.1097/01.mlg.0000172043.51871.d9
Laryngopharyngeal Reflux (LPR): Diagnosis and Tests (2018, September 20). Cleveland Clinic. Retrieved from: https://my.clevelandclinic.org/health/diseases/15024-laryngopharyngeal-reflux-lpr/diagnosis-and-tests
Lechien, J., Saussez, S & Karkos, P. (2018). Laryngopharyngeal Reflux Disease: Clinical Presentation, Diagnosis and Therapeutic Challenges in 2018. Current Opinion in Otolaryngology & Head and Neck Surgery, 26(6), 392-402. doi: 10.1097/MOO.0000000000000486
Marieb, E., Mallatt, J & Wilhelm, P. (2005). Human Anatomy, San Fransico, CA: Pearson Education, Inc.
Moore, K., Dalley, A & Agur, A. (1999). Clinically Oriented Anatomy. Baltimore, Maryland: Lippincott Williams & Wilkins.
Mowat, C & Stanley, A. (2001). Review Article: Spontaneous Bacterial Peritonitis–Diagnosis, Treatment and Prevention. Alimentary Pharmacology & Therapeutics, 15(12), 1851. Retrieved from: http://web.b.ebscohost.com.ezproxy.viu.ca/ehost/pdfviewer/pdfviewer?vid=1&sid=d96ad5e8-b866-48f7-9a8c-4606c7d307a4%40sessionmgr103
Peritonitis. (2018, June 6). Mayo Clinic. Retrieved from: https://www.mayoclinic.org/diseases-conditions/peritonitis/diagnosis-treatment/drc-20376250
Runyon, B., Akriviadis, E., Sattler, F & Cohen, J. (1991). Ascitic Fluid and Serum Cefotaxime and Desacetyl Cefotaxime Levels in Patients Treated for Bacterial Peritonitis. Digestive Diseases and Sciences, 36(12), 1782-1786. doi: https://doi.org/10.1007/BF01296625
Ryan, W. (2010). Appendicitis: Symptoms, Diagnosis, and Treatments. Retrieved from: http://web.b.ebscohost.com.ezproxy.viu.ca/ehost/ebookviewer/ebook/bmxlYmtfXzM2OTM4MF9fQU41?sid=3b20041f-5cce-40c2-be8c-d4e0f08b2c09@sessionmgr120&vid=0&format=EB&rid=1
Sataloff, R., Katz, P., & Hawkshaw, M. (2013). Reflux Laryngitis and Related Disorders: Clinical Reference Guide. San Diego: Plural Publishing, Inc.

