Thorax
Clinical Conditions
CLINICAL CONDITIONS OF THE MUSCULOSKELETAL SYSTEM
Costochondritis
Definition:
Costochondritis (kos-toe-kon-DRY-tis) is an inflammation of the cartilage that connects a rib to the sternum, or breastbone. Pain caused by costochondritis can mimic that of a heart attack or other heart conditions. It is also sometimes known as costosternal syndrome, costosternal chondrodynia or chest wall pain. Sometimes, swelling accompanies the pain and is known as Tietze syndrome.
Symptoms:
The pain associated with costochondritis usually:
- Occurs on the left side of your breastbone
- Is sharp, aching or pressure-like
- Affects more than one rib
- Worsens when you take a deep breath or cough
Causes:
Costochrondritis has no clear cause but theories include injury, physical strain, joint infection, tumours or arthritis.
Treatment:
While costochrondritis typically clears itself up, conservative treatments may be used. These conservative treatments include anti-inflammatory medications, narcotics, stretching and nerve stimulation. In extreme cases where conservative measures fail to provide relief, surgical interventions may be necessary.
Pectus Carinatum
Definition:
Pectus carinatum (PECK-tuss care-uh-NAW-tum) is a sternum and rib cartilage deformity that causes the chest to bow outward. It is sometimes called “pigeon chest” because the chest can look like a bird’s breast. The condition may cause pain.
Symptoms:
In pectus carinatum, the cartilage that holds the ribs to the breastbone pushes out from the chest, causing it to bow outward. Pectus carinatum may be noticeable in babies, but it often isn’t noticeable until a child gets older. The condition may get more obvious as a child grows, particularly during growth spurts. Pectus carinatum usually doesn’t cause any symptoms. When it does, pain is the most common symptom. This pain might occur when a child moves awkwardly, lies down the wrong way or is hit while playing sports.
Causes:
It is caused by a defect in the tough connective tissue (cartilage) that holds the ribs to the breastbone.
Treatment:
Common, conservative treatment for pectus carinatum include chest braces to reshape the child’s chest. Braces use constant, gentle pressure on the sternum to push the chest back into proper shape. Braces are generally required to be worn for a minimum of 12 hours a day, for at least a year. In extreme cases, surgical intervention may be required to reposition the sternum within the thorax.
Pectus Excavatum
Definition:
Pectus excavatum is a condition in which a person’s sternum is sunken into his or her chest. In severe cases, pectus excavatum can look as if the center of the chest has been scooped out, leaving a deep dent. While the sunken breastbone is often noticeable shortly after birth, the severity of pectus excavatum typically worsens during the adolescent growth spurt. Also called funnel chest, pectus excavatum is more common in boys than in girls. Severe cases of pectus excavatum can eventually interfere with the function of the heart and lungs. But even mild cases of pectus excavatum can make children feel self-conscious about their appearance.
Symptoms:
For many people with pectus excavatum, their only sign or symptom is a slight indentation in their chests. In some people, the depth of the indentation worsens in early adolescence and can continue to worsen into adulthood.
In severe cases of pectus excavatum, the breastbone may compress the lungs and heart. Signs and symptoms may include:
- Decreased exercise tolerance
- Rapid heartbeat or heart palpitations
- Recurrent respiratory infections
- Wheezing or coughing
- Chest pain
- Heart murmur
- Fatigue
Causes:
While the cause of pectus excavatum is unclear, it is thought to be an inherited condition, as it sometimes runs in families.
Treatment:
In people who exhibit mild signs and symptoms, physical therapy is often used as a treatment for pectus excavatum. Exercises that improve posture may increase the degree to which the chest can be expanded.
In people who exhibit moderate to severe symptoms, surgical intervention is often recommended. Small incision surgery inserts a curved metal bar to raise the depressed sternum into a more normal position. The metal bar is often removed after 2-3 years. Large incision surgery allows the surgeon to view the sternum directly. The deformed cartilage is removed and the sternum is repositioned using metal strut or mesh supports.
CLINICAL CONDITIONS OF THE CARDIOVASCULAR SYSTEM
Pericarditis
Description:
Pericarditis is a cardiovascular disorder that is characterized by the onset of inflammation in the pericardium of the heart in response to a number of factors, such as bacterial and viral infection, thoracic trauma, and previous poorly administered medical surgeries. This inflammatory response can mediate friction between the serous membranes of the pericardium and the over-accumulation of pericardial fluids. When left untreated, these structural changes can lead the formation of scar adhesions between the two membranes and an increase in intrapericardial pressure, which can impair the normal function of the heart.
Symptoms:
It is important to note that the symptom patterns exhibited by an individual with pericarditis will vary based on how long the condition has persisted for. Acute pericarditis typically lasts for less than three weeks. The condition is classified as incessant pericarditis when it persists for four to six weeks, but does not continue past three months. Once the condition persists for longer than three months, it is classified as chronic pericarditis. Typical symptoms exhibited across all three classifications include:
- Pericardial friction rub (“grinding”) audible cue
- Sharp, piercing chest pain deep to left side of the sternum (this pain typically intensifies during inspiration)
- Heart palpitations (the sensation of the heart losing or gaining an additional beat)
- Low-grade fever
- Coughing
- Sudden weakness or fatigue
- Swelling of the abdomen and/or lower extremities
Mechanism & Diagnosis:
The heart is circumvented by a three-layered sac-like structure known as the pericardium. The pericardium consists of a tough external layer that is composed of primarily dense connective tissue, known as the fibrous pericardium, and two delicate inner layers that are composed of serous membrane, known as the serous pericardium. Together, these layers work in conjunction with one another to: minimize motion of the heart by connecting it to the mediastinum, provide mechanical protection against external forces and infection, prevent overfilling of the heart, and reduce friction between the heart and surrounding structures during cardiovascular movements through the secretion of serous fluid.
In certain situations, an inflammatory response can be induced. The cause of this inflammatory response is determined to be idiopathic (unknown) among current medical research. But epidemiological speculation suggests that the inflammatory response is due to infection in the pericardial wall. According to the research, this infectious response could be accounted for through the presence of various microbial agents, such as tuberculous, systemic diseases, such as urema and neoplastic disease, autoimmune disorders such as lupus, scleroderma, and rheumatoid arthritis, thoracic trauma, and poorly administered medical procedures associated with the heart, such as cardiotomy. In addition to the layers of the pericardium becoming inflamed, the lining covering the serous layers will typically harden and develop a coarse texture. Upon contraction and relaxation of the heart, the two serous layers will rub against one another, producing a high-pitched, grinding sound that can easily be detected by palpating the left sternal border with a stethoscope. If pericaridits persists for a longer period of time, scar adhesions can form between the pericardial layers, limiting the hearts range of motion during the cardiac cycle. This condition is referred to as chronic constrictive pericarditis. A visual representation of the structural changes that occur in the pericardium during pericarditis are provided in Figure 1.
With regards to the diagnostic process, practitioners will typically utilize a variety of approaches to determine the underlying cause of the condition. Some of the modalities include: electrocardiograms (ECG), chest x-rays, echocardiogram, computerized tomography (CT), cardiac magnetic resonance imaging (MRI), and blood samples. Collectively, these modalities can provide practitioners with a visual representation of the structure of the pericardium, as well as insight into any abnormalities that are present in the blood stream, such as high levels of white blood cells, which would be indicative of infection.
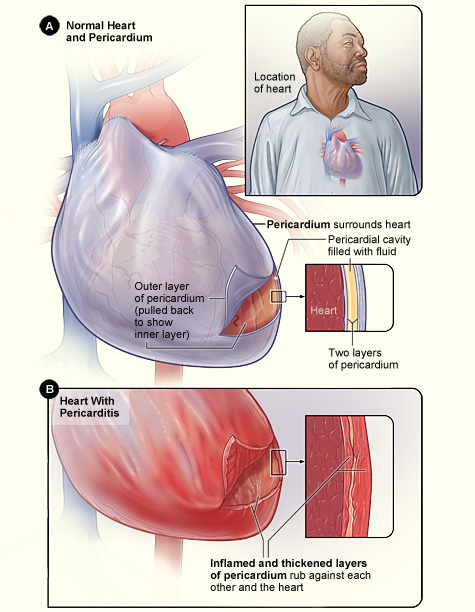
Figure 1. A visual representation of the structural changes that occur in the pericardium of the heart during pericarditis. (Image retrieved from: https://commons.wikimedia.org/wiki/Category:Pericarditis#/media/File:Pericarditis.jpg).
Treatment & Surgical Intervention:
Since the underlying etiology (cause) of pericarditis is determined to be either idiopathic or due to other factors such as systemic disease, poorly administered surgeries, and microbial agents, the underlying cause of the condition should be determined before further treatment is prescribed. For less severe acute pericarditis that is caused by microbial agents, prescribing the patient non-steroidal anti-inflammatory drugs (NSAIDs), such as aspirin and ibuprofen, colchicines, and antibiotics is an effective approach for alleviating the condition. Together, these will help reduce the local inflammatory response, kill off any bacteria that are residing in the pericardial region, and reduce that likelihood of recurrent or relapsing pericarditis from developing. According to Emazio et al (2005), the supplementation of colchicines with conventional treatment for acute pericarditis can lead to a significant reduction in the symptoms exhibited by patients during the initial 72 hours following the onset of pericarditis, and the reoccurrence of the condition over the following 18 months. In addition to this, the supplementation of colchincines has shown to reduce the likelihood of developing more severe conditions associated with pericarditis, such as cardiac tamponade and chronic pericardial constriction. In more severe scenarios where the etiology is a connective tissue disorder, an immune-mediated disease, or urema disease, the prescription of corticosteroids is recommended. Although the implementation of cortiocosteriods has shown to be effective of reducing local inflammation in the pericardial region, the research has also shown that it can increase one’s likelihood of developing relapsing pericarditis.
Cardiac Tamponade
Description:
Cardiac tamponade is a particular disorder that is characterized by the over accumulation of fluid or blood in the pericardial cavity, resulting in an increase in intrapericardial pressure that compresses the heart and causes functional alterations in its capacity to fill and eject blood to the rest of the body.
Symptoms:
- Fatigue and weakness
- Hypotension (low blood pressure)
- Pain that originates at the left side of the chest and eventually migrates to the neck, arms, and back
- Issues associated with inspiration
- Hyperventilation (a condition involving an imbalance in breathing, where one will typically exhale more than they inhale)
- Fainting, dizziness, nausea, and loss of consciousness
Mechanism & Diagnosis:
Under normal conditions, the serous membranes of the pericardium release serous fluid to help cushion the heart and reduce friction between the parietal and visceral layers of the pericardium, providing the heart with an adequate degree of motion during the cardiac cycle. But under certain conditions, an inflammatory response can be mediated in the pericardial wall. As a result of this response, excess pericardial fluid can be released from the serous membranes of the pericardium. This over accumulation of pericardial fluid is referred to as pericardial effusion. In conjunction with the onset of inflammation in the pericardium, an over accumulation of fluid in the pericardial cavity is among one of the more common characteristics exhibited by patients with cardiac tamponade. In some cases, blood will begin to fill the pericardial cavity as a result of thoracic trauma. This accumulation is known as hemocardium, and is drained under normal conditions to help regulate intrapericardial pressure. A visual representation of the accumulation of fluids in the pericardial cavity during cardiac tamponade is provided in Figure 2. But once pericardial pressure reaches a certain threshold, the body’s ability to drain this accumulation of blood and/or fluid becomes impaired. Since there is a limited amount of space available in the pericardial cavity, an increase in pericardial effusion will increase the intrapericardial pressure in the cavity. As a result of this increase in pressure, the chambers of the heart will become compressed. This will consequently compromise venous return, reduce the available ventricular volume, and inhibit optimal contractility within the walls of the ventricles. As a result of these characteristics, total end diastolic volume (EDV), or the total volume of blood present in the left and right ventricles following the ventricular filling phase (diastole), is reduced. This will consequently reduce stroke volume (volume of blood ejected following ventricular contraction) and total cardiac output (volume of blood ejected per minute) of the heart.
When referring to diagnosing this condition, practitioners look for a set of common symptom patterns known as Beck’s triad. The first component of the triad is hypotension and a low pulse pressure, which is attributed to the reduction in stroke volume and mean arterial pressure (MAP) associated with the condition. The second component is distension of the jugular veins, which is attributed to the reduction in the diastolic filling capacity of the right ventricle as a result of the increase in intrapericardial pressure. The final component of the triad is suppressed valvular sounds produced by the heart during the cardiac cycle, which is attributed to the accumulation of fluid and/or blood distorting the sounds.
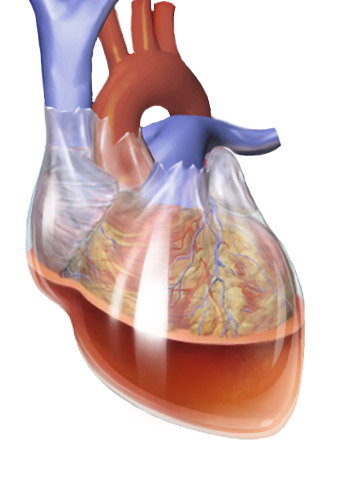
Figure 2. A visual representation of the fluid accumulation commonly exhibited in patients with cardiac tamponade. (Image retrieved from: https://commons.wikimedia.org/w/index.php?curid=31574265).
Treatment & Surgical Intervention:
Like pericarditis, treatment approaches to cardiac tamponade should be structured based on the level of severity exhibited by the condition. For patients that exhibit stable levels of pericardial effusion and/or hemocardium in the pericardial cavity, regular monitoring of the hemodynamic levels via a serial echocardiography coupled with treatment that targets the underlying etiology of the condition should be sufficient. Examples of conventional treatment can include: prescribing antibiotics if bacterial agents are the source of the infection, levothyroxine if hypothyroidism is the etiology (cause), and the prescription of non-steroidal anti-inflammatory drugs (NSAIDs) and colchicines to help alleviate the local inflammatory response. In more severe situations where the patient is exhibiting abnormal hemodynamics, immediate echocardiography-guided pericardiocentesis (EGP) or other means of surgical removal should be considered, such as pericardiectomy. EGP is the process in which practitioners utilize electrocardiography to locate the optimal site of penetration based on where the majority of the blood and/or fluid resides in the pericardium. Once the site of penetration is located, a hypodermic needle is inserted into the site to extract the accumulated blood and/or fluid. According to a study conducted by Tsang et al (2002), which involved the testers undergoing the EGP procedure in 977 different patients exhibiting symptoms of cardiac tamponade, the procedure resulted in an overall success rate of 97%, a total complication rate of 4%, and 1 procedural death out of the entire sample. If EGP is unable to evacuate the accumulated fluid in the pericardial cavity, then other alternatives should be considered, such as pericardiectomy, which involves practitioners surgically removing a section or the entirety of the pericardium. This approach should only be used as a final resort when no other treatment options are working.
Mitral Stenosis
Description:
Mitral stenosis is a particular condition that is characterized by the obstruction of blood flow to the left ventricle of the heart in response to structural alterations that occur in the bicuspid (mitral) valve that resides between the left atrium and left ventricle.
Symptoms:
Patients with mild cases of mitral stenosis don’t usually exhibit any apparent symptoms beyond the snapping sound detected via a stethoscope. Although there are a limited number of symptoms associated with mitral stenosis, the development of more severe conditions associated with the condition can be denoted through the following symptoms:
- Blue colouration of the fingertips and lips
- Spontaneous inflammation and weight gain
- Dizziness, fatigue, weakness, and difficulty inspiring and expiring
- Chronic coughing episodes
- Enlarged liver
- Distension of neck veins
- Hyperhydrosis (excessive sweat production)
- Abnormal growth patterns and loss of appetite in infants
Mechanism & Diagnosis:
During the diastolic portion of the cardiac cycle, blood that is received from the pulmonary circuit collects in the left atrium. Upon a change in the pressure between the left atrium and left ventricle, blood begins to drain into the left ventricle through means of gravitational force and atrial contraction. The structure that divides these two chambers is known as the left atrioventricular or bicuspid (mitral) valve. Under normal conditions, the change in pressure that occurs during atrial contraction (diastole) will cause the value to open, and close during ventricular contraction (systole). The general function of the mitral value is to promote proper blood flow to the left ventricle, and to prevent the back flow (atrioventricular reflux) of blood into the atrium during ventricular contraction. The contraction of the papillary muscles and support offered by the chordae tendineae prior to ventricular contraction are integral in preventing the mitral valve from prolapsing into the left atrium.
Under certain conditions, particularly in those that have experienced rheumatic fever, a disease that is characterized by inflammation of the heart, skin, joints, and brain, the mitral valve will begin to harden, narrow, and develop a certain degree of rigidly. In addition to this, other common physiological processes can occur, such as fusion of the commissures (borders separating leaflets) between the anterior and posterior leaflets of the valve and the fusion and shortening of the subchordal apparatus. This condition is known as mitral stenosis. A visual representation of mitral stenosis is provided in Figure 3. Other etiologies (causes) associated with mitral stenosis include: congenital deformities, such as parachute mitral valve and double orifice mitral valve, and systemic and local diseases such as lupus erythematosus, Fabry’s disease, and mucopolysaccridosis. Mitral stenosis has also shown to be a common condition among pregnant women, typically developing during the second trimester. A common feature with this condition can be depicted through the mitral valve losing its ability to open completely during atrial contraction. Since there is a smaller passageway for blood to travel through during mitral stenosis, a large degree of turbulence along with a pressure gradient is established. As this pressure gradient increases, the diastolic filling period for the left ventricle will decrease. As less blood is being drained into the left ventricle, the left atrium is required to exert more force during contraction to compensate for the loss in blood volume during ventricular filling. As one’s heart rate increases, a large degree stress can be placed on the left atrium, which can lead to the development of more severe conditions, such as the formation of blood clots, atrial fibrillation, and pulmonary congestion. A clinical condition that is often associated with mitral stenosis is mitral valve prolapse. This condition is characterized by the cusps of the mitral valve prolapsing into the left atrial chamber upon contraction of the left ventricle as a result of weakness or elongation developed in the collagen fibers of the cusps and chordate tendineae. This condition is particularly harmless, but can manifest into more severe health complications if left untreated.
When referring to the diagnostic process associated with mitral stenosis, practitioners can detect this condition through a physical examination and echocardiography. Since there is turbulence created upon blood flow between the left atrial and ventricular chambers, simply palpating the left side of the chest with a stethoscope can pick up on the distinct “snapping” (heart murmur) noise that occurs during early and late diastole. Echocardiography, which utilizes ultrasound to create accurate 3-dimensional images of bodily regions, can provide the practitioners with an accurate visual representation of the mitral valve. Electrocardiography and chest x-rays have also shown to be effective tools for detecting this condition.
![]()
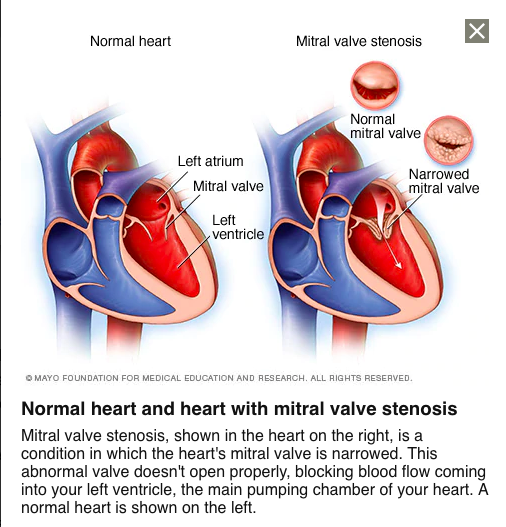
Figure 3. A visual representation of the changes in mitral valve structure between a normal, healthy heart and a heart affected by mitral stenosis. (Image retrieved from: https://www.mayoclinic.org/diseases-conditions/mitral-valve-stenosis/symptoms-causes/syc-20353159).
Treatment & Surgical Intervention:
With regards to treatment, mitral stenosis has been commonly approached through means of Percutaneous Balloon Valvuloplasty (PBV). PBV is an intricate procedure which involves the percutaneous insertion of one or more large balloons into the stenotic valve. Once the balloons are embedded in the stenotic valve, they are then inflated. This will reduce the obstruction created by the stenotic valves, improving blood flow and restoring normal function back in the mitral valve. A visual representation of the PBV procedure is provided in Figure 4. According to Vahanian and Palacios (2004), PBV has shown to exhibit a success rate of upwards of 95% among patients with non-calcified pulmonic, mitral, and aortic stenosis, and approximately 50-65% in patients that exhibit sub-optimal anatomy typically characterized by a large-degree of calcification, fibrotic accumulation, and subvalvular fusion in their mitral valve cusps. When PBV is deemed ineffective or unavailable to certain populations, other alternatives include: surgical repair or replacement. Surgical repair of the mitral valve typically involves cardiologists integrating synthetic tissue to help preserve the structural integrity of the mitral valve. The primary areas of repair typically consist of the chordate tendineae, mitral valve leaflets, and the fibrous ring surrounding the leaflets known as the mitral annulus. Mitral valve replacement typically involves the removal of the affected valve and the surgical implantation of mechanical (i.e., bi-leaflet and tilting disk valves) and biologic (i.e., values extracted from cadavers or synthesized from human tissue) prosthetic valves.
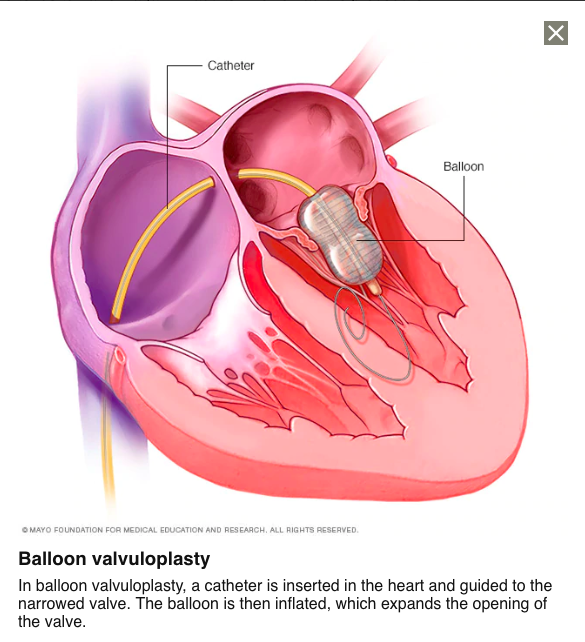
Figure 4. A visual representation of the Percutaneous Balloon Vulvoloplasty (PBV) procedure in a patient that is affected by a stenotic mitral valve. (Image retrieved from: https://www.mayoclinic.org/tests-procedures/valvuloplasty/pyc-20384961).
Bundle Branch Block
Description:
Bundle branch block is a particular medical condition that is characterized by damage to the bundle branches or fascicles of the conductive system of the heart, resulting in the disruption of impulse transmission across the heart, producing an asynchronous contraction cycle between the atrium and ventricle.
Symptoms:
Individuals with bundle branch blocks don’t typically exhibit many noticeable symptoms apart from the information that can be gathered from an electrocardiogram (ECG) and echocardiogram (echo). The summary of the symptoms that can be seen across patients inflicted with a bundle branch block includes:
- Loss of consciousness (syncope)
- The feeling experienced prior to losing consciousness (presyncope)
Mechanism & Diagnosis:
Under normal conditions, the atrial and ventricular chambers of the heart will contract and relax in a coordinated, rhythmical, and sequential manner as represented through electrocardiogram (ECG) patterns. The contraction and relaxation of these two chambers ultimately depends on the activity of the conducting system of the heart. The electrical transmission responsible for facilitating atrial and ventricular contraction originates at a dense collection of specialized muscle cells in the walls of the right atrium, known as the sinoatrial (SA) node. Being the pacemaker of the conducting system, the SA node is responsible for generating and propagating an electrical impulse throughout the walls of the right atrium, stimulating a contractile response in the left and right atrium (depolarization). Following this, the electrical impulse will then travel to the atrioventricular (AV) node, where the impulse will be temporarily delayed and relayed to the AV bundles. This brief delay is integral, as it provides a sufficient amount of filling time for the ventricles prior to contracting. After meeting with the AV bundle, the electrical impulse will then travel down a bifurcation of the AV bundles known as the left and right bundle branches. Travelling distally, there are a number of fascicles that correspond with the AV bundles. These fascicles will further branch off into a vast network of purkinje fibres, which are embedded in the ventricular myocardium from the apex to the base of the heart to ensure that the ventricles are being contracted from an inferior to superior basis.
In certain situations, the AV bundle branches and/or their corresponding fascicles can become damaged due to heart disease, myocardial infarction, and poorly performed surgical procedures. As a result of this damage, these specific structures will lose their ability to effectively relay electrical impulses to the purkinje fibers. In this situation, the body will naturally propagate the impulse along the muscle fibers situated in the walls of the heart, which will increase the amount of time it will take to stimulate ventricular contraction (depolarization). This extended period of transmission will cause ventricular depolarization to become prolonged, causing the ventricle to lose its contractile coordination with respect to the atrium. Since the ventricles will contract at a slower rate, the amount of time the heart beats per minute (heart rate) will be reduced, which will consequently diminish the heart’s capacity to relay blood to the tissues of the systemic circuit (cardiac output).
When referring to the diagnostic process, a bundle branch block can typically be detected through the usage of an electrocardiogram (ECG) and echocardiogram (echo). The electrocardiogram (ECG) will record and analyze the electrical activity of the heart. When referring to ECG patterns, a bundle branch block is usually depicted when the QRS complex exceeds 120ms in length. When referring to the specificity of the blockage, a left bundle branch block (LBBB) is generally illustrated through a broad QRS complex (> 120ms) in the presence of a sinus rhythm or related rhythms originating from above the ventricles, a deep and broad S-wave or rS between leads V1 and V2, a broad, notched (“M-shaped”) R-wave between I, aVL, and leads V5 and V6, and an absence of a Q-wave in leads V5 and V6. A right bundle branch block (RBBB) is generally denoted by a broad QRS complex (>120 ms) in the presence of normal sinus or related rhythms situated above the ventricles, a terminal R-wave or RSR complex in lead V1 and a R-complex proceeding a prolonged, shallow S-wave that occurs in I, aVL, and leads V5 and V6. Common ECG characteristics of both LBBB and RBBBs are provided in Figures 4 and 5. An echocardiogram (echo) can provide meaningful information regarding heart valve function, the thickness of the heart walls, and the location of the heart rhythm. With regards to a bundle branch block, the heart rhythm is typically detected above the ventricles.
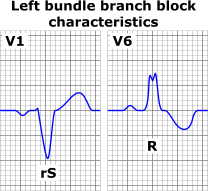

Figures 4 & 5. A visual representation of the ECG patterns that are commonly exhibited in individuals that have obstructions in their left or right bundle branches. (Image retrieved from: https://commons.wikimedia.org/wiki/File:Left_bundle_branch_block_ECG_characteristics.png; https://upload.wikimedia.org/wikipedia/commons/2/29/Right_bundle_branch_block_ECG_characteristics.png).
Treatment & Surgical Intervention:
It is important to note that if a patient that has been previously diagnosed with a bundle branch block is symptom-free, there is no real need to administer any treatment as there is a low probability that their condition will manifest into more severe complications. But if a bundle branch block develops in response to a heart condition, such as cardiac arrest, it could be a pre-cursor for the onset more serious medical conditions. In patients that are at risk for further health complications, the implementation of a pace maker is an effective option. A pacemaker is an intricate mechanism that possesses the dual ability to monitor the electrical activity of the heart, and elicit its own electrical impulses to stimulate ventricular and/or atrial contraction. A pacemaker is percutaneously implanted deep to the skin of the upper left chest. In the case of a patient with a bundle branch blockage, a single-chamber pacemaker is usually implemented. This specialized type of pacemaker circumvents the majority of the conductive circuit, sending the generated electrical impulse straight to the ventricular myocardium. This will help restore normal function to the conductive system of the heart, reducing the likelihood of associated medical complications from developing. Other variations of the pacemaker include: dual-chamber pacemaker, which relays electrical impulses to the right atrium and right ventricle, and the biventricular pacemaker, which relays electrical impulses between the right atrium, and the left and right ventricles. The biventricular pacemaker is often involved in a procedure known as cardiac resynchronization therapy, which is intended to promote contractile synchronization between the left and right ventricles in those that suffer from a bundle branch block or insufficient ventricular pumping capacity.
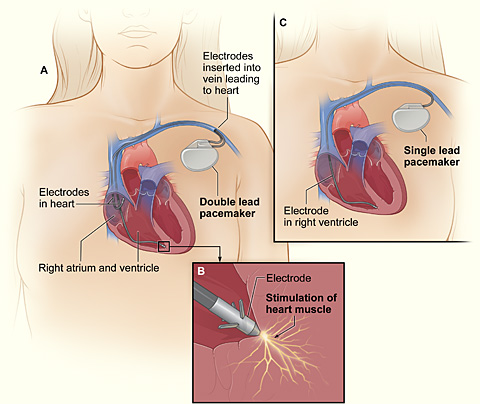
Figure 6. A visual representation of the general orientation and function of single-chamber and dual-chamber pacemakers. (Image retrieved from: https://commons.wikimedia.org/wiki/File:Pacemaker_NIH.jpg).
CLINICAL CONDITIONS OF THE RESPIRATORY SYSTEM
Pleurisy
Description:
Pleurisy (or pleuritis) is a clinical condition that is characterized by the onset of an inflammatory response in the parietal pleura of the pleural cavity as a result of exposure to a variety of different factors, such as viruses, bacteria, and genetic conditions. This inflammation can mediate an over-accumulation of pleural fluid, which can increase intrapleural pressure. If left untreated, this condition can severely degrade breathing mechanics.
Symptoms:
- Chest pain that can radiate to the shoulders and back and seems to exacerbate when one breathes, coughs, or sneezes.
- Reduced breathing capacity (due to the muscle guarding in the diaphragm that occurs as a result of the painful sensation in the chest)
- Joint and muscle soreness
- Headaches, fever, and loss of appetite
Mechanism & Diagnosis:
Like most bodily cavities, the pleural cavity, which encases the left and right lungs, is lined with parietal and visceral serosa. The parietal pleura is oriented on the inner walls of the thoracic cavity, the superior aspect of the diaphragm, and the lateral aspects of the mediastinum. The visceral pleura is continuous with the parietal pleura, and spans the entire outer surface of the left and right lungs. The visceral pleura also expands into the horizontal and/or oblique fissures of the lungs, covering the internal lobes. The potential space that separates the partial and visceral serosa is referred to as the pleural cavity. Collectively, the parietal and visceral pleura secrete a fluid known as pleural fluid. This fluid provides the dual function of reducing the friction between the parietal and visceral serosa during respiratory movements by providing lubrication, and maintaining adherence between the two pleura by creating surface tension between the two membranes.
Under certain conditions, the pleural cavity can become breached via secretions and particles migrating from different regions in the body and diffusing across lung tissue. These secretions and particles are often accompanied by various microbial invaders, such as viruses and bacteria. Upon detection of these pathogenic invaders, the body will mediate an immunological response to prevent further proliferation of the invaders, attempting to protect that respiratory tissue from serious structural damage. This conditioned is referred to as pleurisy or pleuritis. According to the literature, viral agents have shown to be the predominant etiology of pleurisy. Some of the more common viruses associated with pleurisy include: influenza, parainfluenza, coxsackieviruses, mumps, adenovirus, and the Epstein-Barr virus. Other etiologies (causes) associated with pleurisy include: asbestosis, inflammatory bowel disease (IBS), Familial Mediterranean Fever, sickle cell anaemia, tuberculous pleuritis, chronic renal failure, and Sjogren’s syndrome. In conjunction to the recruitment of white blood cells to combat the pathogenic agents, an inflammatory response is initiated in the parietal serosa. Instead of exhibiting a smooth, moist outer coating, the parietal pleura will develop a rough, coarse texture. Since the parietal and visceral pleura are located in such close proximity, the parietal pleura can rub against the delicate visceral plural (plural rub) during respiratory movements, producing a distinct “creaking” noise that can be detected via a stethoscope. In some cases, the parietal and visceral pleura can completely adhere to one another following an inflammatory response. This is commonly referred to as pleural adherence. In the situation of pleural effusion, which is an excess accumulation of fluid in the pleural cavity in response to inflammation, vessel damage, and/or leakage from lung capillaries, an enormous amount of pressure can build-up between the two membranes. This can elicit a sharp, painful sensation that is often coupled with impaired breathing mechanics. A visual representation of the structural changes that occur in the pulmonary pleurae and pleural cavity during pleurisy is provided in Figure 7.
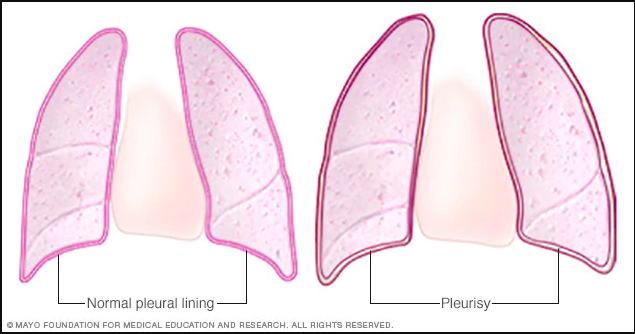
Figure 7. A visual representation of the structural changes that occur in the pulmonary pleurae and pleural cavity following the onset of pleurisy. (Image retrieved from: https://www.mayoclinic.org/-/media/kcms/gbs/patient-consumer/images/2013/08/26/10/10/ds00244-r7_pleurisythu_jpg.jpg).
With regards to the diagnostic process, there are a plethora of different tests that can be utilized for detecting pleurisy. All patients that are displaying any noticeable symptoms associated with pleurisy, such as chest pain and difficulty breathing, should be administered a radiography. Some different types of radiography include: chest x-rays and computerized tomography (CT) scans. Other useful instruments include: ultrasounds and electrocardiograms (ECG). Since there is plural effusion that commonly occurs during pleurisy, many of the screening instruments stated above are effective at locating the accumulation of fluid in the pleural cavity. Additionally, the “creeking” sound that is produced via friction between the parietal and visceral pleurae can also be easily detected through the usage of a stethoscope or ECG. In addition to these modalities, hemocrit, centrifusion, and stain and culture tests can also be performed. Collectively, these tests will provide a thorough analysis of blood composition, providing insight into any potential anaerobic infection, chylothorax, which is a common accumulation in plural effusion, and hemocrit concentrations in the blood. Thoracoscopy and thoracenteisis are also considered to be effective methods of diagnosing pleurisy. Thoracoscopy involves the incision of the chest and insertion of a small camera and stabilizer into the plural cavity to monitor the state of the region and/or collect tissue samples. Thoracentesis involves the injection of a local anaesthetic followed by the insertion of a needle between the ribs to extract accumulated fluid in the pleural cavity. A visual representation of these two procedures is provided in Figures 8 and 9 respectively. Together, these two procedures can provide meaningful information that can be further examined via lab analysis.
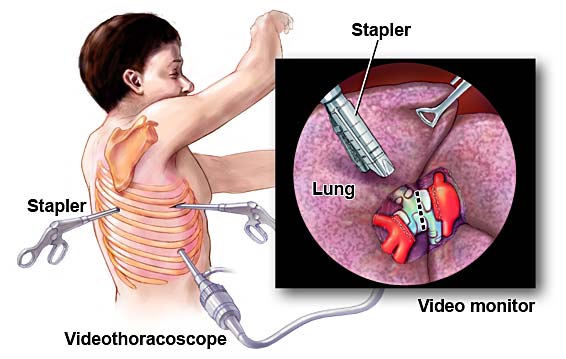
Figure 8. A visual representation of the thoracoscopy procedure. (Image retrieved from: https://www.medgurus.org/thoracoscopy-best-hospitals-doctors-cost-in-india/).
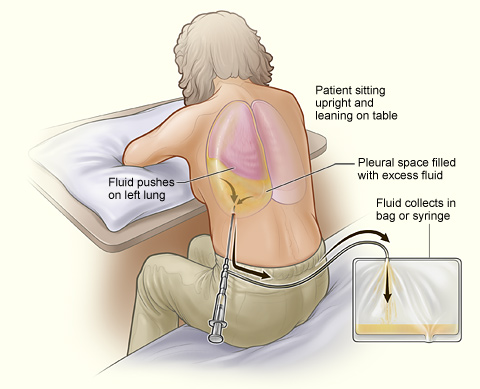
Figure 9. A visual representation of the thoracentesis procedure in a patient that has excess fluid accumulation in their pleural cavity. (Image retrieved from: https://commons.wikimedia.org/wiki/Category:Thoracentesis#/media/File:Thoracentesis.jpg).
Treatment & Surgical Intervention
With regards to treating pleurisy, it is important to first alleviate the symptoms associated with the condition, and then address the underlying cause(s) of the ailment. Since pleurisy is an inflammatory condition, the usage of non-steroidal anti-inflammatory drugs (NSAIDs), corticosteroids, and narcotic analgesics can be an effective option for reducing the inflammatory response in the parietal pleura and alleviating any pain associated with the condition. If bacteria are the suspected cause of the condition, prescribing antibiotics would be a prudent approach when trying to kill off the bacteria populations in the region. If the etiology (cause) is determined to be viral, the best approach would be to allow the body to go through its natural healing cycle while taking symptom-alleviating medications when needed, such as the options stated above.
Asthma:
Description: Asthma is a chronic inflammatory disease that causes the lungs and breathing passages to become sensitive, resulting in difficulty breathing.
Symptoms: Asthma can cause few or many symptoms, depending on the individual, and they can be mild to severe including:
- Shortness of breath
- A feeling of tightness in the chest
- Coughing
- Wheezing
- Bronchospasms (constriction of bronchioles)
A serious asthma emergency could result in the individual experiencing symptoms of struggling to breathe, inability to speak, pale, gray, clammy skin as well as blue lips or nail beds. The individual may also feel tired, experience difficulty speaking, and may faint if the symptoms are prolonged.
Causes: The cause of asthma is unknown, however there are various known triggers that can prompt an ‘asthma attack’. There are triggers that cause inflammation, and triggers that specifically promote the occurrence of a symptom. Inflammatory triggers could be dust mites, pets or other animals, moulds, pollen and air pollutants. Inflammation is also caused by viral infections and bacterial infections in the respiratory system. Triggers that elicit symptoms can be due to certain scents such as perfumes or colognes, smoke, chemical fumes, pollutants, cold air, and exercise. Another trigger for inflammation within the airways is intense emotions.
Treatment: There is no cure for asthma, however there are various medications that can reduce the symptoms an individual experiences. There are two main types of drug therapies that are effective: Rapid onset bronchodilators or ‘quick-relief’ and slow onset bronchodilators which gradually work over the course of 4 to 6 hours and are effective in reducing symptoms for up to 12 hours. These medications can either be administered through an inhaler or a nebulizer.
Bronchiectasis:
Description: This disease occurs in individuals that have experienced trauma to the bronchioles. The ciliated epithelial cells that line the airways become damaged, which in turn, reduces their ability to ‘sweep’ mucus and foreign particles up the respiratory tract. Without this function, bacteria, viruses or debris can become lodged in these areas and develop into infections, which are typically reoccurring. In children, it tends to affect boys more than girls, however as adults, 60% of individuals affected are female.
Symptoms:
- Chronic, persistent cough
- Shortness of breath
- Wheezing or whistling sound produced when breathing
- Increased sputum production
- Chest pain
- Fever
- Weakness
Severe symptoms can include coughing up blood or mucus that is bloody, as well as feeling fatigued, and having ‘clubbed’ fingers and toes. Weight loss may also be seen in individuals with this condition and in some cases respiratory failure can occur.
Causes: Bronchiectasis can develop due to cystic fibrosis, trauma to the bronchioles and ciliated epithelial cells, a physical object lodged in the respiratory tract, immunodeficiency disorders, pneumonia, tuberculosis, measles, fungal infections, or primary ciliary dyskinesia.
Treatment: Antibiotics and bronchodilators should be used to alleviate symptoms, as well as chest physical therapy. Corticosteroid therapy may also be used to reduce inflammation in the region. Supplemental oxygen may be necessary as well as medications that reduce and dilute mucus production and fluid may need to be drained from the lungs frequently. Surgery is another option, in which the segment of the respiratory tract that is damaged is removed.
Chronic Obstructive Pulmonary Disease (COPD):
Description: COPD is an umbrella term referring to various chronic diseases that result in the individual having a feeling of gradual and progressive breathlessness. These include bronchitis, emphysema, and refractory asthma. It is typically caused by the individual being a smoker, however there are multiple other reasons such as environmental factors and genetics that can influence their breathing capacity. This condition affects a population that is predominantly older than 40 years old and can have a debilitating effect on their quality of life down to impeding their daily routine activities.
Symptoms:
- Chronic cough lasting for multiple months
- Phlegm
- Audible distinctions in breathing such as a ‘whistling’ sound or wheezing
- Shortness of breath or inability to catch one’s breath
- Reoccurring and long-lasting lung infections
- Fatigue
- Unintentional weight loss
Causes: Smoking is the culprit behind 80-90% of COPD cases, however, environmental factors such as exposure to harsh chemicals and pollutants can also cause COPD as well as genetic factors such as alpha-1 antitrypsin deficiency and autoimmune diseases.
Treatment: There is not a cure for COPD, however lifestyle factors such as quitting smoking, and prescribed bronchodilators or anticholinergics may alleviate symptoms and improve the individual’s ability to breathe. The individual may also be prescribed antibiotics to assist with any accompanying infections they may develop as these individuals are more susceptible to respiratory infections. Corticosteroids may also help individuals cope with their COPD symptoms.
Figure 8: Animated COPD Patient. (2014). Understanding COPD. Retrieved from https://www.youtube.com/watch?time_continue=213&v=T1G9Rl65M-Q
Bronchitis:
Description: This condition can present itself following a viral or a bacterial infection of the respiratory tract. It is characterized by inflammation and often excessive mucus within the bronchi which impedes normal breathing. Acute bronchitis typically is resolved within 2-3 weeks, however, Chronic bronchitis lasts upwards of 3 months and is recurring for a minimum of two years.
Symptoms:
- Discolored mucus
- Difficulty breathing
- Fever
- Pain or discomfort in the chest
- Chills
Causes: Acute bronchitis is caused by infections of the respiratory tract, however the most prevalent cause for chronic bronchitis is smoking as well as exposure to hazardous airborne pollutants.
Treatment: In the majority of cases of acute bronchitis, the condition is provoked due to a viral infection and will resolve on its own within a few weeks. If the condition is due to a bacterial infection, antibiotics will be prescribed. For chronic bronchitis, bronchodilators or corticosteroids may be necessary as well as pulmonary rehabilitation.
Emphysema:
Description: This disease is differentiated by a decrease in the compliance or elasticity of the lungs reducing the capacity for which the individuals are able to take in air. The alveoli become weak, or are so damaged the walls within the air sacs rupture, resulting in a decrease in surface area for gas exchange to occur as well as a much smaller volume of air is taken in during inspiration. Expiration also experiences complications, as the alveoli have lost their elasticity, they are not able to return to their original form, so the expulsion of air from the air sac does not occur fully and in some cases doesn’t occur at all which results in a lack of oxygen-rich air to the system.
Symptoms:
- Breathlessness
- Increased mucus production
- Wheezing
- Reoccurring lung infections
- Difficulty breathing or fatigue caused by breathing and exertion
- In extreme cases, lips or nails may turn blueish or gray
Symptoms may not be present until after more than 50 percent of lung function is lost.
Causes: The most common cause of emphysema is smoking or being exposed to irritants such as harsh chemicals or pollutants, however alpha-1-antitrypsin deficiency can also cause this in rare cases.
Treatment: There is no cure for emphysema, however smoking cessation is essential. This, paired with bronchodilators, corticosteroids, and pulmonary rehabilitation therapy may reduce or alleviate symptoms. In some cases, supplemental oxygen may be recommended to increase low blood oxygen levels. If the emphysema is very severe, a surgical procedure called a lung volume reduction surgery may be suggested. This procedure removes segments of the lung that have lost all function due to irreparable damage.
Alpha-1 antitrypsin (AAT) deficiency:
Description: This condition is a genetically inherited disorder resulting in lung and liver disease. First signs usually occur between the ages 20 and 50 with substantial damage done to the alveoli, causing them to collapse or the alveolar walls rupture, resulting in emphysema.
Symptoms:
- Breathlessness
- Unprovoked weight loss
- Abnormal breathing and wheezing
- Rapid heartbeat
- ‘Hacking’ cough
- Barrel-shaped chest
- Jaundice
- Cirrhosis
- Swollen abdominopelvic region
- Panniculitis (rare) is a condition affecting the skin resulting in lumps and areas that are hardened and painful.
Causes: Neutrophil elastase is an enzyme secreted by white blood cells to combat infections, however it is prone to attacking healthy tissue, typically in the lungs and liver if not regulated by a protein called alpha-1 antitrypsin. Mutations in the SERPINA1 gene can cause an insufficient production of this protein and the neutrophil elastase causes damage to the delicate tissue of the alveoli. Individuals may also become deficient in alpha-1 antitrypsin if they have prolonged exposure to smoke and pollutants.
Treatment: Bronchodilators, supplemental oxygen and pulmonary rehabilitation all may improve the symptoms the individual experiences. Damaged lung tissue may need to be removed surgically.
Croup (Laryngitis):
Description: This condition presents itself as a swelling of primarily the larynx, but also the trachea and the bronchi, which results in labored breathing.
Symptoms:
- ‘Barking’ cough
- Hoarseness
- Difficulty breathing laying down
- Abnormal audible noises when breathing
- Fever
Causes: Croup is caused by a viral infection to the lower respiratory tract, typically from the influenza virus.
Treatment: Humidifiers may soothe irritated areas of the breathing passageways; however, symptoms tend to clear up on their own within a few days. If the condition does not improve, glucocorticoids or epinephrine may be administered to reduce the swelling in the airways.
Cystic Fibrosis:
Description: This is the most common fatal genetic disease that affects youth and young adults. It impedes normal lung function as well as digestive function with abnormal mucus production which results in excessively sticky mucus that clogs the airways of the lungs and digestive organs.
Symptoms:
Symptoms may not be present until adolescence or adulthood.
- Chronic cough
- Production of abnormally sticky, thick mucus
- Recurring respiratory infections
- Inflammation of the nasal mucous membranes
- Likely to develop pneumonia
- Nasal polyps
- Pneumothorax
- Respiratory failure
- Diabetes
- Obstruction of intestinal pathways and bile duct
- Breathlessness
- Unprovoked weight loss
- Wheezing
- Pancreatic damage and liver damage
- Infertility
Causes: A genetic mutation in the CTFR gene resulting in improper regulation of quantities of salt and water that moves in and out of the cells, which alters the way in which glands produce sweat and mucus.
Treatment: There is no cure for this disease, however chest physical therapy, anti-inflammatory medications, antibiotics, bronchodilators and medication to slow and thin the mucus production can aid the symptoms and improve quality of life.
Idiopathic Pulmonary Fibrosis (IPF):
Description: This disease is characterized by inflammation and scarring within the lungs which causes painful, labored breathing. It predominantly affects the interstitium, which is the tissue surrounding the alveoli.
Symptoms:
- Dyspnea
- Difficulty doing normal daily activities
- Cough that lasts longer than 8 weeks (can be a dry cough or accompanied by mucus)
- Pain in the chest when inhaling
- Unprovoked weight loss
- Fatigue
- ‘Clubbing’ in the fingers and toes
- Lack of appetite
Causes: The reason why people develop this is unclear, however familial predisposition, smoking, and issues with acid reflux have been common risk factors seen. It predominantly affects males and age seems to be a factor as most patients are greater than 50 years old.
Treatment: This condition has no cure, however medications such as Pirfenidone and Nintedanib may impede the progression of the disease from becoming more substantial. Supplemental oxygen, morphine, and pulmonary rehabilitation are common treatments for this disease. Lung transplants may be recommended if scarring is very severe.
Pertussis (Whooping Cough):
Description: This illness is caused by the Bordatella Pertussis bacteria and is very contagious. It can infect people of all ages but could prove to be fatal if an infant develops it. The bacteria attaches to the ciliated epithelial cells housed within the upper respiratory tract and produce secretions that damage the cilia and cause inflammation within the airway making it more difficult to breathe.
Symptoms:
- Violent coughing with a ‘whooping’ sound once all air has been expelled from lungs
- Fever
- Apnea
- Fatigue
- Vomiting after coughing fits
- Cyanosis
Causes: Transmission of this respiratory illness is due to a bacterial infection that is airborne. It can last up to 10 weeks.
Treatment: Antibiotics will be prescribed to get rid of the bacterial infection.
Lung Cancer:
Description: Malignancy targeting the epithelial cells around the airways, mucous glands and alveolar tissue. The primary cause of cancer-related deaths in the United States and Canada, affecting both genders. The most common types of lung cancer are:
- squamous cell carcinoma: approximately 25-30% of all cases, staring in the epithelium lining the bronchioles or bronchi, here masses are formed which end up becoming hollow and hemorrhaging.
- adenocarcinoma: begins in the peripheral lung as tumors developed from bronchial glands and alveoli. This is approximately 40% of the cases.
- small cell carcinoma: 20% of lung cancer cases: spherical clusters of growth originating in the primary bronchi and continuing to grow into the mediastinum. This type of cancer also creates hormones leading to more complications such as Cushing’s syndrome, or inappropriate ADH secretion or SIADH.
Symptoms:
- Pain in the chest
- Breathlessness
- Coughing or wheezing
- Unexplained weight loss
- Coughing that produces blood
- Chronic cough
- Fluid in the chest
- Metastasis (spreading of cancer to other parts of the body)
Symptoms are typically only present once tumors begin to restrict airflow or impede other structures.
Causes: Smoking is the primary cause (90% of all cases) of most lung cancer as well as exposure to toxic pollutants and harsh chemicals. This being said, not all lung cancer develops due to smoking, many individuals that develop it are not smokers. A familial history of lung cancer as well as exposure to radon gas, arsenic, chromium and nickel could result in this diseases’ development. People exposed to asbestos may also develop lung cancer.
Treatment: Radiation therapy, chemotherapy, or surgery may be the chosen treatment options, however depending on the stage and the region it is affecting, palliative care is necessary to improve the quality of life of the individual. Lung transplants are sometimes an option, depending on the state of the patient’s health.
Pneumonia:
Description: This is a respiratory infection that causes inflammation in the alveoli of the lungs. This may be accompanied by the air sacs becoming filled with fluid and/or pus. It can vary in severity from mild to life-threatening, however typically children and seniors are most at risk, as well as individuals with compromised immune systems.
Symptoms:
- Chest pain during normal breathing
- Coughing
- Confusion (typically seen in seniors over the age of 65)
- Sputum
- Chills
- Pleurisy (inflammation of the pleurae)
- Atelectasis (collapsed lung)
- Exhaustion
- Fever accompanied by perspiration and chills
- Nausea
- Breathlessness
- Vomiting or diarrhea
- Sometimes a lung abscess can occur.
Causes: There are 4 different classifications of pneumonia:
- Community-acquired pneumonia: most common form, can be transmitted through bacteria, such as streptococcus pneumoniae, which is the most common type of bacterial pneumonia, fungi, and viruses.
- Hospital-acquired pneumonia: may be more serious due to already compromised immune systems and the potential for antibiotic-resistant bacteria.
- Health care- acquired pneumonia: patients in long term care facilities which also have weakened immune systems and are exposed to potentially more resistant forms of bacteria.
- Aspiration pneumonia: occurs when food or beverages are inhaled into the lungs and can occur with vomit or saliva as well.
Treatment: Antibiotics, pain relievers and anti-inflammatory medication may be necessary to overcome this respiratory illness. If symptoms are severe hospitalization may be encouraged.
Pulmonary Embolism:
Description: When a thrombus or a blood clot formed in another part of the body (typically the deep veins of the legs and sometimes the arms) becomes dislodged, it can travel up through the venous system, through the heart and become wedged in the pulmonary arteries or the respiratory membrane. This occurrence is incredibly dangerous as it can result in completely cutting off the blood flow to the system and acute heart failure can occur depending on how much of the pulmonary circulation is obstructed. If it obstructs 50% of the circulation it is termed an acute minor pulmonary embolism. If the obstruction impedes more than 50% of pulmonary circulation, there is an increase in the right ventricular afterload as it is not used to resistance in the pulmonary system. Pulmonary hypertension and acute right heart failure occur. Tricuspid regurgitation results, compromising the right atrium and the left ventricle will not receive enough blood flow to supply to the system. This is called an acute massive pulmonary embolism. The final type of pulmonary embolism is called a subacute massive pulmonary embolism which is caused by multiple blood clots becoming dislodged and travelling to the lungs over a longer time frame. The slow obstruction in the lungs results in the right ventricle adapting to compensate for increased pressure needed to overcome the resistance. Pulmonary hypertension occurs and the obstructed lung tissue may die, called pulmonary infarction as a result of this type of pulmonary embolism.
Symptoms:
- Individuals may be asymptomatic
- Increase in blood pressure
- Sudden breathlessness
- Pain when trying to inhale or take deep breaths
- Coughing up blood
- Fainting
- Light-headedness
Causes:
- Trauma
- Autoimmune diseases
- Lifestyle factors: smoking, obesity, sedentary
- More common with progressed age
- Hormone therapy, pregnancy and various forms of birth control can cause blood clots
- Recent surgeries, especially involving hip or knee replacements and combined with bed rest
- Genetic predisposition
Treatment: Medications such as: Subcutaneous low molecular weight heparin, intravenous unfractionated heparin, or direct oral factor 10 inhibitors such as rivaroxaban or warfarin. Compression socks and lifestyle changes such as dietary improvements and being more active have also been beneficial for treatment.
Tuberculosis:
Description: The lungs become infected after Mycobacterium Tuberculosis bacteria colonize the airways, interstitial spaces, and the alveoli, affecting the respiratory system and the lymphatic system. It is estimated that 30% of people have tuberculosis, however it never develops into active tuberculosis and is termed latent tuberculosis, as they only are carriers. The infection is supressed by the immune system which contains it within calcified fibrous tubercles within the lungs. If the immune system becomes weakened, they may become infected and develop symptoms.
Symptoms:
- Pain in the chest
- Fever
- Sweating
- Chills
- Coughing (including coughing up blood and/or a ‘racking’ cough)
- Fatigue
- Weight loss
Causes: Mycobacterium Tuberculosis is spread as an airborne disease. Individuals with weakened immune systems are more likely to become sick if they are to come in contact with a contagious individual.
Treatment: Antibiotics must be taken for six to nine months, exactly as prescribed otherwise reinfections can occur with a drug-resistant type of tuberculosis. The most common medications are: isoniazid (INH), rifampin (RIF), ethambutol (EMB), and pyrazinamide (PZA).
Pulmonary alveolar proteinosis (PAP):
Definition: This rare condition is differentiated as pulmonary surfactant, produced by type II pneumocytes in the wall of the alveoli is created in large quantities and never removed. The surfactant accumulates and impedes oxygen rich air from entering the air sac.
Symptoms:
- Dyspnea
- Cyanosis (bluish color in the tips of the fingers)
- Cough (can be dry or with phlegm, blood may be present)
- ‘Clubbing’ of the toes and fingers
- Fatigue
- Unprovoked weight loss
- Pain in the chest
- Respiratory tract infections
- Respiratory Failure
Causes: This condition can be congenital, caused by a genetic mutation of the GM-CSF receptors on alveolar macrophages, resulting in the buildup of surfactant, however it can also be an autoimmune disorder, in which the body’s immune cells attack these receptors. This is common in smokers.
Treatment: Whole lung lavage (WLL) is a standard therapy for this condition in which saline water is used to wash out excess surfactant from one lung while the other receives pure oxygen, and then it is switched. This treatment needs to be done at a minimum of once a year, however depending on the severity, may be needed monthly.
Pneumoconiosis:
Definition: All lung diseases caused by the inhalation of dust into the lungs which, in turn, causes damage in the form of scarring after being deposited in the alveoli. Often referred to as “Black Lung Disease”.
Symptoms:
- Hypoxemia
- Chronic cough
- Tightness in the chest
- Breathlessness
- Fatigue
- Increased mucus production
- Decreased ability for exertion
Causes: Common mineral dusts cause scarring in lung tissue such as asbestos, beryllium, silica and dust from coal resulting in small nodules in the lungs which can eventually grow into large masses. Known as an “occupational disease”.
Treatment: bronchodilators, supplemental oxygen and chest rehabilitation are the most common treatments for this. Lung transplants or surgery to remove damaged areas are also sometimes considered.
Asbestosis:
Description: This chronic disease is characterized by scarring in the lungs that occurs over time from exposure to asbestos. The scar tissue forms around the alveoli, reducing the amount they are able to expand and fill with air.
Symptoms:
- Difficulty breathing
- ‘Crackling’ sound when breathing
- Pain in the chest
- Tightness in the chest
- Chronic dry cough
- Fluid in the lungs
- Weight loss
- Lack of appetite
- Fatigue
- Mesothelioma (Cancer of the mesothelium from exposure to asbestos)
In advanced cases of asbestosis, the individual may also experience pulmonary hypertension due to scar tissue stiffening the areas around arteries, and constricted blood flow. They may also have ‘clubbed toes or fingers’ or have deformities in the nail beds, due to a lack of oxygen to the extremities. Symptoms may occur many years after exposure to the source of asbestos, but the lung damage caused is permanent.
Causes: Asbestosis is a result of breathing in airborne microscopic fragments of asbestos, which causes long term, continuous damage to the alveoli, reducing the amount of oxygen and carbon dioxide that is exchanged.
Treatment: Options are palliative, as this condition progresses with time. Reducing exposure to the source of asbestos and refraining from smoking are the first steps in a treatment plan for asbestosis. Other effective options include utilizing humidifiers to loosen mucus in the lungs, supplemental oxygen, and medications such as bronchodilators and pain killers. A surgical procedure called thoracentesis may be recommended which drains excess fluids from the lungs and reduces some pressure, and in some cases, the option of a lung transplant is considered. Most patients with asbestosis undergo pulmonary rehabilitation programs in attempt to improve their quality of life.
Acute Respiratory Distress Syndrome (ARDS):
Definition: This condition is progressive and affects individuals with rapidly declining health by having fluid leak into the lungs, and build up in the alveoli. It typically occurs following trauma.
Symptoms:
- Severe shortness of breath
- Increased heart rate
- Coughing
- Fever
- Pain in the chest, especially during inhalation
- Blueish or gray fingertips
Causes: ARDS occurs following trauma such as severe infections, pneumonia, physical trauma to the lung itself perhaps following an accident, aspiration, breathing in harsh chemicals or smoke from a fire etc. Smokers and people with weakened immune systems are more likely to develop this.
Treatment: oxygen therapy through a ventilator and laying face down may help increase the chances of survival from ARDS, as well as diuretics to get rid of excess fluid. Extracorporeal membrane oxygenation (ECMO) is a high-risk option but sometimes efficient in stabilizing the body to allow the lungs a chance to heal.
Infant Respiratory Distress Syndrome:
Definition: A respiratory disorder affecting newborns born prematurely typically 6 weeks prior to their due date. Type II Pneumocytes, which are typically formed by the 28th week, create and secrete surfactant which prevents the lungs from collapsing by helping reduce surface tension. If the child is born before the 28th week, they will develop RDS, which sets in within the first 24 hours from birth.
Symptoms:
- Nostril flaring
- Apnea
- Grunting or straining during breathing
- Quick, shallow breathing
- Chest is pulled in between ribs and below ribs after each breath
Causes: The lack of surfactant production causes IRDS which can cause the lungs to collapse, and not enough oxygen to support the body as well as not being able to expel carbon dioxide.
Treatment: Effective treatment includes surfactant replacement therapy, supplemental oxygen and breathing support through a ventilator using a nasal continuous positive airway pressure (NCPAP) machine.
Pneumothorax:
Definition: This condition occurs when air is able to get into the region in between the lung and the chest wall, which causes the lung to collapse.
Symptoms:
- Breathlessness
- Sudden chest pain
Causes: Can be caused by a chest injury, lung diseases such as COPD, cystic fibrosis and pneumonia or mechanical ventilation issues in which the ventilator alters the pressure within the lung.
Treatment: If the lung did not completely collapse, sometimes waiting and monitoring the condition for it to re-expand is the best course of action. If the severity of the condition is great, a needle may be inserted into the lung to expel excess air. If neither of these options alleviate the symptoms and reinflate the lung, surgery may be required.
Pleural Effusion:
Definition: This disorder is characterized by an accumulation of fluid between the layers of the pleura.
Symptoms:
- Coughing
- Breathlessness
- Difficulty breathing laying down
- Chest pain
Causes: there are two subdivisions of pleural effusion:
- Trasudative (watery fluid): caused by heart failure, a pulmonary embolism, liver failure, or following a procedure such as open-heart surgery.
- Exudative (protein-rich fluid): is caused by pneumonia, cancer, pulmonary embolism, or renal failure.
- Tuberculosis, autoimmune disorders, trauma, asbestosis, and infections of the chest wall or abdomen can also cause pleural effusion.
Treatment: Depends on underlying condition, but diuretics and physical drainage of the lung through thoracentesis or thoracostomy are commonly effective for alleviating the symptoms of this condition.
References
Alkindi, F., El-Menyar, A., Al-Suwaidi, J., Patel, A., Gehani, A. A., Singh, R & Arabi, A. (2015). Left Bundle Branch Block in Acute Cardiac Events: Insights from a 23-year Registry. Angiology, 66(9), 811-817. doi:https://doi-org.ezproxy.viu.ca/10.1177/0003319714560223
Bolling, S., Li, S., O’Brien, S., Brennan, J., Prager, R & Gammie, J. S. (2010). Predictors of Mitral Valve Repair: Clinical and Surgeon Factors. Annals of Thoracic Surgery, 90(6), 1904-1912. doi:10.1016/j.athoracsur.2010.07.062
Bundle Branch Block. (2018, May 15). Mayo Clinic. Retrieved from: https://www.mayoclinic.org/diseases-conditions/bundle-branch-block/diagnosis-treatment/drc-20370518
Chandrashekhar, Y., Westaby, S & Narula, J. (2009). Mitral Stenosis. Lancet, 374(9697), 1271-1283. doi:10.1016/S0140-6736(09)60994-6
Imazio, M., Bobbio, M., Cecchi, E., Demarie, D., Demichelis, B., Pomari, F & Belli, R. (2005). Colchicine in Addition to Conventional Therapy for Acute Pericarditis: Results of the Colchicine for Acute Pericarditis (COPE) Trial. Circulation, 112(13), 2012-2016. doi: 10.1161/CIRCULATIONAHA.105.542738
Kass, S., Williams, P & Reamy, B. (2007). Pleurisy. American Family Physician, 75(9), 1357. Retrieved from: http://web.b.ebscohost.com.ezproxy.viu.ca/ehost/pdfviewer/pdfviewer?vid=1&sid=2fa14cd8-f7fe-4287-a959-3b3ca355d06f%40pdc-v-sessmgr02
Khandaker, M., Espinosa, R., Nishimura, R., Sinak, L., Hayes, S., Melduni, R & Oh, J. (2010). Pericardial Disease: Diagnosis and Management. Mayo Clinic Proceedings, 85(6), 572-593. doi:10.4065/mcp.2010.0046
Kim, J & Baggish, A. (2015). Electrocardiographic Right and Left Bundle Branch Block Patterns in Athletes: Prevalence, Pathology, and Clinical Significance. Journal of Electrocardiology, 48(3), 380-384. doi:10.1016/j.jelectrocard.2015.03.015
Marieb, E., Mallatt, J & Wilhelm, P. (2005). Human Anatomy, San Fransico, CA: Pearson Education, Inc.
Moore, K., Dalley, A & Agur, A. (1999). Clinically Oriented Anatomy. Baltimore, Maryland: Lippincott Williams & Wilkins.
Muller, D. W., Farivar, R. S., Jansz, P., Bae, R., Walters, D., Clarke, A & Shaw, M. (2017). Transcatheter Mitral Valve Replacement for Patients with Symptomatic Mitral Regurgitation: a Global Feasibility Trial. Journal of the American College of Cardiology, 69(4), 381-391. doi: 10.1016/j.jacc.2016.10.068
Nishimura, R., Holmes Jr, D & Reeder, G. (1990). Percutaneous Balloon Valvuloplasty. In Mayo Clinic Proceedings, 65(2), 198-220. doi:https://doi.org/10.1016/S0025-6196(12)65015-0
Pacemakers. (N.D.). National Heart, Lung, and Blood Institute. Retrieved from: https://www.nhlbi.nih.gov/health-topics/pacemakers
Pleurisy. (2018, February 6). Mayo Clinic. Retrieved from: https://www.mayoclinic.org/diseases-conditions/pleurisy/symptoms-causes/syc-20351863
Pleurisy. (2018, October 3). Healthline. Retrieved from: https://www.healthline.com/health/pleurisy
Tsang, T. S., Enriquez-Sarano, M., Freeman, W. K., Barnes, M. E., Sinak, L. J., Gersh, B. J & Seward, J. B. (2002). Consecutive 1127 Therapeutic Echocardiographically-Guided Pericardiocenteses: Clinical Profile, Practice Patterns, and Outcomes Spanning 21 Years. In Mayo Clinic Proceedings, 77(5), 429-436. Retrieved from: https://www.sciencedirect.com/science/article/abs/pii/S0025619611622118
Vahanian, A., & Palacios, I. F. (2004). Percutaneous Aapproaches to Valvular Disease. Circulation, 109(13), 1572-1579. Retrieved from: https://www.ahajournals.org/doi/abs/10.1161/01.CIR.0000124794.16806.E3
Alpha-1 Antitrypsin Deficiency. (n.d.). Retrieved from https://www.nhlbi.nih.gov/health-topics/alpha-1-antitrypsin-deficiency
Alpha-1 antitrypsin deficiency – Genetics Home Reference – NIH. (n.d.). Retrieved from https://ghr.nlm.nih.gov/condition/alpha-1-antitrypsin-deficiency
Asbestosis. (n.d.). Retrieved from https://www.lung.ca/lung-health/asbestos
Asbestosis in Lungs: Symptoms, Causes & Treatment Options. (n.d.). Retrieved from https://www.asbestos.com/asbestosis/
Asbestosis Treatment | Reducing Discomfort & Risk of Infections. (n.d.). Retrieved from https://www.asbestosnetwork.com/Health-Issues/Asbestosis/Asbestosis-Treatment.shtml
Asthma Treatment. (2017, March 20). Retrieved from https://acaai.org/asthma/asthma-treatment
Bronchiectasis. (n.d.). Retrieved from https://www.nhlbi.nih.gov/health-topics/bronchiectasis
Bronchitis. (2017, April 11). Retrieved from https://www.mayoclinic.org/diseases-conditions/bronchitis/symptoms-causes/syc-20355566
Common Asthma Triggers. (n.d.). Retrieved from https://www.asthma.ca/get-help/asthma-3/triggers-3/common-asthma-triggers/
COPD (Chronic Obstructive Pulmonary Disease). (n.d.). Retrieved from https://www.physio-pedia.com/COPD_(Chronic_Obstructive_Pulmonary_Disease)
COPD Symptoms. (n.d.). Retrieved from https://www.lung.org/lung-health-and-diseases/lung-disease-lookup/copd/symptoms-causes-risk-factors/symptoms.html
Croup. (n.d.). Retrieved from https://www.healthlinkbc.ca/health-topics/hw31906
Cystic Fibrosis. (n.d.). Retrieved from https://www.lung.ca/lung-health/lung-disease/cystic-fibrosis
Cystic Fibrosis. (n.d.). Retrieved from https://www.nhlbi.nih.gov/health-topics/cystic-fibrosis
Emphysema. (n.d.). Retrieved from https://www.physio-pedia.com/Emphysema
Emphysema. (n.d.). Retrieved from https://www.healthlinkbc.ca/health-topics/zm2394
Emphysema. (2017, April 28). Retrieved from https://www.mayoclinic.org/diseases-conditions/emphysema/symptoms-causes/syc-20355555
How is PE Diagnosed? (n.d.). Retrieved from https://www.stoptheclot.org/learn_more/signs-and-symptoms-of-blood-clots/how_is_pe_diagnosed/
Learn About ARDS. (n.d.). Retrieved from https://www.lung.org/lung-health-and-diseases/lung-disease-lookup/ards/learn-about-ards.html
Lung cancer. (2018, November 16). Retrieved from https://www.mayoclinic.org/diseases-conditions/lung-cancer/symptoms-causes/syc-20374620
Lung Diseases. (n.d.). Retrieved from https://www.lung.org/lung-health-and-diseases/all-diseases.html
MacGill, M. (2017, December 13). Emphysema: Symptoms, treatment, and causes. Retrieved from https://www.medicalnewstoday.com/articles/8934.php
Marieb, E. N., & Hoehn, K. (2013). Human anatomy & physiology. Hoboken, NJ: Pearson Education.
Martini, F., Nath, J. L., Bartholomew, E. F., Ober, W. C., Ober, C. E., Welch, K., & Hutchings, R. T. (2018). Fundamentals of anatomy & physiology. New York: Pearson.
Pertussis (Whooping Cough). (2017, August 07). Retrieved from https://www.cdc.gov/pertussis/index.html
Pneumoconiosis Symptoms, Causes and Risk Factors. (n.d.). Retrieved from https://www.lung.org/lung-health-and-diseases/lung-disease-lookup/pneumoconiosis/pneumoconiosis-symptoms-causes-risks.html
Pneumonia. (2018, March 13). Retrieved from https://www.mayoclinic.org/diseases-conditions/pneumonia/symptoms-causes/syc-20354204
Pneumothorax. (2017, August 04). Retrieved from https://www.mayoclinic.org/diseases-conditions/pneumothorax/symptoms-causes/syc-20350367
Pulmonary Alveolar Proteinosis. (n.d.). Retrieved from https://rarediseases.org/rare-diseases/pulmonary-alveolar-proteinosis/
Respiratory Distress Syndrome. (n.d.). Retrieved from https://www.nhlbi.nih.gov/health-topics/respiratory-distress-syndrome
Smith, M. P. (2017, June 19). Diagnosis and management of bronchiectasis. Retrieved from http://www.cmaj.ca/content/189/24/E828
Tuberculosis. (2018, January 04). Retrieved from https://www.mayoclinic.org/diseases-conditions/tuberculosis/symptoms-causes/syc-20351250
Tuberculosis (TB). (2018, August 17). Retrieved from https://www.cdc.gov/tb/topic/treatment/tbdisease.htm
Understanding PF. (n.d.). Retrieved from https://cpff.ca/understanding-pf/what-is-pulmonary-fibrosis/
What is COPD? | Signs and Symptoms. (n.d.). Retrieved from https://www.copdfoundation.org/What-is-COPD/Understanding-COPD/What-is-COPD.aspx

